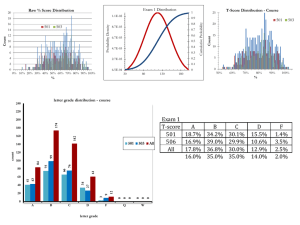
Modeling and Analysis of Entropy Generation in Light
... Some comments on condition d) are in order. The thermal equilibrium among species in the system is reached if one assumes that electrons in the conduction band do not have enough energy to produce effects such as tunneling and others, but they only stay in the conduction band and then recombine with ...
... Some comments on condition d) are in order. The thermal equilibrium among species in the system is reached if one assumes that electrons in the conduction band do not have enough energy to produce effects such as tunneling and others, but they only stay in the conduction band and then recombine with ...
Energy
... • Isothermal change: Such a change takes place at a constant temperature. • The constancy in the temperature is maintained while changes occur because there is a thermal contact between the system and the surrounding, allowing heat flow in such a way that the heat gain by the system equals the heat ...
... • Isothermal change: Such a change takes place at a constant temperature. • The constancy in the temperature is maintained while changes occur because there is a thermal contact between the system and the surrounding, allowing heat flow in such a way that the heat gain by the system equals the heat ...
EGR 107 FALL 2001
... and a pressure of 120kPa. The air is compressed and heat is removed for an isobaric process until the volume is reduced to one third of its original volume. Next the air is heated at constant volume. In the third process, the air is expanded in a reversible, adiabatic process in which Pv C to the ...
... and a pressure of 120kPa. The air is compressed and heat is removed for an isobaric process until the volume is reduced to one third of its original volume. Next the air is heated at constant volume. In the third process, the air is expanded in a reversible, adiabatic process in which Pv C to the ...
Query on Negative Temperature, Internal
... This finds the probability wn of a state of the whole system such that the body concerned is in some definite quantum state (with energy E n ), i.e., a microscopically defined state , and is suitable that the system is assumed to be in equilibrium [2]. So long as assume that the Gibbs distribution h ...
... This finds the probability wn of a state of the whole system such that the body concerned is in some definite quantum state (with energy E n ), i.e., a microscopically defined state , and is suitable that the system is assumed to be in equilibrium [2]. So long as assume that the Gibbs distribution h ...
Thermochemistry
... 1.Introduction to Energy changes Energy is the capacity to do work. There are many/various forms of energy like heat, electric, mechanical, and/ or chemical energy.There are two types of energy: (i)Kinetic Energy(KE) ;the energy in motion. (ii)Potential Energy(PE); the stored/internal energy. Energ ...
... 1.Introduction to Energy changes Energy is the capacity to do work. There are many/various forms of energy like heat, electric, mechanical, and/ or chemical energy.There are two types of energy: (i)Kinetic Energy(KE) ;the energy in motion. (ii)Potential Energy(PE); the stored/internal energy. Energ ...
2. The Thermopile
... The relative Seebeck emf produced in a thermoelectric circuit (RSE) has no relationship with contact potential, or Volta effect [Jastrzebski, 1976; Bridgman, 1934]. Contact potential is measured by the difference in work functions when two different metals are brought sufficiently close so that elec ...
... The relative Seebeck emf produced in a thermoelectric circuit (RSE) has no relationship with contact potential, or Volta effect [Jastrzebski, 1976; Bridgman, 1934]. Contact potential is measured by the difference in work functions when two different metals are brought sufficiently close so that elec ...
Atmospheric Thermodynamics
... introduction into the principles of atmospheric thermodynamics, and to present a “handy” overview and derivation of the quantities used in numerical weather prediction The material presented is kept to a minimum, and focuses on the concept of enthalpy of moist air. This should allow the reader to el ...
... introduction into the principles of atmospheric thermodynamics, and to present a “handy” overview and derivation of the quantities used in numerical weather prediction The material presented is kept to a minimum, and focuses on the concept of enthalpy of moist air. This should allow the reader to el ...
4. Two-level systems - Theoretical Physics
... the ground state, this implies a small heat capacity. As the temperature then approaches the scale temperature it is easy to excite the particles and you get a large heat capacity. At higher temperatures we have essentially the same number of particles in the levels and that situation does not chang ...
... the ground state, this implies a small heat capacity. As the temperature then approaches the scale temperature it is easy to excite the particles and you get a large heat capacity. At higher temperatures we have essentially the same number of particles in the levels and that situation does not chang ...
Review of Chemical Thermodynamics 7.51 September 1999 ∆G
... Thermodynamics allows us to predict how chemical reactions will change as a function of temperature and how changes in the structure of molecules might affect the equilibrium properties of a population of these molecules. There are four basic thermodynamic properties: ∆G — Change in free energy betw ...
... Thermodynamics allows us to predict how chemical reactions will change as a function of temperature and how changes in the structure of molecules might affect the equilibrium properties of a population of these molecules. There are four basic thermodynamic properties: ∆G — Change in free energy betw ...
Lecture Notes
... where dQ is the energy required to produce a temperature change equal to dT. Heat capacity is not an intrinsic property i.e. total heat a material can absorb depends on its volume / mass. Hence another parameter called specific heat, c, it defined as heat capacity per unit mass (J/kg-K, Cal/kg-K). ...
... where dQ is the energy required to produce a temperature change equal to dT. Heat capacity is not an intrinsic property i.e. total heat a material can absorb depends on its volume / mass. Hence another parameter called specific heat, c, it defined as heat capacity per unit mass (J/kg-K, Cal/kg-K). ...
Chapter 10: Entropy and the Second Law of Thermodynamics
... since work is a process that involves moving random motions into more ordered ones. ...
... since work is a process that involves moving random motions into more ordered ones. ...
Thermochemistry Thermochemistry
... • All systems will contain energy – In thermodynamics we are interested in the flow of energy, particularly in the forms of heat and work. – Note that heat and work occur when there is a process. They only exist when something happens. • The system has energy, (often described as the capacity to do ...
... • All systems will contain energy – In thermodynamics we are interested in the flow of energy, particularly in the forms of heat and work. – Note that heat and work occur when there is a process. They only exist when something happens. • The system has energy, (often described as the capacity to do ...
Chapter-9-Handouts
... Internal Energy (E): The capacity to do work or to produce heat. Temperature (T): How hot or cold an object is. Heat (q): The energy that is transferred as a result of a temperature difference between a system and its surroundings. ...
... Internal Energy (E): The capacity to do work or to produce heat. Temperature (T): How hot or cold an object is. Heat (q): The energy that is transferred as a result of a temperature difference between a system and its surroundings. ...
Heat

In physics, heat is energy in a process of transfer between a system and its surroundings, other than as work or with the transfer of matter. When there is a suitable physical pathway, heat flows from a hotter body to a colder one. The pathway can be direct, as in conduction and radiation, or indirect, as in convective circulation.Because it refers to a process of transfer between two systems, the system of interest, and its surroundings considered as a system, heat is not a state or property of a single system. If heat transfer is slow and continuous, so that the temperature of the system of interest remains well defined, it can sometimes be described by a process function.Kinetic theory explains heat as a macroscopic manifestation of the motions and interactions of microscopic constituents such as molecules and photons.In calorimetry, sensible heat is defined with respect to a specific chosen state variable of the system, such as pressure or volume. Sensible heat transferred into or out of the system under study causes change of temperature while leaving the chosen state variable unchanged. Heat transfer that occurs with the system at constant temperature and that does change that particular state variable is called latent heat with respect to that variable. For infinitesimal changes, the total incremental heat transfer is then the sum of the latent and sensible heat increments. This is a basic paradigm for thermodynamics, and was important in the historical development of the subject.The quantity of energy transferred as heat is a scalar expressed in an energy unit such as the joule (J) (SI), with a sign that is customarily positive when a transfer adds to the energy of a system. It can be measured by calorimetry, or determined by calculations based on other quantities, relying on the first law of thermodynamics.