Entropy. Temperature. Chemical Potential
... we view the closed system as composed of coupled subsystems (all macroscopic), then by definition these equilibria are obtained via an exchange of energy (U ), volume (V ) and particles (N ) among the subsystems. Equilibrium corresponds to the maximum value of the total entropy as a function of thes ...
... we view the closed system as composed of coupled subsystems (all macroscopic), then by definition these equilibria are obtained via an exchange of energy (U ), volume (V ) and particles (N ) among the subsystems. Equilibrium corresponds to the maximum value of the total entropy as a function of thes ...
Lecture 20_Review
... When you measure the weight of objects using scales, the buoyant force should be accounted for. Suppose you are standing on a weighing scale and suddenly all the atmosphere vanished. The reading on the scale would A) Increase B) Decrease C)Remain the same D)Quickly reduce to zero Answer: A In the p ...
... When you measure the weight of objects using scales, the buoyant force should be accounted for. Suppose you are standing on a weighing scale and suddenly all the atmosphere vanished. The reading on the scale would A) Increase B) Decrease C)Remain the same D)Quickly reduce to zero Answer: A In the p ...
Modeling and control of O2/CO2 gas turbine cycle for CO2 capture
... ing fluid in the gas turbine. We will investigate dynamics and control of this semi-closed O2 /CO2 gas turbine cycle, where CO2 capture is achieved since some CO2 must be removed from the cycle to avoid accumulation. In particular, we look at the design of a predictive controller that aims at achiev ...
... ing fluid in the gas turbine. We will investigate dynamics and control of this semi-closed O2 /CO2 gas turbine cycle, where CO2 capture is achieved since some CO2 must be removed from the cycle to avoid accumulation. In particular, we look at the design of a predictive controller that aims at achiev ...
Chapter 5 13edx
... out in a sealed “bomb” such as this one. • The heat absorbed (or released) by the water is a very good approximation of the enthalpy change for the reaction. • qrxn = – Ccal × ∆T Thermochemistry © 2015 Pearson Education, Inc. ...
... out in a sealed “bomb” such as this one. • The heat absorbed (or released) by the water is a very good approximation of the enthalpy change for the reaction. • qrxn = – Ccal × ∆T Thermochemistry © 2015 Pearson Education, Inc. ...
Thermal Diffusion and Partial Molar Enthalpy Variations of n
... This value is not the same as the result in eq 13.26 2.2. Fluxes and Forces from Nonequilibrium Thermodynamics. Consider next the butane adsorbed in the zeolite phase. The amount of zeolite is constant in the system, and it is taken as frame of reference (thus, its mass flux is zero). We are interes ...
... This value is not the same as the result in eq 13.26 2.2. Fluxes and Forces from Nonequilibrium Thermodynamics. Consider next the butane adsorbed in the zeolite phase. The amount of zeolite is constant in the system, and it is taken as frame of reference (thus, its mass flux is zero). We are interes ...
Abstraction and its Limits: Finding Space For Novel Explanation
... The toy theory is also misleading in another way; there is an obvious sense in which the transformed variables of (7)-(9) are the wrong variables in which to describe basic mechanics. The example would be more telling, and more interesting, if our new variables themselves lead to successful abstract ...
... The toy theory is also misleading in another way; there is an obvious sense in which the transformed variables of (7)-(9) are the wrong variables in which to describe basic mechanics. The example would be more telling, and more interesting, if our new variables themselves lead to successful abstract ...
2 - PSU MNE
... This chapter is devoted to a brief review of materials thermodynamics, with emphasis on chemical equilibrium. It is intended to serve as the basis of the many applications of thermodynamics that will be encountered in subsequent chapters. ...
... This chapter is devoted to a brief review of materials thermodynamics, with emphasis on chemical equilibrium. It is intended to serve as the basis of the many applications of thermodynamics that will be encountered in subsequent chapters. ...
Midterm 2: Tue Nov 15 (Chs 11, 13, 14, 15, 19, 20)
... A)The density of a large body of liquid (e.g. ocean) remains about constant throughout its volume B) The density of a large body of gas (e.g. our atmosphere) remains about constant throughout. C) The pressure in the ocean remains about constant throughout its volume D) The pressure in the atmosphere ...
... A)The density of a large body of liquid (e.g. ocean) remains about constant throughout its volume B) The density of a large body of gas (e.g. our atmosphere) remains about constant throughout. C) The pressure in the ocean remains about constant throughout its volume D) The pressure in the atmosphere ...
thermochemistry
... out in a sealed “bomb” such as this one. • The heat absorbed (or released) by the water is a very good approximation of the enthalpy change for the reaction. • qrxn = – Ccal × ∆T Thermochemistry © 2015 Pearson Education, Inc. ...
... out in a sealed “bomb” such as this one. • The heat absorbed (or released) by the water is a very good approximation of the enthalpy change for the reaction. • qrxn = – Ccal × ∆T Thermochemistry © 2015 Pearson Education, Inc. ...
Magnetoconvection in HCLL Blankets
... Density changes due to temperature variations, which cause convective motions, are described by the Boussinesq approximation, which states that the fluid density is a linear function of temperature in the gravitational body force term, ρβ(T − T0 )g, and constant elsewhere. Here ρ is the density at t ...
... Density changes due to temperature variations, which cause convective motions, are described by the Boussinesq approximation, which states that the fluid density is a linear function of temperature in the gravitational body force term, ρβ(T − T0 )g, and constant elsewhere. Here ρ is the density at t ...
Taylor blast wave paper I
... The propagation and decay of a blast wave in air has been studied for the case when the maximum excess over atmospheric pressure does not exceed 2atm. At great distances R from the explosion centre the pressure excess decays as in a sound wave proportionally to R-l. At points nearer to the centre it ...
... The propagation and decay of a blast wave in air has been studied for the case when the maximum excess over atmospheric pressure does not exceed 2atm. At great distances R from the explosion centre the pressure excess decays as in a sound wave proportionally to R-l. At points nearer to the centre it ...
Sub Module 2.12 Measurement of vacuum
... manometers, Bourdon gages for the measurement of vacuum as long as it is not high vacuum. In the case of the U tube with mercury as the manometer liquid we can practically go all the way down to -760 mm mercury but there is no way of using it to measure vacuum pressures involving less than a fractio ...
... manometers, Bourdon gages for the measurement of vacuum as long as it is not high vacuum. In the case of the U tube with mercury as the manometer liquid we can practically go all the way down to -760 mm mercury but there is no way of using it to measure vacuum pressures involving less than a fractio ...
Thermodynamics Institute of Lifelong Learning, University of Delhi
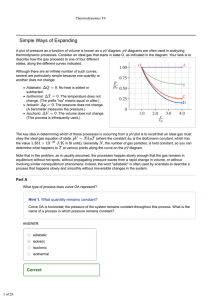
... Path: The sequence of steps taken by a system during a thermodynamic process starting from the initial state, through an intermediate state to the final state is known as a path. The path can consist of a single or more steps. Classification of Processes Based upon Path Depending upon the path follo ...
... Path: The sequence of steps taken by a system during a thermodynamic process starting from the initial state, through an intermediate state to the final state is known as a path. The path can consist of a single or more steps. Classification of Processes Based upon Path Depending upon the path follo ...
Energetics and Equilibria
... (such a Newton's Laws) it cannot be "proved". The law really is a statement about experience ; the "proof", such that it is, is that nobody has ever observed an event which contradicts the Second Law. All laws have their limitations – for example Newton's Laws do not apply to individual particles, l ...
... (such a Newton's Laws) it cannot be "proved". The law really is a statement about experience ; the "proof", such that it is, is that nobody has ever observed an event which contradicts the Second Law. All laws have their limitations – for example Newton's Laws do not apply to individual particles, l ...