Chapter I - Introduction: The Atom
... They are also representative of all elementary atomic particles in that they differ from one another physically because of the way mass-energy is distributed within the atom. All atoms are put together in similar ways. Mass in all atoms is distributed among three elementary particles called the pro ...
... They are also representative of all elementary atomic particles in that they differ from one another physically because of the way mass-energy is distributed within the atom. All atoms are put together in similar ways. Mass in all atoms is distributed among three elementary particles called the pro ...
Hard metal
... Entropy and enthalpy changes provide driving force for the macrocyclic effect but the balance between the two is complex. Metal-ligand bonding is optimized when the size of the macrocyclic cavity and metal ion radius is closely matched. This promotes a favorable negative DH for complexation Fo ...
... Entropy and enthalpy changes provide driving force for the macrocyclic effect but the balance between the two is complex. Metal-ligand bonding is optimized when the size of the macrocyclic cavity and metal ion radius is closely matched. This promotes a favorable negative DH for complexation Fo ...
Welcome to CHMC31 course, a course that brings to you... world of transition elements. Below you will find a more... Intermediate Inorganic Chemistry (CHMC31Y3)
... and bonding. Useful to learn a lot about the elements and their reactivity). Miessler, G.L., and D.A. Tarr. Inorganic Chemistry. 3rd ed. Upper Saddle River: Pearson Prentice Hall, 2004. (A good text for our topics 2, 3 (UV-Vis), 6, and 8; it is on course reserves in UTSC library) Huheey, J.E., E.A. ...
... and bonding. Useful to learn a lot about the elements and their reactivity). Miessler, G.L., and D.A. Tarr. Inorganic Chemistry. 3rd ed. Upper Saddle River: Pearson Prentice Hall, 2004. (A good text for our topics 2, 3 (UV-Vis), 6, and 8; it is on course reserves in UTSC library) Huheey, J.E., E.A. ...
Intermediate Inorganic Chemistry (CHMC39Y)
... and bonding. Useful to learn a lot about the elements and their reactivity). Miessler, G.L., and D.A. Tarr. Inorganic Chemistry. 3rd ed. Upper Saddle River: Pearson Prentice Hall, 2004. (A good text for our topics 2, 3 (UV-Vis), 6, and 8; it is on course reserves in UTSC library) Huheey, J.E., E.A. ...
... and bonding. Useful to learn a lot about the elements and their reactivity). Miessler, G.L., and D.A. Tarr. Inorganic Chemistry. 3rd ed. Upper Saddle River: Pearson Prentice Hall, 2004. (A good text for our topics 2, 3 (UV-Vis), 6, and 8; it is on course reserves in UTSC library) Huheey, J.E., E.A. ...
- Department of Chemistry, York University
... a. Ions as Measures of Electron Density Ions are susceptible to spectroscopic detection, but free electrons are not. - When approximate electro-neutrality prevails, the determination of molecular ion abundance can provide a partial picture of the free-electron abundance. - Electron density is though ...
... a. Ions as Measures of Electron Density Ions are susceptible to spectroscopic detection, but free electrons are not. - When approximate electro-neutrality prevails, the determination of molecular ion abundance can provide a partial picture of the free-electron abundance. - Electron density is though ...
CHAPTER 1 INTRODUCTION 1.1 Research Background
... These carbene were synthesized by Schrock in 1974 for the first time through a reaction between neopentyl lithium and dichlorotris (neopentyl) tantalium (V). These carbenes are usually formed with high oxidation state with middle and early transition metals such as Ti(IV) and Ta(V) with π-donor liga ...
... These carbene were synthesized by Schrock in 1974 for the first time through a reaction between neopentyl lithium and dichlorotris (neopentyl) tantalium (V). These carbenes are usually formed with high oxidation state with middle and early transition metals such as Ti(IV) and Ta(V) with π-donor liga ...
Document
... subscripts by 2 or 3, we would have obtained the formulas C3H6O3 and C2H4O2, respectively. Although the ratio of carbon to hydrogen to oxygen atoms in each of these formulas is correct (1:2:1), neither is the simplest formula because the subscripts are not in the smallest possible whole-number ratio ...
... subscripts by 2 or 3, we would have obtained the formulas C3H6O3 and C2H4O2, respectively. Although the ratio of carbon to hydrogen to oxygen atoms in each of these formulas is correct (1:2:1), neither is the simplest formula because the subscripts are not in the smallest possible whole-number ratio ...
Mass Spectrometry and Organic
... Analyzing Ion Clusters: a way to rule candidate structures in or out Mass spectrometry “sees” all the isotopomers as distinct ions An ion with all 12C is one mass unit different from an ion with one 13C and the rest 12C Since the isotope distribution in nature is known* for all the elements (13C is ...
... Analyzing Ion Clusters: a way to rule candidate structures in or out Mass spectrometry “sees” all the isotopomers as distinct ions An ion with all 12C is one mass unit different from an ion with one 13C and the rest 12C Since the isotope distribution in nature is known* for all the elements (13C is ...
Infrared, Raman, and Luminescence Spectra of Phenyl Isocyanide
... tors for the coulombic interactions between electrons (Vee ), nuclei,
and finally electrons and nuclei
... tors for the coulombic interactions between electrons (Vee ), nuclei
Coordination Compounds (NCERT)
... The degenerate d-orbitals (in a spherical field environment) split into two levels i.e., eg and t2g in the presence of ligands. The splitting of the degenerate levels due to the presence of ligands is called the crystal-field splitting while the energy difference between the two levels (eg and t2g) ...
... The degenerate d-orbitals (in a spherical field environment) split into two levels i.e., eg and t2g in the presence of ligands. The splitting of the degenerate levels due to the presence of ligands is called the crystal-field splitting while the energy difference between the two levels (eg and t2g) ...
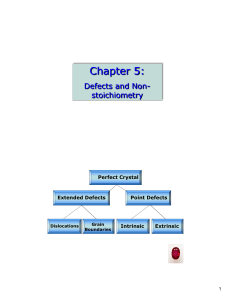
Chapter 5:
... is 6.12×10-27, whereas at 1000 K this increases to 1.37×10-8 At room temperature there are very few Schottky defects, even at 1000K there are only about 1 or 2 defects per hundred million sites. Depending on the value of DH, a Schottky or Frenkel defect may be present. The lower DH dominates, but in ...
... is 6.12×10-27, whereas at 1000 K this increases to 1.37×10-8 At room temperature there are very few Schottky defects, even at 1000K there are only about 1 or 2 defects per hundred million sites. Depending on the value of DH, a Schottky or Frenkel defect may be present. The lower DH dominates, but in ...
Transition metals and coordination chemistry
... Coordination Chemistry The Nobel Prize in Chemistry 1913 Alfred Werner (University of Zurich, Switzerland) "in recognition of his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemist ...
... Coordination Chemistry The Nobel Prize in Chemistry 1913 Alfred Werner (University of Zurich, Switzerland) "in recognition of his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemist ...
Chapter 23
... align opposite each other, but the spins are not equal, so there is a net magnetic field. • This can occur because magnetic centers have different numbers of unpaired electrons; more sites align in one direction than the other; both of these conditions apply. • Examples are NiMnO3, Y3Fe5O12, a ...
... align opposite each other, but the spins are not equal, so there is a net magnetic field. • This can occur because magnetic centers have different numbers of unpaired electrons; more sites align in one direction than the other; both of these conditions apply. • Examples are NiMnO3, Y3Fe5O12, a ...
No Slide Title
... Electron Paramagnetic Resonance (EPR) is an important tool in experimental studies of systems containing unpaired electrons[1]. The traditional application areas for EPR include studies of transition metal complexes, stable organic radicals, transient reaction intermediates, as well as solid state a ...
... Electron Paramagnetic Resonance (EPR) is an important tool in experimental studies of systems containing unpaired electrons[1]. The traditional application areas for EPR include studies of transition metal complexes, stable organic radicals, transient reaction intermediates, as well as solid state a ...
Heterobimetallic chemistry: Heterobimetallic complexes derived
... values for monomer formulation, respectively. On the other hand, for the remaining complexes, the experimental values were found to be close to the theoretical values calculated for dimeric formulation. However, the experimental values are much higher than the values calculated either for the monome ...
... values for monomer formulation, respectively. On the other hand, for the remaining complexes, the experimental values were found to be close to the theoretical values calculated for dimeric formulation. However, the experimental values are much higher than the values calculated either for the monome ...
Theory of resonance Raman scattering and fluorescence from
... 30-33 and references given therein. More recently, the timedependent reformulation of the KHD formula 34 has been extended to include vibronic coupling in the excited states. 35,36 In these works only the pure (coherent) Raman emission has been considered, which arises from SE emission processes whi ...
... 30-33 and references given therein. More recently, the timedependent reformulation of the KHD formula 34 has been extended to include vibronic coupling in the excited states. 35,36 In these works only the pure (coherent) Raman emission has been considered, which arises from SE emission processes whi ...
Excited States of Pt(PF3)4 and Their Role in Focused Electron Beam
... the calculations of response propertiesan essential point when addressing positions of high-lying states.23 This level of theory failed in describing the situation far from the minimum, i.e., for calculating potential energy curves for dissociation of a ligand. First, LDA is known to show overbindi ...
... the calculations of response propertiesan essential point when addressing positions of high-lying states.23 This level of theory failed in describing the situation far from the minimum, i.e., for calculating potential energy curves for dissociation of a ligand. First, LDA is known to show overbindi ...
Nitric oxide NO +2 Neutral Dinitrogen trioxide N2O3 +3 Acidic
... 2 MARK QUESTION (SHORT ANSWER TYPE QUESTION) 1. Why is HF acid stored in wax coated glass bottles? This is because HF does not attack wax but reacts with glass.It dissolves SiO2 present in glass forming hydrofluorosilicic acid. SiO2 +6HFÆH2SiF6+2H2O 2. What is laughing gas? Why is it so called? ...
... 2 MARK QUESTION (SHORT ANSWER TYPE QUESTION) 1. Why is HF acid stored in wax coated glass bottles? This is because HF does not attack wax but reacts with glass.It dissolves SiO2 present in glass forming hydrofluorosilicic acid. SiO2 +6HFÆH2SiF6+2H2O 2. What is laughing gas? Why is it so called? ...
Tuning PCP-Ir Complexes: The impact of an N
... results in the stabilization of ylidic canonical forms II and III (Scheme 1). As a consequence of this, the terminal carbon atom is strongly nucleophilic, which results in an end-on coordination mode when acting as ligand for transition metals.[4a-b,5] ...
... results in the stabilization of ylidic canonical forms II and III (Scheme 1). As a consequence of this, the terminal carbon atom is strongly nucleophilic, which results in an end-on coordination mode when acting as ligand for transition metals.[4a-b,5] ...
Crystal-Field Theory
... • Weak field ligands tend to favor adding electrons to the higher energy orbitals (high spin complexes) because < pairing energy. • Strong field ligands tend to favor adding electrons to lower energy orbitals (low spin complexes) because > pairing energy. ...
... • Weak field ligands tend to favor adding electrons to the higher energy orbitals (high spin complexes) because < pairing energy. • Strong field ligands tend to favor adding electrons to lower energy orbitals (low spin complexes) because > pairing energy. ...
Chemistry 324 Review Problems 5
... 2. Suggest a reason why the Cr3+ ion in Q1 shows such remarkable differences in the two complexes given. Normally, first row, octahedral transition metal ions undergo dissociative exchange (Id mechanism) but this is strongly disfavoured electronically for d3 ions due to the large loss in CFSE. This ...
... 2. Suggest a reason why the Cr3+ ion in Q1 shows such remarkable differences in the two complexes given. Normally, first row, octahedral transition metal ions undergo dissociative exchange (Id mechanism) but this is strongly disfavoured electronically for d3 ions due to the large loss in CFSE. This ...
No Slide Title
... If the bonds form from overlap of 3 2p orbitals on nitrogen with the 1s orbital on each hydrogen atom, what would the molecular geometry of NH3 be? If use the 3 2p orbitals predict 90o Actual H-N-H bond angle is 107.3o ...
... If the bonds form from overlap of 3 2p orbitals on nitrogen with the 1s orbital on each hydrogen atom, what would the molecular geometry of NH3 be? If use the 3 2p orbitals predict 90o Actual H-N-H bond angle is 107.3o ...
Jahn–Teller effect
-3D-balls.png?width=300)
The Jahn–Teller effect, sometimes also known as Jahn–Teller distortion, describes the geometrical distortion of molecules and ions that is associated with certain electron configurations. This electronic effect is named after Hermann Arthur Jahn and Edward Teller, who proved, using group theory, that orbital nonlinear spatially degenerate molecules cannot be stable. The Jahn–Teller theorem essentially states that any nonlinear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. For a description of another type of geometrical distortion that occurs in crystals with substitutional impurities see article off-center ions.