Syntheses and characterization of [4
... This tilt causes the orbitals of the Cp ligands to interact differently with the d-orbitals of the central metal than those in parallel metallocenes. Bent metallocenes can be prepared by bonding groups to the central metal in between the two Cp ligands or by connecting the two Cp rings with a short ...
... This tilt causes the orbitals of the Cp ligands to interact differently with the d-orbitals of the central metal than those in parallel metallocenes. Bent metallocenes can be prepared by bonding groups to the central metal in between the two Cp ligands or by connecting the two Cp rings with a short ...
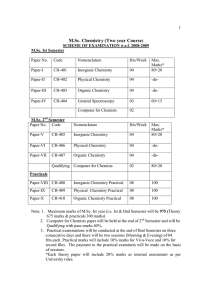
M.Sc. Chemistry (Two year Course)
... Schrodinger wave equation for a particle in a three dimensional box and the concept of degeneracy of energy levels. Schrodinger wave equation for linear harmonic oscillator, solution by polynomial method, zero point energy and its consequence. Schrodinger wave equation for three dimensional Rigid ro ...
... Schrodinger wave equation for a particle in a three dimensional box and the concept of degeneracy of energy levels. Schrodinger wave equation for linear harmonic oscillator, solution by polynomial method, zero point energy and its consequence. Schrodinger wave equation for three dimensional Rigid ro ...
227
... orientation at the surface, and the electric field polarization at the metal surface. These constrains will be later introduced as surface selection rules or propensity rules. On the other hand, the most challenging case arises when the molecule is chemically adsorbed onto the rough surface. A surfa ...
... orientation at the surface, and the electric field polarization at the metal surface. These constrains will be later introduced as surface selection rules or propensity rules. On the other hand, the most challenging case arises when the molecule is chemically adsorbed onto the rough surface. A surfa ...
From metal complexes to fullerene arrays: exploring the exciting
... is relatively simple. A certain number of molecular subunits are assembled in a suitable spatial arrangement. Light excitation of a specific molecular fragment may modify electronic interactions among the components and give rise to phenomena such as energy or electron transfer. In these terms, indee ...
... is relatively simple. A certain number of molecular subunits are assembled in a suitable spatial arrangement. Light excitation of a specific molecular fragment may modify electronic interactions among the components and give rise to phenomena such as energy or electron transfer. In these terms, indee ...
CARBON-HYDROGEN-TRANSITION METAL BONDS
... receive the two electrons of the C-H bond. It is to be presumed that this orbital will be essentially of d-character for transition metal compounds. The orbital should be a very good acceptor and the energy and disposition should approach that of the C-H bonding electrons as far as possible. If ther ...
... receive the two electrons of the C-H bond. It is to be presumed that this orbital will be essentially of d-character for transition metal compounds. The orbital should be a very good acceptor and the energy and disposition should approach that of the C-H bonding electrons as far as possible. If ther ...
5. J. Am. Chem. Soc., 2009, 131, 16544
... (1:1:1:1) in CD3CN/CDCl3 (3:1) and were sonicated at 50 °C for 2 h. The resultant yellow solution was characterized by 1H NMR and electrospray ionization mass spectroscopy. Three different ligands in the presence of a metal ion may form six different complexes (Scheme 2). However, the number is redu ...
... (1:1:1:1) in CD3CN/CDCl3 (3:1) and were sonicated at 50 °C for 2 h. The resultant yellow solution was characterized by 1H NMR and electrospray ionization mass spectroscopy. Three different ligands in the presence of a metal ion may form six different complexes (Scheme 2). However, the number is redu ...
Syntheses, spectral characterization, thermal properties and DNA
... ligands L1/L2 or L3 to give the corresponding metal complexes. The elemental analysis data for ligands and complexes is given in Table.1. All the compounds show the analytical results close to the theoretical values indicating the presence of two types of ligands. 3.2. IR Spectra The IR spectral dat ...
... ligands L1/L2 or L3 to give the corresponding metal complexes. The elemental analysis data for ligands and complexes is given in Table.1. All the compounds show the analytical results close to the theoretical values indicating the presence of two types of ligands. 3.2. IR Spectra The IR spectral dat ...
The unique electronic structure of Ca10 (Pt4As8)(Fe2
... plots in Fig. 3(b), particularly along the cuts #1-#4 and #11#16. The dispersions of these electron-like bands are identical as expected. Based on these observations, the ν Fermi pocket should be attributed to the states in the Pt4 As8 layer. This finding qualitatively agrees with a recent first pri ...
... plots in Fig. 3(b), particularly along the cuts #1-#4 and #11#16. The dispersions of these electron-like bands are identical as expected. Based on these observations, the ν Fermi pocket should be attributed to the states in the Pt4 As8 layer. This finding qualitatively agrees with a recent first pri ...
Hydrides: Solid State Transition Metal Complexes
... and heavy alkaline earth metals are often sensitive to air and moisture. A decisive parameter for hydride formation is pressure. The synthesis of hydrides based on light transition elements usually requires higher hydrogen pressures than those based on heavier congeners. Examples are the palladium c ...
... and heavy alkaline earth metals are often sensitive to air and moisture. A decisive parameter for hydride formation is pressure. The synthesis of hydrides based on light transition elements usually requires higher hydrogen pressures than those based on heavier congeners. Examples are the palladium c ...
Photochemistry of tetrasulfido complexes of molybdenum (VI
... ligands to the metal.' However, a t these high formal oxidation states of the metal, the bonding in these complexes must have large covalent contributions. I t follows that some of the MO's are strongly delocalized between metal and ligands. L M C T transitions may then not be associated with the tr ...
... ligands to the metal.' However, a t these high formal oxidation states of the metal, the bonding in these complexes must have large covalent contributions. I t follows that some of the MO's are strongly delocalized between metal and ligands. L M C T transitions may then not be associated with the tr ...
Design of molecular structure and synthetic approaches
... single source precursors (SSP) – the species permitting to specifically facilitate the formation of the desired material in such aspects as chemical and phase composition, morphology or domain structure via preservation of the chemical composition or structure features of the building blocks from pr ...
... single source precursors (SSP) – the species permitting to specifically facilitate the formation of the desired material in such aspects as chemical and phase composition, morphology or domain structure via preservation of the chemical composition or structure features of the building blocks from pr ...
Absence of photoemission from the Fermi level in potassium
... order to clarify their electronic ground state. Apart from crystal structure issues there is also structural changes of the molecular backbone itself upon charge addition. This is a natural consequence of the filling of orbitals with anti-bonding character and the accompanied relaxation of the molec ...
... order to clarify their electronic ground state. Apart from crystal structure issues there is also structural changes of the molecular backbone itself upon charge addition. This is a natural consequence of the filling of orbitals with anti-bonding character and the accompanied relaxation of the molec ...
Oxidative Addition - vrg
... present. Unlike most of the other substrate molecules that break a single bond and form two separate anionic ligands upon the oxidative addition, these ligands have double or triple bonds and only one of the π-bonds is broken leaving the σ-bond intact. The ligand does pick up two electrons from the ...
... present. Unlike most of the other substrate molecules that break a single bond and form two separate anionic ligands upon the oxidative addition, these ligands have double or triple bonds and only one of the π-bonds is broken leaving the σ-bond intact. The ligand does pick up two electrons from the ...
Binuclear Metal Carbonyl DAB Complexes. III. Activation of a C=N
... lone pairs on nitrogen. Only one example has been reported where the nelectrons were involved in the metal-to-ligand bonding [7], while for some reactions of coordinated DAB ligands R-coordinated intermediates were assumed [8]. In this preliminary paper on ruthenium carbonyl DAB complexes, the earli ...
... lone pairs on nitrogen. Only one example has been reported where the nelectrons were involved in the metal-to-ligand bonding [7], while for some reactions of coordinated DAB ligands R-coordinated intermediates were assumed [8]. In this preliminary paper on ruthenium carbonyl DAB complexes, the earli ...
Questa è la versione dell`autore dell`opera: [Chemical Reviews
... Resonance (EPR) techniques have turned to be most powerful to achieve this goal, providing of course that the adsorbate-surface system be paramagnetic. The present review intends to describe and classify the variety of inorganic radicals which are formed at oxide surfaces, emphasizing the importanc ...
... Resonance (EPR) techniques have turned to be most powerful to achieve this goal, providing of course that the adsorbate-surface system be paramagnetic. The present review intends to describe and classify the variety of inorganic radicals which are formed at oxide surfaces, emphasizing the importanc ...
Enabling enhanced emission and low-threshold lasing
... (6–9), as well as more fundamental studies of cavity quantum electrodynamics (10–12) and various types of enhanced spectroscopy (13) and sensing (14). In all, it is frequently sought to alter (15–18) and often to enhance this interaction by allowing it to occur in a typically nanostructured cavity, ...
... (6–9), as well as more fundamental studies of cavity quantum electrodynamics (10–12) and various types of enhanced spectroscopy (13) and sensing (14). In all, it is frequently sought to alter (15–18) and often to enhance this interaction by allowing it to occur in a typically nanostructured cavity, ...
Visible-Light Excitation of Infrared Lanthanide
... Attempts to enforce a time delay (10 µs) in the luminescence measurements resulted in the complete disappearance of both bands. Attempts to measure the lifetime of the emission band at 723 nm failed due to weak intensity and an apparent poly-exponential decay profile; however, the long timescale of ...
... Attempts to enforce a time delay (10 µs) in the luminescence measurements resulted in the complete disappearance of both bands. Attempts to measure the lifetime of the emission band at 723 nm failed due to weak intensity and an apparent poly-exponential decay profile; however, the long timescale of ...
Ceria co-doping: Synergistic or average effect?
... approach allows us to study systems with realistic doped/co-doped defect concentrations within the temperatures of interest for SOFC applications (600-1000 C◦ ), and to accumulate sufficiently long trajectories to calculate the conductivity. This is in contrast to most of the previous computational ...
... approach allows us to study systems with realistic doped/co-doped defect concentrations within the temperatures of interest for SOFC applications (600-1000 C◦ ), and to accumulate sufficiently long trajectories to calculate the conductivity. This is in contrast to most of the previous computational ...
CfE Advanced Higher Chemistry
... Electromagnetic radiation is a form of energy. Light, x-rays, radio signals and microwaves are all forms of electromagnetic radiation. Visible light is only a small part of the range of the electromagnetic spectrum. Figure 1.1: The electromagnetic spectrum ...
... Electromagnetic radiation is a form of energy. Light, x-rays, radio signals and microwaves are all forms of electromagnetic radiation. Visible light is only a small part of the range of the electromagnetic spectrum. Figure 1.1: The electromagnetic spectrum ...
covalent - Typepad
... 1. In the compound sodium fluoride, NaF, the sodium atom loses one electron and the fluorine atom gains one electron to form ions that are isoelectric with a. helium. c. neon. b. oxygen. d. calcium. 2. Atoms of copper and iron a. generally form stable bonds with transition elements. b. have stable e ...
... 1. In the compound sodium fluoride, NaF, the sodium atom loses one electron and the fluorine atom gains one electron to form ions that are isoelectric with a. helium. c. neon. b. oxygen. d. calcium. 2. Atoms of copper and iron a. generally form stable bonds with transition elements. b. have stable e ...
Downloadable Full Text - DSpace@MIT
... cyclization transition state (Path B, Scheme 1). For the reaction with acetylene, coordination of BEt3 to the aldehyde oxygen destabilizes the reactant π complex 7 and the oxidative cyclization transition state TS1-B by 14.2 and 2.4 kcal/mol, respectively, in terms of free energies (Figure 2a). Howe ...
... cyclization transition state (Path B, Scheme 1). For the reaction with acetylene, coordination of BEt3 to the aldehyde oxygen destabilizes the reactant π complex 7 and the oxidative cyclization transition state TS1-B by 14.2 and 2.4 kcal/mol, respectively, in terms of free energies (Figure 2a). Howe ...
Exam 3 Review Problems
... 56. All of the following statements concerning crystal field theory are true EXCEPT a. the crystal field splitting is larger in low‐spin complexes than high‐spin complexes. b. low‐spin complexes contain the maximum number of unpaired electrons. c. in an isolated atom or ion, the five d orbitals ...
... 56. All of the following statements concerning crystal field theory are true EXCEPT a. the crystal field splitting is larger in low‐spin complexes than high‐spin complexes. b. low‐spin complexes contain the maximum number of unpaired electrons. c. in an isolated atom or ion, the five d orbitals ...
Molecular Quantum Mechanics
... examples to provide the reader with further opportunities to see formulae in action. We have added new problems for each chapter. We have expanded the discussion on numerous occasions within the body of the text to provide further clarification for or insight into mathematical results. We have set a ...
... examples to provide the reader with further opportunities to see formulae in action. We have added new problems for each chapter. We have expanded the discussion on numerous occasions within the body of the text to provide further clarification for or insight into mathematical results. We have set a ...
Transition Metals and their reactions
... • Most transition metal colours are due to d-d electron transitions. The energy gap between the split d-orbitals corresponds to visible light (E=hf). • Some of the really strong colours are due to charge transfer (such as in MnO4-). ...
... • Most transition metal colours are due to d-d electron transitions. The energy gap between the split d-orbitals corresponds to visible light (E=hf). • Some of the really strong colours are due to charge transfer (such as in MnO4-). ...
Naming complexes and isomerisation pdf
... Coordination number four The most common coordination geometries are either tetrahedral or square-planar. Examples of tetrahedral complexes are Li(H2O)4+, BeF42-, CoBr42- and Ni(CO)4. Tetrahedral geometry is preferred for valence electron configurations d0 and d10. Square planar is preferred for d8 ...
... Coordination number four The most common coordination geometries are either tetrahedral or square-planar. Examples of tetrahedral complexes are Li(H2O)4+, BeF42-, CoBr42- and Ni(CO)4. Tetrahedral geometry is preferred for valence electron configurations d0 and d10. Square planar is preferred for d8 ...
Jahn–Teller effect
-3D-balls.png?width=300)
The Jahn–Teller effect, sometimes also known as Jahn–Teller distortion, describes the geometrical distortion of molecules and ions that is associated with certain electron configurations. This electronic effect is named after Hermann Arthur Jahn and Edward Teller, who proved, using group theory, that orbital nonlinear spatially degenerate molecules cannot be stable. The Jahn–Teller theorem essentially states that any nonlinear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. For a description of another type of geometrical distortion that occurs in crystals with substitutional impurities see article off-center ions.