Pierre Thuéry
... metallacycle with the metal atoms separated by 9.4967(5) Å (Fig. 2). Coordination of the third arm of HNTA results in the formation of nearly equilateral uranium triangles, the third side, parallel to the b axis, being 9.9608(7) Å in length. These adjacent triangles form a ribbon or ladder-like asse ...
... metallacycle with the metal atoms separated by 9.4967(5) Å (Fig. 2). Coordination of the third arm of HNTA results in the formation of nearly equilateral uranium triangles, the third side, parallel to the b axis, being 9.9608(7) Å in length. These adjacent triangles form a ribbon or ladder-like asse ...
C - mvhs-fuhsd.org
... D. Atoms have a positively charged nucleus surrounded by an electron cloud. E. No two electrons in one atom can have the same four quantum numbers. 65. The following properties are observed for an unknown element, Z: at room temperature, it is gray, lustrous solid. The compound ZCl2 dissolves in wat ...
... D. Atoms have a positively charged nucleus surrounded by an electron cloud. E. No two electrons in one atom can have the same four quantum numbers. 65. The following properties are observed for an unknown element, Z: at room temperature, it is gray, lustrous solid. The compound ZCl2 dissolves in wat ...
I. scientific notation. – a shorthand that scientists use when dealing
... Chapter 2 Stoichiometry–describes the quantitative relationships among elements 1. in compounds (composition stoichiometry) 2. in chemical changes (reaction stoichiometry) Dalton’s Atomic Theory – summarized experimental observations and interpretations in the nature of atoms: 1. an element is compo ...
... Chapter 2 Stoichiometry–describes the quantitative relationships among elements 1. in compounds (composition stoichiometry) 2. in chemical changes (reaction stoichiometry) Dalton’s Atomic Theory – summarized experimental observations and interpretations in the nature of atoms: 1. an element is compo ...
10/18/11 - Note: Once it is downloaded, click SET
... valence electrons- electrons found in the outermost shell (energy level) of an atom - also determines the atom’s properties - outer shell electrons participate in chemicals reactions There are 7 shells (energy levels) ...
... valence electrons- electrons found in the outermost shell (energy level) of an atom - also determines the atom’s properties - outer shell electrons participate in chemicals reactions There are 7 shells (energy levels) ...
Chapter 08
... Shared electron pairs are shown either as dashes or as pairs of dots. Lone pairs are shown as pairs of dots on atoms. H with 2 e− ...
... Shared electron pairs are shown either as dashes or as pairs of dots. Lone pairs are shown as pairs of dots on atoms. H with 2 e− ...
Chapter 9: Molecular Geometry and Hybridization of Atomic Orbitals
... Now, there are two orbitals in the Be-atom available for bonding. One Cl-atom would share the 2s orbital, and the other Cl-atom would share the 2p orbital. This will result in two non-equivalent Be-Cl bonds. However, experiments suggest that the two Be-Cl bonds are equivalent in every respect. ...
... Now, there are two orbitals in the Be-atom available for bonding. One Cl-atom would share the 2s orbital, and the other Cl-atom would share the 2p orbital. This will result in two non-equivalent Be-Cl bonds. However, experiments suggest that the two Be-Cl bonds are equivalent in every respect. ...
High Energy Phenomena in Supergiant X-ray Binaries - HAL-Insu
... General characteristics SFXTs constitute a new class of ∼ 12 sources identified among the recently discovered IGRs. They are HMXBs hosting NS orbiting around sgOB companion stars, exhibiting peculiar characteristics compared to “classical” HMXBs: rapid outbursts lasting only for hours, faint quiesce ...
... General characteristics SFXTs constitute a new class of ∼ 12 sources identified among the recently discovered IGRs. They are HMXBs hosting NS orbiting around sgOB companion stars, exhibiting peculiar characteristics compared to “classical” HMXBs: rapid outbursts lasting only for hours, faint quiesce ...
Experiment 4 - Macalester College
... the mouse so that everyone gains a solid proficiency with MacSpartan Pro. In addition, it is important that all students learn how to use the Molecular Visions® model kit. All information documented in your laboratory notebook should be turned in as your report for this experiment. Make sure that ea ...
... the mouse so that everyone gains a solid proficiency with MacSpartan Pro. In addition, it is important that all students learn how to use the Molecular Visions® model kit. All information documented in your laboratory notebook should be turned in as your report for this experiment. Make sure that ea ...
Subject Area Assessment Guides
... ionic bond. Chemical bonds between atoms can be almost entirely covalent, almost entirely ionic, or in between these two extremes. The triple bond in nitrogen molecules (N2) is nearly 100 percent covalent. A salt such as sodium chloride (NaCl) has bonds that are nearly completely ionic. However, the ...
... ionic bond. Chemical bonds between atoms can be almost entirely covalent, almost entirely ionic, or in between these two extremes. The triple bond in nitrogen molecules (N2) is nearly 100 percent covalent. A salt such as sodium chloride (NaCl) has bonds that are nearly completely ionic. However, the ...
Figure 1. Map showing the region of the Fourth Quadrant selected
... gravity is able to overwhelm magnetic support. Turbulence is not considered in this picture since it is considered unimportant on the scale of quasi-static cores. However the formation of isolated, low-mass stars, for which this theory appears to work, is a special case, as most stars exist in binar ...
... gravity is able to overwhelm magnetic support. Turbulence is not considered in this picture since it is considered unimportant on the scale of quasi-static cores. However the formation of isolated, low-mass stars, for which this theory appears to work, is a special case, as most stars exist in binar ...
as a PDF
... seemed ridiculous. The laborious discovery and separation of the elements by the classical techniques of fractional crystallization and precipitation naturally led to the view that the lanthanides were all very much alike. But by 1933, Klemm had exposed inadequacies in this similarity paradigm when ...
... seemed ridiculous. The laborious discovery and separation of the elements by the classical techniques of fractional crystallization and precipitation naturally led to the view that the lanthanides were all very much alike. But by 1933, Klemm had exposed inadequacies in this similarity paradigm when ...
CHEMISTRY 103 – Practice Problems #3 Chapters 8 – 10 http
... c. 3 d. 4 e. 5 27. There are two possible structures, one polar and one nonpolar, for each chemical given. Draw the two possible electron domain geometry structures, one polar and one nonpolar, for each chemical (i.e., two electron domain geometry structures for “a” and two for “b”). a. PCl2F3 b. Xe ...
... c. 3 d. 4 e. 5 27. There are two possible structures, one polar and one nonpolar, for each chemical given. Draw the two possible electron domain geometry structures, one polar and one nonpolar, for each chemical (i.e., two electron domain geometry structures for “a” and two for “b”). a. PCl2F3 b. Xe ...
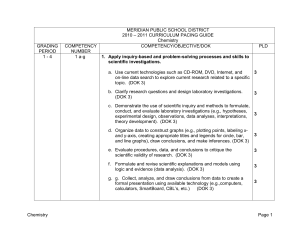
MERIDIAN PUBLIC SCHOOL DISTRICT
... beta, gamma) and the properties of the emissions (e.g., composition, mass, charge, penetrating power) The concept of half-life for a radioactive isotope (e.g., carbon-14 dating) based on the principle that the decay of any individual atom is a random process Page 4 ...
... beta, gamma) and the properties of the emissions (e.g., composition, mass, charge, penetrating power) The concept of half-life for a radioactive isotope (e.g., carbon-14 dating) based on the principle that the decay of any individual atom is a random process Page 4 ...
Synthesis and crystal structure of
... solvents were dried over liquid sodium/potassium alloy) resulted in an almost immediate decomposition to give black indium metal. The 13C NMR spectrum of 1 shows six signals, thus indicating free rotation and equilibration of the benzyl groups in solution, as previously observed for the thallium ana ...
... solvents were dried over liquid sodium/potassium alloy) resulted in an almost immediate decomposition to give black indium metal. The 13C NMR spectrum of 1 shows six signals, thus indicating free rotation and equilibration of the benzyl groups in solution, as previously observed for the thallium ana ...
down
... bond order, bond energy, bond length, and vibrational force constant for series of H2 → Ne2 Bond energy : peak at N2 and smaller peak at H2 Force constant : similar than bond energy trend(but it is more complicated trend for lighter molecules) Bond length : increased as bond energy and force c ...
... bond order, bond energy, bond length, and vibrational force constant for series of H2 → Ne2 Bond energy : peak at N2 and smaller peak at H2 Force constant : similar than bond energy trend(but it is more complicated trend for lighter molecules) Bond length : increased as bond energy and force c ...
PHYS 1400 Sample Exam Questions: Properties of Matter (Atoms) 1
... B) have an extremely small mass, compared to protons or neutrons. C) orbit randomly, and can be anywhere at any distance from the nucleus. D) are the “glue” that keeps the neutrons orbiting the nucleus. 24. Beryllium has an atomic number of 4. This means A) there are 4 particles in the nucleus: 2 pr ...
... B) have an extremely small mass, compared to protons or neutrons. C) orbit randomly, and can be anywhere at any distance from the nucleus. D) are the “glue” that keeps the neutrons orbiting the nucleus. 24. Beryllium has an atomic number of 4. This means A) there are 4 particles in the nucleus: 2 pr ...
Document
... INTRODUCTION: The overview of the “Why, Where, and What” of bonding It is important that atoms bond. Why? Because they need to bond in order to make _____________, _______________, and other more complex forms of matter. For example, if atoms didn’t bond, you would be quite thirsty all the time! Yes ...
... INTRODUCTION: The overview of the “Why, Where, and What” of bonding It is important that atoms bond. Why? Because they need to bond in order to make _____________, _______________, and other more complex forms of matter. For example, if atoms didn’t bond, you would be quite thirsty all the time! Yes ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.