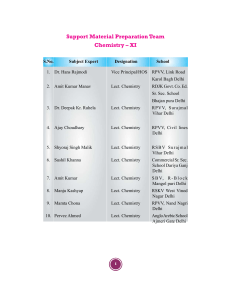
Sructural and chemisorption properties of metallic surfaces and metallic overlayers
... and W(IlO)) have been studied using various surface analytical techniques. The results for the Pd/Al system show that the Pd film grows on Al(IlO) in a layer-bylayer mode. The binding energies of the Pd 4d electrons for a very thin Pd film are very similar to th at for an atomic-like state. The film ...
... and W(IlO)) have been studied using various surface analytical techniques. The results for the Pd/Al system show that the Pd film grows on Al(IlO) in a layer-bylayer mode. The binding energies of the Pd 4d electrons for a very thin Pd film are very similar to th at for an atomic-like state. The film ...
Fe(III) (Oxy)hydroxide Compositional Series
... Cornell.36 A 1 M solution of NaOH (Sigma-Aldrich) was added at a rate of 2 mL/min to a 0.2 M solution of Fe(NO3) (Sigma-Aldrich) with constant stirring until the pH reached 7.5. The solution was repeatedly washed with deionized water and centrifuged to remove the remaining electrolytes, followed by ...
... Cornell.36 A 1 M solution of NaOH (Sigma-Aldrich) was added at a rate of 2 mL/min to a 0.2 M solution of Fe(NO3) (Sigma-Aldrich) with constant stirring until the pH reached 7.5. The solution was repeatedly washed with deionized water and centrifuged to remove the remaining electrolytes, followed by ...
Version 1.6 - Clark Science Center
... of the materials. Color is of interest to us because it teaches us what the structures are, and how easily electrons are moved around. The latter leads directly to asking questions about reactivity: Which molecules will react with each other, what energy changes take place during that reaction, and ...
... of the materials. Color is of interest to us because it teaches us what the structures are, and how easily electrons are moved around. The latter leads directly to asking questions about reactivity: Which molecules will react with each other, what energy changes take place during that reaction, and ...
as a PDF
... between the radiolytic reduction process of both types of ions and the electron-transfer reaction from the less noble metal atoms to the other metal ions.8-10 Then, this preferential reduction of the more noble metal first results in a segregation between the metals and eventually in a core-shell st ...
... between the radiolytic reduction process of both types of ions and the electron-transfer reaction from the less noble metal atoms to the other metal ions.8-10 Then, this preferential reduction of the more noble metal first results in a segregation between the metals and eventually in a core-shell st ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
Sample pages 1 PDF
... can be inferred from the observations. Although their interpretation is not unique and unambiguous, the plausible range of values is between ∼3 and a few hundred gravitational radii. There is no commonly accepted mechanism of truncation of the disk and formation of the corona, with a number of plaus ...
... can be inferred from the observations. Although their interpretation is not unique and unambiguous, the plausible range of values is between ∼3 and a few hundred gravitational radii. There is no commonly accepted mechanism of truncation of the disk and formation of the corona, with a number of plaus ...
- StarBooks
... Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and other properties. Atoms of different elements are different in all respects. Atom is the smallest unit that takes part in chemical combinati ...
... Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical in shape, size, mass and other properties. Atoms of different elements are different in all respects. Atom is the smallest unit that takes part in chemical combinati ...
structure and thermodynamics of lanthanide
... sphere20. This model of solvent separated ion pairing was supported by studies of the spectra of Nd(iii) which showed• no change up to concentrations of 5M chloride ion2 1 A spectral change was observed at higher concentrations which may be related to a change in coordination number from nine to eig ...
... sphere20. This model of solvent separated ion pairing was supported by studies of the spectra of Nd(iii) which showed• no change up to concentrations of 5M chloride ion2 1 A spectral change was observed at higher concentrations which may be related to a change in coordination number from nine to eig ...
Appendices and Glossary
... carbon atoms and two moles of oxygen atoms. The atom ratio and the mole ratio of the elements are identical! The chemical formula of an ionic compound does not tell us the number of atoms in a molecule because ionic substances are not molecular. However, it still gives the mole ratio of the elements ...
... carbon atoms and two moles of oxygen atoms. The atom ratio and the mole ratio of the elements are identical! The chemical formula of an ionic compound does not tell us the number of atoms in a molecule because ionic substances are not molecular. However, it still gives the mole ratio of the elements ...
Concept Development Studies in Chemistry
... elements cannot be decomposed? What does it mean to combine elements into a compound? We want to understand more about the nature of elements and compounds so we can describe the processes by which elements combine to form compounds, by which compounds are decomposed into elements, and by which comp ...
... elements cannot be decomposed? What does it mean to combine elements into a compound? We want to understand more about the nature of elements and compounds so we can describe the processes by which elements combine to form compounds, by which compounds are decomposed into elements, and by which comp ...
4) What is the term for the procedure of collecting data and recording
... A sample of rose gold is: 12.0 grams gold, 5.0 grams silver, and 7.0 grams copper by mass. What is the percent of copper in the sample? A) 12% B) 29% C) 50% D) 58% E) 75% Stainless steel is composed of iron, manganese, chromium, and nickel. If a 2.00 g sample was analyzed and found to contain 2.75% ...
... A sample of rose gold is: 12.0 grams gold, 5.0 grams silver, and 7.0 grams copper by mass. What is the percent of copper in the sample? A) 12% B) 29% C) 50% D) 58% E) 75% Stainless steel is composed of iron, manganese, chromium, and nickel. If a 2.00 g sample was analyzed and found to contain 2.75% ...
Fatimah Final Thesis
... chalcogen–chalcogen bonds, has concentrated mostly on sulfur due to its ability to catenate and to bind to several metal centers. Similar bonding modes have been observed also for Se and Te after the mid-1970s.4,5 The properties of metal–sulfur structures have been studied extensively in chemistry. ...
... chalcogen–chalcogen bonds, has concentrated mostly on sulfur due to its ability to catenate and to bind to several metal centers. Similar bonding modes have been observed also for Se and Te after the mid-1970s.4,5 The properties of metal–sulfur structures have been studied extensively in chemistry. ...
ChemQuest 1 Information: Qualitative vs. Quantitative Critical
... (i.e. two hydrogen and one oxygen atom bonding to form a water molecule), but mixtures are formed by a physical change (i.e. stirring salt and water together. 6. How are pure substances different from mixtures? Pure substances are not mixed with anything else, but mixtures are composed of two or mor ...
... (i.e. two hydrogen and one oxygen atom bonding to form a water molecule), but mixtures are formed by a physical change (i.e. stirring salt and water together. 6. How are pure substances different from mixtures? Pure substances are not mixed with anything else, but mixtures are composed of two or mor ...
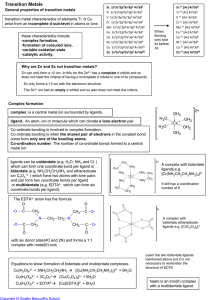
Transition Metals
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
... Zn can only form a +2 ion. In this ion the Zn2+ has a complete d orbital and so does not meet the criteria of having a incomplete d orbital in one of its compounds. Sc only forms a +3 ion with the electronic structure The Sc3+ ion had an empty d orbital and so also does not meet the criteria ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.