Section 2 Oxidation Numbers
... • In order to indicate the general distribution of electrons among the bonded atoms in a molecular compound or a polyatomic ion, _________ ________ are assigned to the atoms composing the compound or ion. • Unlike ionic charges, _______ ________ do not have an ______ ________ meaning: rather, ...
... • In order to indicate the general distribution of electrons among the bonded atoms in a molecular compound or a polyatomic ion, _________ ________ are assigned to the atoms composing the compound or ion. • Unlike ionic charges, _______ ________ do not have an ______ ________ meaning: rather, ...
On the Nature of the Change in the Wave Function in a
... 1 or hole 2 is not a common factor. It should be remembered that some causative factor is implied by the very different electron distributions in Gedankenexperiments 1 and 2. It is reasonable to conclude that knowledge by the observer regarding the particular path of the electron through the wall is ...
... 1 or hole 2 is not a common factor. It should be remembered that some causative factor is implied by the very different electron distributions in Gedankenexperiments 1 and 2. It is reasonable to conclude that knowledge by the observer regarding the particular path of the electron through the wall is ...
Balancing Reaction Equations Oxidation State Reduction
... We use the oxidation number (oxidation state) to keep track of electron shifts in chemical reactions. It is defined as “the charge which an atom appears to have when the net electric charge on a chemical species is apportioned according to certain rules”. Important because: the binding of atoms can ...
... We use the oxidation number (oxidation state) to keep track of electron shifts in chemical reactions. It is defined as “the charge which an atom appears to have when the net electric charge on a chemical species is apportioned according to certain rules”. Important because: the binding of atoms can ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
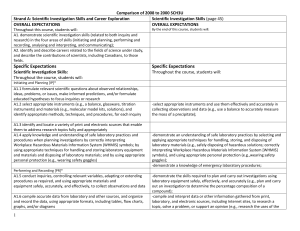
... Gather information and discuss on :a) Group 17 elements and their physical and chemical properties. b) the similarities in chemical properties of Group 17 elements c) the relationship between chemical properties of group 17 elements with their electron arrangements Carry out experiment to investigat ...
... Gather information and discuss on :a) Group 17 elements and their physical and chemical properties. b) the similarities in chemical properties of Group 17 elements c) the relationship between chemical properties of group 17 elements with their electron arrangements Carry out experiment to investigat ...
Intensities of analogous Rydberg series in CF3Cl, CF3Br and in
... atom and the sum of the intensities of all nl–n 0 l transitions that comprise all the different irreducible representations involved in the molecule. A ÔsupermultipletÕ is [28] the group of multiplets with different L-value but the same spin multiplicity that arise from a given electron configuration i ...
... atom and the sum of the intensities of all nl–n 0 l transitions that comprise all the different irreducible representations involved in the molecule. A ÔsupermultipletÕ is [28] the group of multiplets with different L-value but the same spin multiplicity that arise from a given electron configuration i ...
Chem G 9
... neutrons will have different mass numbers and are called isotopes. Students should appreciate that a natural sample of an element is likely to contain a mixture of two or more isotopes. In determining the atomic mass of the element we must take into account that it is a mixture of isotopes with diff ...
... neutrons will have different mass numbers and are called isotopes. Students should appreciate that a natural sample of an element is likely to contain a mixture of two or more isotopes. In determining the atomic mass of the element we must take into account that it is a mixture of isotopes with diff ...
Moseley`s law refuted
... From Moseley’s data it is impossible to infer that atoms possess Z extra-nuclear electrons, electrons that are arranged in shells or orbital’s. Also impossible to infer that there are atomic nuclei with Z protons. Fame of Moseley Soon after Rutherford's landmark experiment of discovering the proton ...
... From Moseley’s data it is impossible to infer that atoms possess Z extra-nuclear electrons, electrons that are arranged in shells or orbital’s. Also impossible to infer that there are atomic nuclei with Z protons. Fame of Moseley Soon after Rutherford's landmark experiment of discovering the proton ...
Amines
... The basic properties of amines A base is • an electron donor. • it is the lone pair on the nitrogen that gives amines their basic properties ...
... The basic properties of amines A base is • an electron donor. • it is the lone pair on the nitrogen that gives amines their basic properties ...
View PDF - CiteSeerX
... calculations of this type one carries out a kind of mathematical experiment, results of which could be compared with the real physical experimental data. The successful comparison convinces us once more that quantum mechanics is a valid theory being capable of reproducing reality in its quantitative ...
... calculations of this type one carries out a kind of mathematical experiment, results of which could be compared with the real physical experimental data. The successful comparison convinces us once more that quantum mechanics is a valid theory being capable of reproducing reality in its quantitative ...
Briefing Session on 2012 HKDSE Examination (December 2012)
... involving calculation and data analysis. These include mass/mole/concentration calculations for a titration experiment, calculation of the enthalpy changes of reactions, and calculations on chemical equilibrium. Many candidates were weak in redox chemistry. They were confused about the concept ...
... involving calculation and data analysis. These include mass/mole/concentration calculations for a titration experiment, calculation of the enthalpy changes of reactions, and calculations on chemical equilibrium. Many candidates were weak in redox chemistry. They were confused about the concept ...
Detailed TOC
... 3.3 Representing Compounds: Chemical Formulas and Molecular Models Types of Chemical Formulas Molecular Models 3.4 An Atomic-Level Perspective of Elements and Compounds 3.5 Ionic Compounds: Formulas and Names Writing Formulas for Ionic Compounds Naming Ionic Compounds Naming Binary Ionic Compounds N ...
... 3.3 Representing Compounds: Chemical Formulas and Molecular Models Types of Chemical Formulas Molecular Models 3.4 An Atomic-Level Perspective of Elements and Compounds 3.5 Ionic Compounds: Formulas and Names Writing Formulas for Ionic Compounds Naming Ionic Compounds Naming Binary Ionic Compounds N ...
chemistry
... questions or answers prior to the examination and that you have neither given nor received assistance in answering any of the questions during the examination. Your answer sheet and answer booklet cannot be accepted if you fail to sign this ...
... questions or answers prior to the examination and that you have neither given nor received assistance in answering any of the questions during the examination. Your answer sheet and answer booklet cannot be accepted if you fail to sign this ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... throughout a given sample and from one sample to another. A chemical element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into s ...
... throughout a given sample and from one sample to another. A chemical element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into s ...
George Facer`s A level Chemistry
... under reflux because ammonia gas would be liberated. This would then escape because it would not be condensed by the reflux condenser. The halogenoalkane and the ammonia solution must therefore be heated in a sealed container. Alternatively, a concentrated ammonia solution can be used and the mixtur ...
... under reflux because ammonia gas would be liberated. This would then escape because it would not be condensed by the reflux condenser. The halogenoalkane and the ammonia solution must therefore be heated in a sealed container. Alternatively, a concentrated ammonia solution can be used and the mixtur ...
AP CHEMISTRY COURSE SYLLABUS
... radii, Ionic radii, electron affinities. Reasons for oxidation numbers based on Quantum model ...
... radii, Ionic radii, electron affinities. Reasons for oxidation numbers based on Quantum model ...
Comparison of 2008 to 2000 SCH3U_ud
... C2.3 investigate synthesis, decomposition, single displacement, and double displacement reactions, by testing the products of each reaction (e.g., test for products such as gases, the presence of an acid, or the presence of a base) [PR, AI] C2.4 predict the products of different types of synthesis ...
... C2.3 investigate synthesis, decomposition, single displacement, and double displacement reactions, by testing the products of each reaction (e.g., test for products such as gases, the presence of an acid, or the presence of a base) [PR, AI] C2.4 predict the products of different types of synthesis ...
Chemistry II Aqueous Reactions and Solution Chemistry Chapter 4
... carries a partial negative charge. The hydrogen side of the molecule has a partial positive charge. ...
... carries a partial negative charge. The hydrogen side of the molecule has a partial positive charge. ...
Multiscale Modeling of Biological Functions: From Enzymes to Molecular Machines
... small part of a molecular quantum mechanically, while representing the rest classically. I felt that this should allow me to finally make a progress towards my old dream of studying enzymes. At that time, Mike returned from his PhD at the Medical Research Council (MRC) to the Weizmann Institute, and ...
... small part of a molecular quantum mechanically, while representing the rest classically. I felt that this should allow me to finally make a progress towards my old dream of studying enzymes. At that time, Mike returned from his PhD at the Medical Research Council (MRC) to the Weizmann Institute, and ...
Implementation of the SCC-DFTB Method for
... if desired. Similarly, while the default link atom to be used is a hydrogen atom, this can be changed by the user at run time as necessary. The electrostatic interactions of the MM link pair atom are replaced by those of the link atom, while the van der Waals interactions remain with the MM link pai ...
... if desired. Similarly, while the default link atom to be used is a hydrogen atom, this can be changed by the user at run time as necessary. The electrostatic interactions of the MM link pair atom are replaced by those of the link atom, while the van der Waals interactions remain with the MM link pai ...
answers to part a of the national high school
... manganese can be extracted from a particular ore, although, it has to be said, that braunite is not commonly used as a source of manganese. Note that the formulae given for minerals often uses the convention in which the oxide component is given before any other constituent of the mineral, thus brau ...
... manganese can be extracted from a particular ore, although, it has to be said, that braunite is not commonly used as a source of manganese. Note that the formulae given for minerals often uses the convention in which the oxide component is given before any other constituent of the mineral, thus brau ...
Aqueous Reactions and Solution Stoichiometry (Chapter 4)
... Some substances dissociate (split apart) in solution to form ions. Since such solutions can conduct an electric current, the dissolved substance is classified as an electrolyte. These substances consist of ions that are held together by the electrostatic forces of attraction between their opposite c ...
... Some substances dissociate (split apart) in solution to form ions. Since such solutions can conduct an electric current, the dissolved substance is classified as an electrolyte. These substances consist of ions that are held together by the electrostatic forces of attraction between their opposite c ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.