QUANTUM THEORY OF ATOMS AND MOLECULES
... energy from the ground to the first excited vibrational states. 3. Calculate the difference between the zero-point energy (in kJ mol) of C-H and C-D bond stretches, given a C-H vibrational stretching frequency of 2900 cm. Hence explain why C-H bonds react more rapidly than C-D bonds in many orga ...
... energy from the ground to the first excited vibrational states. 3. Calculate the difference between the zero-point energy (in kJ mol) of C-H and C-D bond stretches, given a C-H vibrational stretching frequency of 2900 cm. Hence explain why C-H bonds react more rapidly than C-D bonds in many orga ...
to Ch 3.1_Atoms_The Building Blocks of Matter
... 3.1 The Atom: From Philosophical Idea to Scientific Theory Dalton’s Atomic Theory • All matter is composed of extremely small particles called atoms. • Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. ...
... 3.1 The Atom: From Philosophical Idea to Scientific Theory Dalton’s Atomic Theory • All matter is composed of extremely small particles called atoms. • Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. ...
Electron Configurations
... • Where the electrons are in the energy levels and orbitals. • The configuration that requires the least energy is the most stable - called groundstate electron configuration. • 3 specific rules are used to find an atom’s electron configuration: – Aufbau principle (German for build up) – Pauli exclu ...
... • Where the electrons are in the energy levels and orbitals. • The configuration that requires the least energy is the most stable - called groundstate electron configuration. • 3 specific rules are used to find an atom’s electron configuration: – Aufbau principle (German for build up) – Pauli exclu ...
File
... Atoms can’t be broken into smaller pieces. Atoms are indivisible. In any element, all the atoms are exactly alike. Atoms of different elements are different. Atoms of two or more elements can combine to form compounds. Atoms of each element have a unique mass. The masses of the elements in a compoun ...
... Atoms can’t be broken into smaller pieces. Atoms are indivisible. In any element, all the atoms are exactly alike. Atoms of different elements are different. Atoms of two or more elements can combine to form compounds. Atoms of each element have a unique mass. The masses of the elements in a compoun ...
CHAPTER 4: Structure of the Atom
... The Bohr Model of the Hydrogen Atom Successes and Failures of the Bohr Model Characteristic X-Ray Spectra and Atomic Number Atomic Excitation by Electrons In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theor ...
... The Bohr Model of the Hydrogen Atom Successes and Failures of the Bohr Model Characteristic X-Ray Spectra and Atomic Number Atomic Excitation by Electrons In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to Planck’s theor ...
Praxis II Chemistry prep
... 1. In the following pairs of ions, identify which is most likely to exist and explain why you think so. Ca2+ or Ca2S2+ or S24. Why is the radius of a positive ion smaller than the radius of its parent atom? ...
... 1. In the following pairs of ions, identify which is most likely to exist and explain why you think so. Ca2+ or Ca2S2+ or S24. Why is the radius of a positive ion smaller than the radius of its parent atom? ...
chapter_2_2009
... – When an atom gives away an electron, it ends up with more protons than electrons and gains a positive charge; cation – When an atom accepts an electron, it ends up with more electrons than protons and gains a negative charge; anion – This process is called ionization. An ionic bond – The attractio ...
... – When an atom gives away an electron, it ends up with more protons than electrons and gains a positive charge; cation – When an atom accepts an electron, it ends up with more electrons than protons and gains a negative charge; anion – This process is called ionization. An ionic bond – The attractio ...
What is Chemistry? Chemistry
... _______________________________________, commonly known as table salt. A _____________________________ is formed when valence electrons of atoms interact and become stable. Ionic Compounds – substances composed of cations and anions forming ionic bonds. Certain combinations of elements form io ...
... _______________________________________, commonly known as table salt. A _____________________________ is formed when valence electrons of atoms interact and become stable. Ionic Compounds – substances composed of cations and anions forming ionic bonds. Certain combinations of elements form io ...
εn = ε KE + ε PE = ε PE ε PE = ε PE (1 )
... REPULSION which will occur when 2 electrons are placed in the SAME SPATIAL ORBITAL -- such REPULSION would RAISE the energy of the atom This result is summarised in HUND'S RULE: Other things being equal, THE STATE OF LOWEST ENERGY corresponds to the MAXIMUM NUMBER OF UNPAIRED, PARALLEL SPINS Thus th ...
... REPULSION which will occur when 2 electrons are placed in the SAME SPATIAL ORBITAL -- such REPULSION would RAISE the energy of the atom This result is summarised in HUND'S RULE: Other things being equal, THE STATE OF LOWEST ENERGY corresponds to the MAXIMUM NUMBER OF UNPAIRED, PARALLEL SPINS Thus th ...
Summary from Organic Chemistry Packet:
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
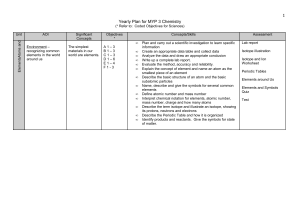
Yearly Plan for MYP 1 Science
... Interpret chemical notation for elements, atomic number, mass number, charge and how many atoms Describe the term isotope and illustrate an isotope, showing its protons, neutrons and electrons Describe the Periodic Table and how it is organized Identify products and reactants. Give the symbols for s ...
... Interpret chemical notation for elements, atomic number, mass number, charge and how many atoms Describe the term isotope and illustrate an isotope, showing its protons, neutrons and electrons Describe the Periodic Table and how it is organized Identify products and reactants. Give the symbols for s ...
O - gearju.com
... and look up the values in Figure 9.5. Solution (a) The electronegativity difference between H and Cl is 0.9, which is appreciable but not large enough (by the 2.0 rule) to qualify HCl as an ionic compound. Therefore, the bond between H and Cl is polar covalent. (b) The electronegativity difference b ...
... and look up the values in Figure 9.5. Solution (a) The electronegativity difference between H and Cl is 0.9, which is appreciable but not large enough (by the 2.0 rule) to qualify HCl as an ionic compound. Therefore, the bond between H and Cl is polar covalent. (b) The electronegativity difference b ...
O - gearju.com
... and look up the values in Figure 9.5. Solution (a) The electronegativity difference between H and Cl is 0.9, which is appreciable but not large enough (by the 2.0 rule) to qualify HCl as an ionic compound. Therefore, the bond between H and Cl is polar covalent. (b) The electronegativity difference b ...
... and look up the values in Figure 9.5. Solution (a) The electronegativity difference between H and Cl is 0.9, which is appreciable but not large enough (by the 2.0 rule) to qualify HCl as an ionic compound. Therefore, the bond between H and Cl is polar covalent. (b) The electronegativity difference b ...
On the Sympathetic Cooling of Atomic and Molecular Ions with
... very efficiently by collisions with the ultracold atoms in a high vacuum environment. The method is very general and can be used in cases where direct laser translational cooling of molecular ions is impractical. Once cooled, precision Doppler free spectroscopy and studies of reactions of molecular ...
... very efficiently by collisions with the ultracold atoms in a high vacuum environment. The method is very general and can be used in cases where direct laser translational cooling of molecular ions is impractical. Once cooled, precision Doppler free spectroscopy and studies of reactions of molecular ...
Chapter 2
... Atomic Number and Atomic Mass • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be appro ...
... Atomic Number and Atomic Mass • Atoms of the various elements differ in number of subatomic particles • An element’s atomic number is the number of protons in its nucleus • An element’s mass number is the sum of protons plus neutrons in the nucleus • Atomic mass, the atom’s total mass, can be appro ...
SLE133 – “Chemistry in Our World” Summary Notes Week 1
... A uniform Mixture having the same composition ...
... A uniform Mixture having the same composition ...
Electronic structure (download)
... The Quantum Mechanics: waves of uncertainty System developed that incorporated these concepts and produced an orbital picture of the electrons No longer think of electrons as particles with precise location, but as waves which have probability of being in some region of the atom – the orbital Impos ...
... The Quantum Mechanics: waves of uncertainty System developed that incorporated these concepts and produced an orbital picture of the electrons No longer think of electrons as particles with precise location, but as waves which have probability of being in some region of the atom – the orbital Impos ...
Final Exam - Seattle Central College
... – Recognize that London forces increase with more electrons—use size to determine relative number of electrons for different molecules. – Know the terms: evaporation, boiling point, vapor pressure, volatile, nonvolatile – Recognize how IMF’s influence vapor pressure and boiling point. – Given differ ...
... – Recognize that London forces increase with more electrons—use size to determine relative number of electrons for different molecules. – Know the terms: evaporation, boiling point, vapor pressure, volatile, nonvolatile – Recognize how IMF’s influence vapor pressure and boiling point. – Given differ ...
5 Electrons in Atoms
... sequence through an element that has not yet been identified, but whose atoms would completely fill 7p orbitals. How many electrons would such an atom have? Write its electron configuration using noble-gas notation for the previous ...
... sequence through an element that has not yet been identified, but whose atoms would completely fill 7p orbitals. How many electrons would such an atom have? Write its electron configuration using noble-gas notation for the previous ...
THE PERIODIC TABLE abbr
... Helium (2 valence electrons) is in the same column as neon (8 valence electrons) because both have full outer energy levels. This gives them similar properties. ...
... Helium (2 valence electrons) is in the same column as neon (8 valence electrons) because both have full outer energy levels. This gives them similar properties. ...
Chemistry Readings
... When a metal reacts with a non-metal, the metal will lose electrons to form a positive ion while the non-metal will gain electrons forming a negative ion. Together they form an ionic compound. This is the reaction between Magnesium and Oxygen. Magnesium is in Group IIA. A Magnesium atom will lose 2 ...
... When a metal reacts with a non-metal, the metal will lose electrons to form a positive ion while the non-metal will gain electrons forming a negative ion. Together they form an ionic compound. This is the reaction between Magnesium and Oxygen. Magnesium is in Group IIA. A Magnesium atom will lose 2 ...
Symbols of Elements
... tunneling microscope (STM). This instrument generates an image of the atomic structure. ...
... tunneling microscope (STM). This instrument generates an image of the atomic structure. ...
Semester Exam Practice Questions
... 48. The formula mass of magnesium chloride, MgCl2, is __________. a. 59.8 amu c. 95.2 amu b. 76.4 amu d. 125.8 amu 49. If one molecule of NH3 has a mass of 17.0 g/mol, what is the mass of 6.02 x 1023 molecules of NH3? a. 2.82 g c. 102 g b. 17.0 g d. 2.82 x 10-25 g 50. Which of the following statemen ...
... 48. The formula mass of magnesium chloride, MgCl2, is __________. a. 59.8 amu c. 95.2 amu b. 76.4 amu d. 125.8 amu 49. If one molecule of NH3 has a mass of 17.0 g/mol, what is the mass of 6.02 x 1023 molecules of NH3? a. 2.82 g c. 102 g b. 17.0 g d. 2.82 x 10-25 g 50. Which of the following statemen ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.