An Introduction to Chemistry
... enough to be seen with an optical microscope is considered to be microscopic. • MACROSCOPIC: Anything that is large enough to be seen with the naked eye is considered to be macroscopic. ...
... enough to be seen with an optical microscope is considered to be microscopic. • MACROSCOPIC: Anything that is large enough to be seen with the naked eye is considered to be macroscopic. ...
Quantum no and orbitals
... Aufbau Principle • Aufbau Principle: an electron will occupy the lowest-energy orbital that can receive it. ...
... Aufbau Principle • Aufbau Principle: an electron will occupy the lowest-energy orbital that can receive it. ...
Detection and Quantized Conductance of Neutral Atoms Near a Charged... Trygve Ristroph, Anne Goodsell, J. A. Golovchenko, and Lene Vestergaard...
... The evolving ability to fabricate rigid micrometer and even nanometer scale structures allows for studies and utilization of atomic physics phenomena at length scales well below optical and cold-atom de Broglie wavelengths and for the miniaturization of atom traps and guides [1–7]. The success of ma ...
... The evolving ability to fabricate rigid micrometer and even nanometer scale structures allows for studies and utilization of atomic physics phenomena at length scales well below optical and cold-atom de Broglie wavelengths and for the miniaturization of atom traps and guides [1–7]. The success of ma ...
s 1
... In excited state, one or both electrons will be in higher shell (e.g., 1s12s1). Configuration must therefore be written in terms of particle #1 in a state defined by four quantum numbers (called ). State of particle #2 called . ...
... In excited state, one or both electrons will be in higher shell (e.g., 1s12s1). Configuration must therefore be written in terms of particle #1 in a state defined by four quantum numbers (called ). State of particle #2 called . ...
Intermolecular and Ionic Forces
... (ice). This kit is specifically designed to show the difference between covalent bonds O-H bonds within a water molecule and the hydrogen bridges holding two different H2O molecules together. Groups will rotate through this station. The water molecule models must stay intact. After you are finished ...
... (ice). This kit is specifically designed to show the difference between covalent bonds O-H bonds within a water molecule and the hydrogen bridges holding two different H2O molecules together. Groups will rotate through this station. The water molecule models must stay intact. After you are finished ...
Metal-Ligand and Metal-Metal Bonding Lecture Notes
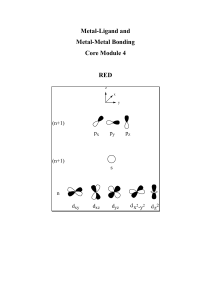
... higher energy starting orbital character). When we talk about splitting of metal ‘d-orbitals’ in crystal field theory we are ignoring the ligand character that is present in some of the ‘d-orbitals’, however it is still a good first approximation and the relative energies between d-orbitals are corr ...
... higher energy starting orbital character). When we talk about splitting of metal ‘d-orbitals’ in crystal field theory we are ignoring the ligand character that is present in some of the ‘d-orbitals’, however it is still a good first approximation and the relative energies between d-orbitals are corr ...
Chapter 6. Electronic Structure of Atoms
... It cannot explain the spectra of atoms other than hydrogen. ...
... It cannot explain the spectra of atoms other than hydrogen. ...
ExamView - Chap_Test_v2.tst
... compound may be used more than once. a. ionic compound b. covalent compound ____ 23. this compound forms when metals react with non-metals ____ 24. molecules exist in this compound ____ 25. the name of this compound may include Roman numerals ____ 26. electrons are transferred when this compound for ...
... compound may be used more than once. a. ionic compound b. covalent compound ____ 23. this compound forms when metals react with non-metals ____ 24. molecules exist in this compound ____ 25. the name of this compound may include Roman numerals ____ 26. electrons are transferred when this compound for ...
lect2_htm
... This effect is important - avoided crossings are often the reason why chemical reactions have a barrier. The Rule applies to molecular orbitals as well as wavefunctions for whole molecules. For example, consider a situation where a molecule has two molecular orbitals. The energies of these MOs are ...
... This effect is important - avoided crossings are often the reason why chemical reactions have a barrier. The Rule applies to molecular orbitals as well as wavefunctions for whole molecules. For example, consider a situation where a molecule has two molecular orbitals. The energies of these MOs are ...
Notes on Electron Configurations
... field‐emission electron microscopy II. M. Mikhailovskij, E. V. Sadanov, T. I. Mazilova, V. A. Ksenofontov, and M Mikhailovskij E V Sadanov T I Mazilova V A Ksenofontov and O. A. Velicodnaja, Department of Low Temperatures and Condensed State, National Scientific Center, Kharkov Institute for Phys ...
... field‐emission electron microscopy II. M. Mikhailovskij, E. V. Sadanov, T. I. Mazilova, V. A. Ksenofontov, and M Mikhailovskij E V Sadanov T I Mazilova V A Ksenofontov and O. A. Velicodnaja, Department of Low Temperatures and Condensed State, National Scientific Center, Kharkov Institute for Phys ...
Chapter 8
... Na+, Al3+, F-, O2-, and N3- are all isoelectronic with Ne What neutral atom is isoelectronic with H - ? ...
... Na+, Al3+, F-, O2-, and N3- are all isoelectronic with Ne What neutral atom is isoelectronic with H - ? ...
1s 2 2s 2 2p 6 3s 2 3p 3d 4s 4p 4d 4f 5s 5p 5d 5f Ni = 28 e
... To know the maximum amount of electrons that could be in any principal quantum level (and the number of elements that could be represented) use the formula 2n2 if n=1, then 2 electrons will fit ...
... To know the maximum amount of electrons that could be in any principal quantum level (and the number of elements that could be represented) use the formula 2n2 if n=1, then 2 electrons will fit ...
The Complete Notes - Joliet Junior College
... Physical and Chemical Properties – what’s the difference? Analogy: We all posses ‘as is’ physical properties, or characteristics, that define us. For example, Dr. Mills is 5’11” and has green eyes. As with people, each chemical also possesses a unique set of ‘as is’ physical properties that define ...
... Physical and Chemical Properties – what’s the difference? Analogy: We all posses ‘as is’ physical properties, or characteristics, that define us. For example, Dr. Mills is 5’11” and has green eyes. As with people, each chemical also possesses a unique set of ‘as is’ physical properties that define ...
Lesson 2a - Freeman Public Schools
... Electrons closest to the nucleus are most strongly attracted Each shell has distinct properties The number of electrons has an upper limit Shells closest to the nucleus fill first ...
... Electrons closest to the nucleus are most strongly attracted Each shell has distinct properties The number of electrons has an upper limit Shells closest to the nucleus fill first ...
File - Grade 12 Chemistry
... Dispersion forces are very weak intermolecular forces that exist between molecules. When a carbon atom is bonded to another carbon atom, or to a hydrogen atom, the bond is not considered to be polar because the electronegativity difference between carbon atoms is zero and between carbon and hydrogen ...
... Dispersion forces are very weak intermolecular forces that exist between molecules. When a carbon atom is bonded to another carbon atom, or to a hydrogen atom, the bond is not considered to be polar because the electronegativity difference between carbon atoms is zero and between carbon and hydrogen ...
Capacitive Coupling of Atomic Systems to Mesoscopic Conductors
... by the lack of probes which do not share the same environment. The Rydberg atoms could be used as a sensitive probe of these fluctuations that is controllably separated from the device itself. If the atoms are excited into a state with a large angular momentum where the Stark shift is linear, a sing ...
... by the lack of probes which do not share the same environment. The Rydberg atoms could be used as a sensitive probe of these fluctuations that is controllably separated from the device itself. If the atoms are excited into a state with a large angular momentum where the Stark shift is linear, a sing ...
9th class bridge course 74-112
... takes part in a chemical reaction. All the points put forward in Dalton’s atomic theory have been contradicted by modern research, except that atom is the smallest unit of matter, which takes part in a chemical reaction. These particles were affected by the electric and magnetic fields but in the di ...
... takes part in a chemical reaction. All the points put forward in Dalton’s atomic theory have been contradicted by modern research, except that atom is the smallest unit of matter, which takes part in a chemical reaction. These particles were affected by the electric and magnetic fields but in the di ...
Final Exam - KFUPM Faculty List
... B) have very high third ionization energies yes: they all have ns2 configuration, and thus the third ionization cuts out an electron from the core and is thus very high C) gain 2 electrons to attain a noble gas electron configuration no: they lose 2 electrons to get a noble gas configuration (double ...
... B) have very high third ionization energies yes: they all have ns2 configuration, and thus the third ionization cuts out an electron from the core and is thus very high C) gain 2 electrons to attain a noble gas electron configuration no: they lose 2 electrons to get a noble gas configuration (double ...
Chemistry 14C Winter 2017 Final Exam Part A Solutions Page 1
... (a) Yes (b) No (c) Yes (d) Yes (e) No. X-ray crystallography reveals the positions of atoms in space. Anything which can be proven from these positions can be proven/verified. X-ray crystallography cannot 'see' or count electrons, so anything dependent on knowing electron locations cannot be proven. ...
... (a) Yes (b) No (c) Yes (d) Yes (e) No. X-ray crystallography reveals the positions of atoms in space. Anything which can be proven from these positions can be proven/verified. X-ray crystallography cannot 'see' or count electrons, so anything dependent on knowing electron locations cannot be proven. ...
Prediction of a Metastable Helium Compound: HHeF
... repulsive state corresponding to HF + Ng. Thus, a higher kinetic barrier may arise from a less repulsive state or a stronger NgH+F- ion-pair interaction. Since helium has a significantly higher ionization potential than neon, it is rather surprising that HHeF is more stable than HNeF. The unusual st ...
... repulsive state corresponding to HF + Ng. Thus, a higher kinetic barrier may arise from a less repulsive state or a stronger NgH+F- ion-pair interaction. Since helium has a significantly higher ionization potential than neon, it is rather surprising that HHeF is more stable than HNeF. The unusual st ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.