Mole Relationships in chemistry
... the atoms in one of more substances O Chemical equation represents this process with the reactants on the left side of the arrow and the products on the right side of the arrow ...
... the atoms in one of more substances O Chemical equation represents this process with the reactants on the left side of the arrow and the products on the right side of the arrow ...
Ch. 10 – Stoichiometry Stoichiometry – relates molar ratios between
... The coefficients of a balanced equation relate the moles (numbers) of any compound to the moles (numbers) of any other compound in the equation. These molar ratios are used to 'convert' between any two compounds, whether they are reactants or products. This allows us to calculate moles of reac ...
... The coefficients of a balanced equation relate the moles (numbers) of any compound to the moles (numbers) of any other compound in the equation. These molar ratios are used to 'convert' between any two compounds, whether they are reactants or products. This allows us to calculate moles of reac ...
Nomenclature
... • Some compounds contain H2O in their structure. These compounds are called hydrates. • This is different from (aq) because the H2O is part of the molecule (not just surrounding it). • The H2O can usually be removed if heated. • A dot separates water: e.g. CuSO4•5H2O is ...
... • Some compounds contain H2O in their structure. These compounds are called hydrates. • This is different from (aq) because the H2O is part of the molecule (not just surrounding it). • The H2O can usually be removed if heated. • A dot separates water: e.g. CuSO4•5H2O is ...
Microsoft Word
... All acetates are soluble except for Be(CH3COO)2 All phosphates are insoluble except for those of Group I elements and NH4+. All carbonates are insoluble except for those of Group I elements and NH4+. All hydroxides are insoluble except for those of NH4+, Group I, Sr(OH)2, and Ba(OH)2; Ca(OH)2 is sli ...
... All acetates are soluble except for Be(CH3COO)2 All phosphates are insoluble except for those of Group I elements and NH4+. All carbonates are insoluble except for those of Group I elements and NH4+. All hydroxides are insoluble except for those of NH4+, Group I, Sr(OH)2, and Ba(OH)2; Ca(OH)2 is sli ...
Unit Powerpoint
... In the Table of Standard Reduction Potentials that zinc has a negative E° indicating that it is not as good at competing for electrons as hydrogen. Zn2+(aq) + 2e- → Zn(s) E° = -0.76 V Therefore if zinc and hydrogen are paired together in an electrochemical cell, the hydrogen would be reduced (gain t ...
... In the Table of Standard Reduction Potentials that zinc has a negative E° indicating that it is not as good at competing for electrons as hydrogen. Zn2+(aq) + 2e- → Zn(s) E° = -0.76 V Therefore if zinc and hydrogen are paired together in an electrochemical cell, the hydrogen would be reduced (gain t ...
The regularities of the Rydberg energy levels of many
... separately in one-valence electron atoms (ions); while in WBEPM theory, through separation of the weakest bound electron and non-weakest bound electrons, one can solve the one-electron Schrödinger equation of the weakest bound electron. This is very convenient for the discussion about the excited st ...
... separately in one-valence electron atoms (ions); while in WBEPM theory, through separation of the weakest bound electron and non-weakest bound electrons, one can solve the one-electron Schrödinger equation of the weakest bound electron. This is very convenient for the discussion about the excited st ...
Chapter 3 - Whitwell High School
... One of the most spectacular reactions of aluminum, the thermite reaction, is with iron oxide, Fe2O3, by which metallic iron is made. So much heat is generated that the iron forms in the liquid state. A certain welding operation requires at least 86.0 g of Fe be produced. What is the minimum mass in ...
... One of the most spectacular reactions of aluminum, the thermite reaction, is with iron oxide, Fe2O3, by which metallic iron is made. So much heat is generated that the iron forms in the liquid state. A certain welding operation requires at least 86.0 g of Fe be produced. What is the minimum mass in ...
An Introduction to Redox
... The students will develop an understanding of the structure of atoms, compounds, chemical reactions, and the interactions of energy and matter. Benchmark 3: The student will gain a basic concept of chemical reactions. The student … 1. understands a chemical reaction occurs when one or more subs ...
... The students will develop an understanding of the structure of atoms, compounds, chemical reactions, and the interactions of energy and matter. Benchmark 3: The student will gain a basic concept of chemical reactions. The student … 1. understands a chemical reaction occurs when one or more subs ...
Topic 4 Chemistry of the Elements of the Main Group
... H can form hydrides (negatively charged H) with a whole range of polarity, from ionic hydrides Cs-H ( = 1.4) to non-polar covalent hydrides B-H ( = 0.1). H can form hydrogen compounds (H is positively charged) with a whole range of polarity, from ionic hydrogen halides HF ( = 1.9) to non-polar ...
... H can form hydrides (negatively charged H) with a whole range of polarity, from ionic hydrides Cs-H ( = 1.4) to non-polar covalent hydrides B-H ( = 0.1). H can form hydrogen compounds (H is positively charged) with a whole range of polarity, from ionic hydrogen halides HF ( = 1.9) to non-polar ...
Solution Preparation Final Goueth
... 31. A self-contained breathing apparatus uses potassium superoxide, KO 2, to convert the carbon dioxide and water in exhaled air into oxygen, as shown by the equation. 4 KO2(s) + 2 H2O (g) + 4 CO2(s) ---> 4 KHCO3(s) + 3 O2 (g) How many molecules of oxygen gas will be produced from the 0.0468 g of ca ...
... 31. A self-contained breathing apparatus uses potassium superoxide, KO 2, to convert the carbon dioxide and water in exhaled air into oxygen, as shown by the equation. 4 KO2(s) + 2 H2O (g) + 4 CO2(s) ---> 4 KHCO3(s) + 3 O2 (g) How many molecules of oxygen gas will be produced from the 0.0468 g of ca ...
LAB- Beanium_CP Chemistry
... asked to gather data about the “isotopes” and organize the data. If atoms were as large as beans they could be sorted, counted, and massed. In this experiment we will sort, count, and mass three different kinds of beans and imagine that we are observing different isotopes of the same element (let’s ...
... asked to gather data about the “isotopes” and organize the data. If atoms were as large as beans they could be sorted, counted, and massed. In this experiment we will sort, count, and mass three different kinds of beans and imagine that we are observing different isotopes of the same element (let’s ...
Chemistry - School District of Springfield Township
... o Experiments have revealed that the mass of an atom is concentrated in a tiny positively charged nucleus (consisting of protons and neutrons) with a negative cloud of electrons moving around the nucleus. o The number of protons in the nucleus (and electrons in the cloud) is called the atomic number ...
... o Experiments have revealed that the mass of an atom is concentrated in a tiny positively charged nucleus (consisting of protons and neutrons) with a negative cloud of electrons moving around the nucleus. o The number of protons in the nucleus (and electrons in the cloud) is called the atomic number ...
State Standard - SchoolNotes.com
... Essential Question: How are atoms structured? How can atomic nuclei change? Understand: Chemical elements are the fundamental building materials of matter. Elemental properties are determined by the structure of the nucleus and distribution of electrons. One element can change into another through o ...
... Essential Question: How are atoms structured? How can atomic nuclei change? Understand: Chemical elements are the fundamental building materials of matter. Elemental properties are determined by the structure of the nucleus and distribution of electrons. One element can change into another through o ...
Chapter 7 - HCC Learning Web
... • Have highly negative electron affinities – Exist as anions in nature ...
... • Have highly negative electron affinities – Exist as anions in nature ...
A simple and effective approach to calculate the energy of complex
... On one hand, this is an interesting exercise for nongraduate students. We show as to attain data even for complex atoms that are well compared with the experiment in diverse cases: binding energies, energy of internal (sub) shells and ionization energies of atoms when three or more of the valence el ...
... On one hand, this is an interesting exercise for nongraduate students. We show as to attain data even for complex atoms that are well compared with the experiment in diverse cases: binding energies, energy of internal (sub) shells and ionization energies of atoms when three or more of the valence el ...
New Approaches in Deep Laser Cooling of Magnesium Atoms for
... 33D3). The semiclassical approach (dashed line) gives the minimum kinetic energy of an atom at the level of Emin≈30×Erec, that is several times smaller than the estimated Doppler limit for this transition ED≈87.5×Erec. The corresponding effective temperature is about 150 μK. This is rather far from ...
... 33D3). The semiclassical approach (dashed line) gives the minimum kinetic energy of an atom at the level of Emin≈30×Erec, that is several times smaller than the estimated Doppler limit for this transition ED≈87.5×Erec. The corresponding effective temperature is about 150 μK. This is rather far from ...
H2O - WCCUSD.net
... Chemical reactions involve energy. Sometimes the energy produced by the reaction is more than what goes into making the reaction happen. When that happens, energy is released, usually as heat, and ...
... Chemical reactions involve energy. Sometimes the energy produced by the reaction is more than what goes into making the reaction happen. When that happens, energy is released, usually as heat, and ...
2014 Academic Challenge Sectional Chemistry Exam Solution Set 1
... B. Only the second molecule is polar. In the third and fourth structures, the square planar shape causes any bond dipoles to cancel out. ...
... B. Only the second molecule is polar. In the third and fourth structures, the square planar shape causes any bond dipoles to cancel out. ...
2003
... A current frontier in the field of ultracold gases is the study of ultracold molecules. In 2003, several groups succeeded in converting ultracold atoms into ultracold molecules by magnetically tuning a molecular level close to zero binding energy (Feshbach resonance). Atoms can then form molecules w ...
... A current frontier in the field of ultracold gases is the study of ultracold molecules. In 2003, several groups succeeded in converting ultracold atoms into ultracold molecules by magnetically tuning a molecular level close to zero binding energy (Feshbach resonance). Atoms can then form molecules w ...
7th Chemistry Unit Test Study Guide Test Date: Friday, Nov. 16
... In the following equation, which substances The Pilgrims were researching chemical reactions. They read that if you heat a small amount of calcium carbonate (CaCO3), it will produce calcium oxide (CaO) and carbon ...
... In the following equation, which substances The Pilgrims were researching chemical reactions. They read that if you heat a small amount of calcium carbonate (CaCO3), it will produce calcium oxide (CaO) and carbon ...
AP Chemistry - Oak Park Unified School District
... Write the number next to the word that fits the paragraph. Measurement in Chemistry Science knowledge is advanced by observing patterns, (1), and constructing explanations, (2); which are supported by repeatable (3) evidence. Measurements are made using the metric system, where the standard units ar ...
... Write the number next to the word that fits the paragraph. Measurement in Chemistry Science knowledge is advanced by observing patterns, (1), and constructing explanations, (2); which are supported by repeatable (3) evidence. Measurements are made using the metric system, where the standard units ar ...
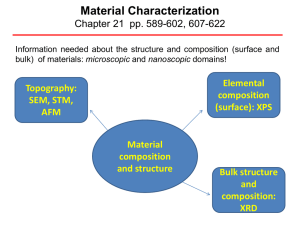
Material Characterization
... X-ray Photoelectron Spectroscopy (XPS) XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its "as received" state, or after some treatment XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot detect hyd ...
... X-ray Photoelectron Spectroscopy (XPS) XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its "as received" state, or after some treatment XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot detect hyd ...
AP CHEMISTRY – Source: 1999 AP Exam CHAPTER 8 TEST
... ANALYSIS: Immediately rule out B, C and E. There is no dipole moment for any of these molecules! C & E b/c they are diatomic. There is NO difference in electronegativity, therefore, no dipole moment can exist! For B, CO2 has a bond dipole for each of the two C=O bonds. However, this is a linear mole ...
... ANALYSIS: Immediately rule out B, C and E. There is no dipole moment for any of these molecules! C & E b/c they are diatomic. There is NO difference in electronegativity, therefore, no dipole moment can exist! For B, CO2 has a bond dipole for each of the two C=O bonds. However, this is a linear mole ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.