ch02 lecture 7e
... PROBLEM: Silicon (Si) has three naturally occurring isotopes: 28Si, 29Si, and 30Si. Determine the number of protons, neutrons, and electrons in each silicon isotope. PLAN: The mass number (A) is given for each isotope and is equal to the number of protons + neutrons. The atomic number Z, found on th ...
... PROBLEM: Silicon (Si) has three naturally occurring isotopes: 28Si, 29Si, and 30Si. Determine the number of protons, neutrons, and electrons in each silicon isotope. PLAN: The mass number (A) is given for each isotope and is equal to the number of protons + neutrons. The atomic number Z, found on th ...
Carbene Singlets, Triplets, and the Physics that
... rest of the molecule. Adapted from Lowry & Schueller, p 560. ...
... rest of the molecule. Adapted from Lowry & Schueller, p 560. ...
+2 - Fort Thomas Independent Schools
... 1. Write formulas for the binary ionic compounds formed between the following elements: a. potassium and iodine b. magnesium and chlorine c. sodium and sulfur d. aluminum and sulfur e. aluminum and nitrogen 2. Name the binary ionic compounds indicated by the following formulas: a. AgCl ...
... 1. Write formulas for the binary ionic compounds formed between the following elements: a. potassium and iodine b. magnesium and chlorine c. sodium and sulfur d. aluminum and sulfur e. aluminum and nitrogen 2. Name the binary ionic compounds indicated by the following formulas: a. AgCl ...
The MOLE
... 1. What is the mass of 4.5 moles of Au? 2. How many atoms of Au will have a mass of 89.60g? 3. How many moles of gold do you have if you have 5.24 x 1026 atoms of gold? 4. What will be the mass of 5.24 x1026 atoms of Gold? ...
... 1. What is the mass of 4.5 moles of Au? 2. How many atoms of Au will have a mass of 89.60g? 3. How many moles of gold do you have if you have 5.24 x 1026 atoms of gold? 4. What will be the mass of 5.24 x1026 atoms of Gold? ...
Lab Stuff:
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
Lab Stuff
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
... What types of substances are removed from mixtures using filtration? Adsorption? Distillation? Think about the foul water lab! ...
Week 2 end - University of Guelph
... Covalent Bonds – bonds resulting from the sharing of electrons between atoms. e.g., Cl – Cl, H–C C–H, H–(C=O)–H The distance between the atoms is the bond length (depends on the atoms). Not all covalent bonds are equal – they involve different nuclei and different electron distributions, (e.g, C-C, ...
... Covalent Bonds – bonds resulting from the sharing of electrons between atoms. e.g., Cl – Cl, H–C C–H, H–(C=O)–H The distance between the atoms is the bond length (depends on the atoms). Not all covalent bonds are equal – they involve different nuclei and different electron distributions, (e.g, C-C, ...
Module P7.1 The atomic basis of matter
... In the latter half of the 19th century, James Clerk Maxwell ☞ developed the first truly systematic and detailed kinetic theory of gases, which allowed for the random motions of individual molecules but nevertheless predicted the distribution of molecular speeds and related the average speed of a mo ...
... In the latter half of the 19th century, James Clerk Maxwell ☞ developed the first truly systematic and detailed kinetic theory of gases, which allowed for the random motions of individual molecules but nevertheless predicted the distribution of molecular speeds and related the average speed of a mo ...
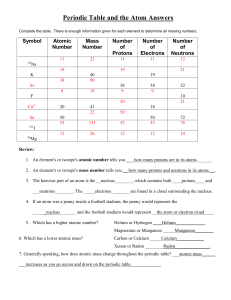
Periodic Table and the Atom Answers
... 13) Which of these elements has the highest first ionization energy? a) oxygen b) oxygen c) fluorine d) carbon e) boron 14) Which of these elements has the highest electronegativity? a) lithium b) nitrogen c) potassium d) arsenic e) beryllium ...
... 13) Which of these elements has the highest first ionization energy? a) oxygen b) oxygen c) fluorine d) carbon e) boron 14) Which of these elements has the highest electronegativity? a) lithium b) nitrogen c) potassium d) arsenic e) beryllium ...
A new Bloch period for interacting cold atoms in 1D optical lattices
... The response of a quantum system to a static field has been a longstanding problem since the early days of quantum mechanics. A topic of particular interest in this wide field is the dynamics of a quantum particle in a periodic potential induced by a static force (modelling a crystal electron in an ...
... The response of a quantum system to a static field has been a longstanding problem since the early days of quantum mechanics. A topic of particular interest in this wide field is the dynamics of a quantum particle in a periodic potential induced by a static force (modelling a crystal electron in an ...
UV Spectroscopy
... - orbitals are the lowest energy occupied molecular orbitals * - orbitals are the highest energy unoccupied molecular orbitals - orbitals are of somewhat higher energy occupied molecular orbitals * - orbitals are lower in energy (unoccupied molecular orbitals) than * n - orbitals; Unshared pa ...
... - orbitals are the lowest energy occupied molecular orbitals * - orbitals are the highest energy unoccupied molecular orbitals - orbitals are of somewhat higher energy occupied molecular orbitals * - orbitals are lower in energy (unoccupied molecular orbitals) than * n - orbitals; Unshared pa ...
CHEMISTRY OF MAIN GROUP ELEMENTS Classification -1 s
... Structures of acids to be given here. Phosphorous forms Phosphorous trioxide P4O6 and Phosphorous pentoxideP4O10 Phosphorus does not enter into pπ - pπ bonds as in nitrogen dioxide P4O6 Structure of above Compounds. ...
... Structures of acids to be given here. Phosphorous forms Phosphorous trioxide P4O6 and Phosphorous pentoxideP4O10 Phosphorus does not enter into pπ - pπ bonds as in nitrogen dioxide P4O6 Structure of above Compounds. ...
2007 - SAASTA
... other trace gases that, in concentration in the atmosphere, can create a strong greenhouse ...
... other trace gases that, in concentration in the atmosphere, can create a strong greenhouse ...
Electrochemistry Oxidation – Reduction and Oxidation Numbers
... 4. Monatomic ions are assigned oxidation numbers equal to the ion charge. e.g., Na+ = +1, Al3+ = +3, Cl- = -1, N3- = -3 5. Oxygen in a compound or ion usually has an oxidation state of –2. (Peroxides are the exception, in which case the oxidation number is –1.) 6. Hydrogen in a compound or ion usual ...
... 4. Monatomic ions are assigned oxidation numbers equal to the ion charge. e.g., Na+ = +1, Al3+ = +3, Cl- = -1, N3- = -3 5. Oxygen in a compound or ion usually has an oxidation state of –2. (Peroxides are the exception, in which case the oxidation number is –1.) 6. Hydrogen in a compound or ion usual ...
The Quantum-Mechanical Model of the Atom
... 1. No electrons are emitted if the frequency of light used is less than νo, regardless of the intensity of the light. 2. For light with a frequency≥ νo , electrons are emitted. The number of electrons increases with the intensity of the light. 3. For light with a frequency > νo , the electrons are e ...
... 1. No electrons are emitted if the frequency of light used is less than νo, regardless of the intensity of the light. 2. For light with a frequency≥ νo , electrons are emitted. The number of electrons increases with the intensity of the light. 3. For light with a frequency > νo , the electrons are e ...
University of Lusaka
... An atom consists of a nucleus of protons and neutrons, surrounded by electrons. Each of the elements in the periodic table is classified according to its atomic number, which is the number of protons in that element's nucleus. Protons have a charge of +1, electrons have a charge of -1, and neutrons ...
... An atom consists of a nucleus of protons and neutrons, surrounded by electrons. Each of the elements in the periodic table is classified according to its atomic number, which is the number of protons in that element's nucleus. Protons have a charge of +1, electrons have a charge of -1, and neutrons ...
Science 10 - SharpSchool
... 1. metals are good conductors, strong, malleable (pound into thin sheet), ductile (can draw into a wire, bendable), have high luster; are found on left side of stair case 2. non metals are poor conductors, non-lustrous, weak, etc…opposite properties to metals; found on right side of ...
... 1. metals are good conductors, strong, malleable (pound into thin sheet), ductile (can draw into a wire, bendable), have high luster; are found on left side of stair case 2. non metals are poor conductors, non-lustrous, weak, etc…opposite properties to metals; found on right side of ...
Chemistry Review
... save yourself a lot of trouble later if you learn their formulas AND charges now: ...
... save yourself a lot of trouble later if you learn their formulas AND charges now: ...
Quantum Chemistry and Spectroscopy
... The description is mostly valid to any C=O group. Below is a table of few common chromophores. As one can see most of them are on the UV range. Well we know that, most of organic compounds are transparent. And UV radiation, especially strong UV, will broke most molecules. (Visible light 400 -700 nm ...
... The description is mostly valid to any C=O group. Below is a table of few common chromophores. As one can see most of them are on the UV range. Well we know that, most of organic compounds are transparent. And UV radiation, especially strong UV, will broke most molecules. (Visible light 400 -700 nm ...
TITLE: Molecules star in quantum movie STANDFIRST: The
... Jönssen, using a copper grating with a period of 1 µm (fig. 1a)3,4, and the build-up of the interference pattern when there was only one electron in the apparatus at a time was first recorded by a group in Bologna5 (1976) and later by a group at Hitachi6 (1989). Both the Bologna and Hitachi groups a ...
... Jönssen, using a copper grating with a period of 1 µm (fig. 1a)3,4, and the build-up of the interference pattern when there was only one electron in the apparatus at a time was first recorded by a group in Bologna5 (1976) and later by a group at Hitachi6 (1989). Both the Bologna and Hitachi groups a ...
BeaniumIsotopeLab
... 1. Was the average mass of the beans a whole number or a decimal? Explain. 2. Explain any differences between the atomic mass of your BEANIUM sample and that of a different lab group. Explain why the difference would be smaller if larger samples were used. 3. What is an isotope? 4. What is the relat ...
... 1. Was the average mass of the beans a whole number or a decimal? Explain. 2. Explain any differences between the atomic mass of your BEANIUM sample and that of a different lab group. Explain why the difference would be smaller if larger samples were used. 3. What is an isotope? 4. What is the relat ...
On the transverse mode of an atom laser
... Research on atom lasers is an active and fascinating area in atomic physics [1, 2]. Several laboratories around the world are now using continuous output couplers to produce atom laser beams from Bose-Einstein condensates. It is therefore important to characterize the qualities of these beams. Recen ...
... Research on atom lasers is an active and fascinating area in atomic physics [1, 2]. Several laboratories around the world are now using continuous output couplers to produce atom laser beams from Bose-Einstein condensates. It is therefore important to characterize the qualities of these beams. Recen ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.