1985 Free Response Answers
... 1) At 25 °C the solubility product constant, Ksp, for strontium sulfate, SrSO4, is 7.6 x 10¯7. The solubility product constant for strontium fluoride, SrF2, is 7.9 x 10¯10 (a) What is the molar solubility of SrSO4 in pure water at 25 °C? (b) what is the molar solubility of SrF2 in pure water at 25 ° ...
... 1) At 25 °C the solubility product constant, Ksp, for strontium sulfate, SrSO4, is 7.6 x 10¯7. The solubility product constant for strontium fluoride, SrF2, is 7.9 x 10¯10 (a) What is the molar solubility of SrSO4 in pure water at 25 °C? (b) what is the molar solubility of SrF2 in pure water at 25 ° ...
File
... equilibrium constant is changed. When the skeleton equation above is balanced and 27. The critical temperature of a substance is the all coefficients reduced to their lowest whole(A) temperature at which the vapor pressure of number terms, what is the coefficient for H+? the liquid is equal to the e ...
... equilibrium constant is changed. When the skeleton equation above is balanced and 27. The critical temperature of a substance is the all coefficients reduced to their lowest whole(A) temperature at which the vapor pressure of number terms, what is the coefficient for H+? the liquid is equal to the e ...
Heats of Formation WS
... Heats of Formation 1. For each of the following compounds, write a balanced thermochemical equation depicting the formation of one mole of the compound from its elements in their standard states and use the appendix to obtain the value of ∆Hfº. [a] NO2 (g) ...
... Heats of Formation 1. For each of the following compounds, write a balanced thermochemical equation depicting the formation of one mole of the compound from its elements in their standard states and use the appendix to obtain the value of ∆Hfº. [a] NO2 (g) ...
Lecture 2
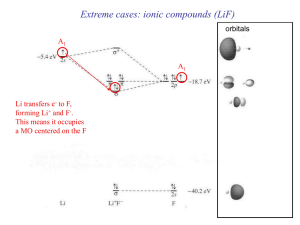
... or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bonding ...
... or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bonding ...
Hydrothermal Reactions from Sodium Hydrogen Carbonate to Phenol
... However, the information on how these organic molecules formed from the simplest inorganic molecules is still at a very beginning stage and defies clarity. Abiotic synthesis of organic compounds from CO2 under hydrothermal conditions has been proposed as a source of the precursor compounds from whic ...
... However, the information on how these organic molecules formed from the simplest inorganic molecules is still at a very beginning stage and defies clarity. Abiotic synthesis of organic compounds from CO2 under hydrothermal conditions has been proposed as a source of the precursor compounds from whic ...
August 2007
... A student constructed an electrochemical cell as shown. The aqueous cell solutions had a concentration of 1 mol/L with respect to the metal ions present. The solution in one half-cell is initially an orange colour due to the mixture of the pale green Fe2+ ions and the orange Fe3+ ions. The other hal ...
... A student constructed an electrochemical cell as shown. The aqueous cell solutions had a concentration of 1 mol/L with respect to the metal ions present. The solution in one half-cell is initially an orange colour due to the mixture of the pale green Fe2+ ions and the orange Fe3+ ions. The other hal ...
Gas-Forming reactions Reactions that form a
... Gas-Forming reactions Reactions that form a gas as one of the products are gas-forming reaction. Most common examples involve metal carbonates and acids. CaCO3 + 2CH3COOH(aq) → Ca(CH3COO)2(aq) + H2CO3(aq) H2CO3(aq) → Overall equation Ionic equation: Net ionic equation: ...
... Gas-Forming reactions Reactions that form a gas as one of the products are gas-forming reaction. Most common examples involve metal carbonates and acids. CaCO3 + 2CH3COOH(aq) → Ca(CH3COO)2(aq) + H2CO3(aq) H2CO3(aq) → Overall equation Ionic equation: Net ionic equation: ...
Checking the Kinetics of Acetic Acid Production by
... the concentration. One reason for this variation is that the number of ions in the solution might not be proportional to the concentration of the electrolyte. For instance, the concentration of ions in a solution of a weak acid depends on the concentration of the acid in a complicated way, and doubl ...
... the concentration. One reason for this variation is that the number of ions in the solution might not be proportional to the concentration of the electrolyte. For instance, the concentration of ions in a solution of a weak acid depends on the concentration of the acid in a complicated way, and doubl ...
SODIUM HYDROGEN CARBONATE
... The CO2 can be recycled to produce more NaHCO3 by the Solvay process. In recent years, only about 15% of Na2CO3 was produced by the Solvay process. The remainder was mined from large deposits of trona, Na2CO3∙NaHCO3∙H2O, which were discovered in 1938 near the Green River in Wyoming. It is cheaper to ...
... The CO2 can be recycled to produce more NaHCO3 by the Solvay process. In recent years, only about 15% of Na2CO3 was produced by the Solvay process. The remainder was mined from large deposits of trona, Na2CO3∙NaHCO3∙H2O, which were discovered in 1938 near the Green River in Wyoming. It is cheaper to ...
Module 1 - Hartismere
... 3.2g of copper sulphate (CuSO4) ? 0.135g of water ? 25g of Calcium carbonate (CaCO3) ? ...
... 3.2g of copper sulphate (CuSO4) ? 0.135g of water ? 25g of Calcium carbonate (CaCO3) ? ...
Acid-Base Equilibria - Riverside Local Schools
... from the Latin word acidus, meaning sour or tart. Bases, in contrast, have a bitter taste and feel slippery (soap is a good example). The word base comes from an old English meaning of the word, which is "to bring low." (We still use the word debase in this sense, meaning to lower the value of somet ...
... from the Latin word acidus, meaning sour or tart. Bases, in contrast, have a bitter taste and feel slippery (soap is a good example). The word base comes from an old English meaning of the word, which is "to bring low." (We still use the word debase in this sense, meaning to lower the value of somet ...
Chapter 1 questions
... Nicotine is a highly toxic chemical which is strongly addictive when ingested, generally through the inhalation of burning tobacco. The relative molecular mass of nicotine is approximately 160. Quantitative analysis of this compound yields the following percentages by mass of its constituent element ...
... Nicotine is a highly toxic chemical which is strongly addictive when ingested, generally through the inhalation of burning tobacco. The relative molecular mass of nicotine is approximately 160. Quantitative analysis of this compound yields the following percentages by mass of its constituent element ...
aldehyde, ketones and carboxylic acids
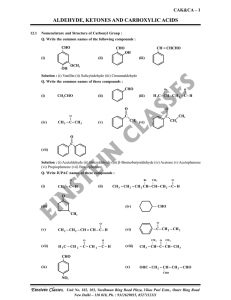
... intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehydes are more reactive than Ketones ? Solution : There are two reasons for this, they are as follows : 1. Steri ...
... intermediate captures a proton from the reaction medium to give the electrically neutral product. The net result is addition of Nu– and H+ across the carbon oxygen double bond. Q. Why Aldehydes are more reactive than Ketones ? Solution : There are two reasons for this, they are as follows : 1. Steri ...
Topic 2
... same atomic number (Z). The #protons defines the identity of an atom and can be found on the periodic table (large number in top of element box). – The neutron is a nuclear particle having a mass almost identical to that of a proton, but no electric charge. The charge of the nucleus comes from the # ...
... same atomic number (Z). The #protons defines the identity of an atom and can be found on the periodic table (large number in top of element box). – The neutron is a nuclear particle having a mass almost identical to that of a proton, but no electric charge. The charge of the nucleus comes from the # ...
Oxidation And Degradation Products Of Common Oxygen Scavengers
... When sodium hydroxide is present in the boiler water, the acetic acid produced is trapped as sodium acetate and removed by blowdown. The other products are volatile and pass through the condensate system and are vented. In autoclave studies, with very high concentrations of DEHA, very small quantiti ...
... When sodium hydroxide is present in the boiler water, the acetic acid produced is trapped as sodium acetate and removed by blowdown. The other products are volatile and pass through the condensate system and are vented. In autoclave studies, with very high concentrations of DEHA, very small quantiti ...