1 Assignment 5 Hydrogen – The Unique Element
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
1 Assignment 4 Hydrogen – The Unique Element
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
... Both molecular and saline hydrides are quite reactive. Group 1 and 2 hydrides react vigorously with water to produce hydrogen gas and a metal hydroxide. This means that they can be used as drying agents for solvents – the most commonly used in this regard is CaH2. p-Block molecular hydrides have dif ...
problem 8 on 2003B exam
... These materials were produced by Educational Testing Service® (ETS®), which develops and administers the examinations of the Advanced Placement Program for the College Board. The College Board and Educational Testing Service (ETS) are dedicated to the principle of equal opportunity, and their progra ...
... These materials were produced by Educational Testing Service® (ETS®), which develops and administers the examinations of the Advanced Placement Program for the College Board. The College Board and Educational Testing Service (ETS) are dedicated to the principle of equal opportunity, and their progra ...
Physical and Chemical equilibrium
... The reaction quotient is defined as the ratio of concentration of the reaction species at any point of time other than the equilibrium stage. It is represented by Q. Thus, inserting the starting concentrations of reactant and product in the equilibrium constant expression gives ...
... The reaction quotient is defined as the ratio of concentration of the reaction species at any point of time other than the equilibrium stage. It is represented by Q. Thus, inserting the starting concentrations of reactant and product in the equilibrium constant expression gives ...
B - eko.olunet.org
... Diversity of Coloured Compounds 1. Metal X forms three oxides: A, B and C. Compound B is green and it is used in oil colour production. The oxidation state of X in both hydroxide D and oxide B is the same. Compound E is obtained upon dissolving D in hydrochloric acid. When D is dissolved in potassiu ...
... Diversity of Coloured Compounds 1. Metal X forms three oxides: A, B and C. Compound B is green and it is used in oil colour production. The oxidation state of X in both hydroxide D and oxide B is the same. Compound E is obtained upon dissolving D in hydrochloric acid. When D is dissolved in potassiu ...
articles - Geoscience Research Institute
... the earliest rocks should contain an unusually large proportion of carbon or organic chemicals. This is not the case.9 Abelson9 and Cloud10 further state that the primitive atmosphere may have been an oxidizing environment. In other words, the elements of the primitive atmosphere had combined with o ...
... the earliest rocks should contain an unusually large proportion of carbon or organic chemicals. This is not the case.9 Abelson9 and Cloud10 further state that the primitive atmosphere may have been an oxidizing environment. In other words, the elements of the primitive atmosphere had combined with o ...
WHAT YOU EAT - Montana State University Extended University
... membranes that surround all cells. Fats are also key sources of energy for animals (recall that most of the cells in our bodies prefer to burn fat as fuel virtually all of the time). And, energy ...
... membranes that surround all cells. Fats are also key sources of energy for animals (recall that most of the cells in our bodies prefer to burn fat as fuel virtually all of the time). And, energy ...
chemical reactions
... K2CrO4 (aq) + Ba(NO3)2 (aq) BaCrO4 (s) + 2KNO3 (aq) Precipitate These reactions will be further discussed in Chapter 8 ...
... K2CrO4 (aq) + Ba(NO3)2 (aq) BaCrO4 (s) + 2KNO3 (aq) Precipitate These reactions will be further discussed in Chapter 8 ...
2016 Chemistry Examination Paper
... (ii) Describe how a bromine solution could be used to determine which of two colourless liquids is DHMA and which is oleic acid. _______________________________________________________________________________________________________ ___________________________________________________________________ ...
... (ii) Describe how a bromine solution could be used to determine which of two colourless liquids is DHMA and which is oleic acid. _______________________________________________________________________________________________________ ___________________________________________________________________ ...
Chemistry II Exams and Keys Corrected 2016 Season
... 1. Which of the following instrumental techniques is most suitable in determining the concentration of Cr6+ ions in aqueous solutions? A. Visible Spectroscopy B. X-Ray Crystallography ...
... 1. Which of the following instrumental techniques is most suitable in determining the concentration of Cr6+ ions in aqueous solutions? A. Visible Spectroscopy B. X-Ray Crystallography ...
rate law determination of crystal violet hydroxylation
... To find the reaction order of CV+, m, and the pseudo rate constants, k1 and k2, differential rate laws expressed in equations 3 & 4 must be integrated. (You should review integrated rate laws in your lecture text before continuing.) Integrated rate laws, when arranged in line equation form, result i ...
... To find the reaction order of CV+, m, and the pseudo rate constants, k1 and k2, differential rate laws expressed in equations 3 & 4 must be integrated. (You should review integrated rate laws in your lecture text before continuing.) Integrated rate laws, when arranged in line equation form, result i ...
Principles of Chemistry 1 and 2 Notes
... (turning it on/off) Generally speaking: Compounds that possess dipole moments are said to be polar and compounds that do not possess dipole moments are said to be non polar. Examples: Diatomic molecules: CO, NO, HCl, HF, etc…. all have dipole moments and hence are polar. Exceptions for the general r ...
... (turning it on/off) Generally speaking: Compounds that possess dipole moments are said to be polar and compounds that do not possess dipole moments are said to be non polar. Examples: Diatomic molecules: CO, NO, HCl, HF, etc…. all have dipole moments and hence are polar. Exceptions for the general r ...
Practice Test Material - Directorate of Education
... A welding gas contains carbon and hydrogen only. Burning a small sample of it in oxygen gives 3.38g carbon dioxide, 0.690g of water and no other products. A volume of 10.0L (measured at STP) of this welding gas is found to weigh 11.6g. Calculate : ...
... A welding gas contains carbon and hydrogen only. Burning a small sample of it in oxygen gives 3.38g carbon dioxide, 0.690g of water and no other products. A volume of 10.0L (measured at STP) of this welding gas is found to weigh 11.6g. Calculate : ...
Discussion 9, Mahaffy et al., Chapter 15
... a. Oxidation is loss of electrons (acts as a reducing agent) b.Reduction is gain of electrons (acts as a oxidizing agent) Assigning Oxidation numbers c. Oxidation number is 0 for atoms in an element. d.The sum of all oxidation numbers in a molecule or ion must add up to the total charge. e. In compo ...
... a. Oxidation is loss of electrons (acts as a reducing agent) b.Reduction is gain of electrons (acts as a oxidizing agent) Assigning Oxidation numbers c. Oxidation number is 0 for atoms in an element. d.The sum of all oxidation numbers in a molecule or ion must add up to the total charge. e. In compo ...
CHEM_01A_ExptD_Copper_Cycle_F14
... During this experiment, you will be asked to classify the reactions taking place into one of the categories described in the ‘Introduction’. When you classify each reaction, you might find it helpful to ...
... During this experiment, you will be asked to classify the reactions taking place into one of the categories described in the ‘Introduction’. When you classify each reaction, you might find it helpful to ...
xmas review questions 01516 with hints
... 39. When dilute nitric acid was added to a solution of one of the following chemicals, a gas was evolved. This gas turned a drop of limewater, Ca(OH)2, cloudy, due to the formation of a white precipitate. The chemical was (A) household ammonia, NH3 (B) baking soda, NaHCO3 (C) table salt, NaCl (D) ep ...
... 39. When dilute nitric acid was added to a solution of one of the following chemicals, a gas was evolved. This gas turned a drop of limewater, Ca(OH)2, cloudy, due to the formation of a white precipitate. The chemical was (A) household ammonia, NH3 (B) baking soda, NaHCO3 (C) table salt, NaCl (D) ep ...
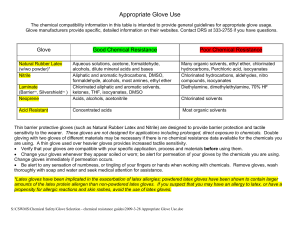
Appropriate Glove Use
... hydrocarbons, Perchloric acid, isocyanates Chlorinated hydrocarbons, aldehydes, nitro compounds, isocyanates Diethylamine, dimethylethylamine, 70% HF ...
... hydrocarbons, Perchloric acid, isocyanates Chlorinated hydrocarbons, aldehydes, nitro compounds, isocyanates Diethylamine, dimethylethylamine, 70% HF ...
chem equation Pkt Student2
... will require trial and error, the following guidelines may be helpful) a) balance the different types of atoms ________________ b) first, balance the atoms of elements that are combined and that appear only ___________on each side of the equation c) balance ____________________________that appear on ...
... will require trial and error, the following guidelines may be helpful) a) balance the different types of atoms ________________ b) first, balance the atoms of elements that are combined and that appear only ___________on each side of the equation c) balance ____________________________that appear on ...
Module 2 Alcohols, halogenoalkanes and analysis
... Halogenoalkanes are important starting materials for many synthetic routes. This is because they are readily converted into alcohols and other functional groups – you will become familiar with many of these compounds during your Advanced level work. For centuries, alcohols have been widely known and ...
... Halogenoalkanes are important starting materials for many synthetic routes. This is because they are readily converted into alcohols and other functional groups – you will become familiar with many of these compounds during your Advanced level work. For centuries, alcohols have been widely known and ...