Name - Chemistry 302
... 2. Break reaction into to half-reactions: oxidation half-reaction and reduction half-reaction 3. Balance all species EXCEPT HYDROGEN AND OXYGEN. 4. Balance OXYGEN by adding water to the necessary sides of the half-reactions. 5. Balance HYDROGEN by adding protons (H+) to the necessary sides 5a. If in ...
... 2. Break reaction into to half-reactions: oxidation half-reaction and reduction half-reaction 3. Balance all species EXCEPT HYDROGEN AND OXYGEN. 4. Balance OXYGEN by adding water to the necessary sides of the half-reactions. 5. Balance HYDROGEN by adding protons (H+) to the necessary sides 5a. If in ...
The Study of pH Influence on Bovine Liver Catalase by Means of UV
... located in molecule is correlation time Wc [7]. According to expectation the highest correlation time value is obtained when pH 7 (Wc = 3.51*10-10). However, the change of pH both toward acidic and alkaline environment causes decrease of correlation time value (Wc = 1.21*10-10 for pH 2, Wc = 1.84*10 ...
... located in molecule is correlation time Wc [7]. According to expectation the highest correlation time value is obtained when pH 7 (Wc = 3.51*10-10). However, the change of pH both toward acidic and alkaline environment causes decrease of correlation time value (Wc = 1.21*10-10 for pH 2, Wc = 1.84*10 ...
Lithium chloride ionic association in dilute aqueous solution: a
... where kB is the Boltzmann constant, T is the temperature of the system. However, in the dilute aqueous systems, a simple molecular dynamics simulation would always result in a problematic ionic radial distribution function for the sake of inadequate sampling or some traps during the propagation of t ...
... where kB is the Boltzmann constant, T is the temperature of the system. However, in the dilute aqueous systems, a simple molecular dynamics simulation would always result in a problematic ionic radial distribution function for the sake of inadequate sampling or some traps during the propagation of t ...
Bulgarian Chemical Communications, Volume 41, Number 4 (pp
... The degradation of diazo dyes Brilliant Yellow (BY) and Bismark Brown (BB) was investigated by the photoFenton-like process Fe2+/ammonium persulphate (APS)/UV in acidic pH medium. The influence of various reaction parameters like pH, concentration of Fe2+ ions/APS, structure of the dye and effect of ...
... The degradation of diazo dyes Brilliant Yellow (BY) and Bismark Brown (BB) was investigated by the photoFenton-like process Fe2+/ammonium persulphate (APS)/UV in acidic pH medium. The influence of various reaction parameters like pH, concentration of Fe2+ ions/APS, structure of the dye and effect of ...
Post Lab Questions
... Your assignments are a reflection of you, your commitment to quality and your interest in the class. All assignments will be turned in on flat, smooth paper with no tears. Notebook paper will not have spiral notebook fuzz. All assignments are to be done in ink, blue or black only. Assignments should ...
... Your assignments are a reflection of you, your commitment to quality and your interest in the class. All assignments will be turned in on flat, smooth paper with no tears. Notebook paper will not have spiral notebook fuzz. All assignments are to be done in ink, blue or black only. Assignments should ...
STEREOCHEMISTRY - M E S KVM College Valanchery.
... 2. Differential Absorption. When a racemic mixture is placed on a chromatographic column and if the column consists of chiral substances, then the enantiomers should move along the column at different rates. Separation has been successfully accomplished with paper, column, thin-layer and gas and liq ...
... 2. Differential Absorption. When a racemic mixture is placed on a chromatographic column and if the column consists of chiral substances, then the enantiomers should move along the column at different rates. Separation has been successfully accomplished with paper, column, thin-layer and gas and liq ...
Solutions
... **Like dissolves like** Substances with similar intermolecular attractive forces tend to be soluble in one another. -Polar liquids tend to dissolve in polar solvents. -Hydrogen bonding interaction between solute and solvent may lead to high solubility. (In alcohols as the length of the carbon chain ...
... **Like dissolves like** Substances with similar intermolecular attractive forces tend to be soluble in one another. -Polar liquids tend to dissolve in polar solvents. -Hydrogen bonding interaction between solute and solvent may lead to high solubility. (In alcohols as the length of the carbon chain ...
Table of Contents
... ___________________ that traps the solid particles while the liquid passes through in a process called filtering. Some simple methods also exist for separating homogeneous mixtures. A solid dissolved in a liquid solution can be separated by letting it dry out in the process of ___________________. M ...
... ___________________ that traps the solid particles while the liquid passes through in a process called filtering. Some simple methods also exist for separating homogeneous mixtures. A solid dissolved in a liquid solution can be separated by letting it dry out in the process of ___________________. M ...
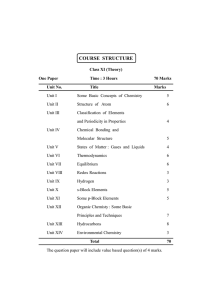
COURSE STRUCTURE
... (c) Calculate the volume of 0.1 M NaOH solution is required to neutralise 100 mL of concentrated aqueous sulphuric acid which contains 98% H2SO4 by mass. The density of conc. H2SO4 is 1.84 g/mL. NaOH reacts with H2SO4 according to the following equation : 2NaOH + H2SO4 Na2SO4 + 2H2O (At. mass/g mo ...
... (c) Calculate the volume of 0.1 M NaOH solution is required to neutralise 100 mL of concentrated aqueous sulphuric acid which contains 98% H2SO4 by mass. The density of conc. H2SO4 is 1.84 g/mL. NaOH reacts with H2SO4 according to the following equation : 2NaOH + H2SO4 Na2SO4 + 2H2O (At. mass/g mo ...
Significant Figures
... ferrous chloride or iron(II) chloride ferric chloride or iron(III) chloride ...
... ferrous chloride or iron(II) chloride ferric chloride or iron(III) chloride ...
An Introduction to Chemical Science
... observations to be made, etc. The right-hand page is blank, and on that the pupil makes a record of his work. These notes are examined at the time, or subsequently, by the teacher, and the pupil is not allowed to take the book from the laboratory; nor can he use any other book on Chemistry while exp ...
... observations to be made, etc. The right-hand page is blank, and on that the pupil makes a record of his work. These notes are examined at the time, or subsequently, by the teacher, and the pupil is not allowed to take the book from the laboratory; nor can he use any other book on Chemistry while exp ...
study material(2014-15) class xii-chemistry
... Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials prepared by each participant were thoroughly reviewed by their co-particip ...
... Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials prepared by each participant were thoroughly reviewed by their co-particip ...
CHAPTER 3 STOICHIOMETRY:
... Yes; because there are equal numbers of both N and O atoms in the two boxes ...
... Yes; because there are equal numbers of both N and O atoms in the two boxes ...
chemistry-resource
... Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials prepared by each participant were thoroughly reviewed by their co-particip ...
... Reviewed Support Materials of the previous year. In order to ensure that the participants come well-prepared for the Workshop, the topics/chapters were distributed among them well in advance. During the Workshop the materials prepared by each participant were thoroughly reviewed by their co-particip ...
tro2_ppt_lecture_04 - Louisiana Tech University
... Limiting Reactant • For reactions with multiple reactants, it is likely that one of the reactants will be completely used before the others. • When this reactant is used up, the reaction stops and no more product is made. • The reactant that limits the amount of product is called the limiting react ...
... Limiting Reactant • For reactions with multiple reactants, it is likely that one of the reactants will be completely used before the others. • When this reactant is used up, the reaction stops and no more product is made. • The reactant that limits the amount of product is called the limiting react ...
Chem 33 Lab - Santa Clara University
... of experiments. Your lab grade will reflect the caliber of experiments you have chosen. Avoid doing all the easy ones or all the hard ones. Remember that you will usually have to be working on more than one experiment at any one time. You must carefully analyze the procedures of the experiments you ...
... of experiments. Your lab grade will reflect the caliber of experiments you have chosen. Avoid doing all the easy ones or all the hard ones. Remember that you will usually have to be working on more than one experiment at any one time. You must carefully analyze the procedures of the experiments you ...
chemistry
... detail would be almost impossible, and a waste of time. Chemists classify matter by its behaviour and properties, and in doing so, simplify the process of understanding and prediction. If you read a description of matter which indicates that it is a solid, nonmetallic molecular compound, then (by th ...
... detail would be almost impossible, and a waste of time. Chemists classify matter by its behaviour and properties, and in doing so, simplify the process of understanding and prediction. If you read a description of matter which indicates that it is a solid, nonmetallic molecular compound, then (by th ...