Solubility Rules The following rules are used in several chemistry
... Sulfide Sulfite Silicate Silver ...
... Sulfide Sulfite Silicate Silver ...
Atoms, Molecules and Ions - Moodle @ FCT-UNL
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
Chapter 2 PowerPoint
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
... (b) There are two silicon atoms and six bromine atoms present, so the formula is Si2Br6. ...
Chapter 20 – The Representative Elements
... strong acid and a very powerful oxidizing agent. It oxidizes almost all metals it comes in contact with, except gold and platinum. Unlike the reactions of metals with other strong acid (such as HCl and H 2SO4), the reactions of nitric acid with metals do not produce H2 gas. This is because the nitra ...
... strong acid and a very powerful oxidizing agent. It oxidizes almost all metals it comes in contact with, except gold and platinum. Unlike the reactions of metals with other strong acid (such as HCl and H 2SO4), the reactions of nitric acid with metals do not produce H2 gas. This is because the nitra ...
WELCOME TO AP CHEMISTRY
... not know how to do a problem, ask a friend to explain it to you. You will be doing him/her a favor because the ability to explain a concept to someone else is a measure of that person’s true understanding. Use the text book as your primary resource. Please memorize nomenclature rules for ionic compo ...
... not know how to do a problem, ask a friend to explain it to you. You will be doing him/her a favor because the ability to explain a concept to someone else is a measure of that person’s true understanding. Use the text book as your primary resource. Please memorize nomenclature rules for ionic compo ...
Name AP Chemistry Take Home Quiz – Due Thursday, 1/9/2014
... a. CN-(aq) is a stronger base than C2H3O2-(aq) b. HCN(aq) is a stronger acid than HC2H3O2(aq) c. The conjugate base of CN-(aq) is C2H3O2-(aq) d. The equilibrium constant will increase with an increase in temperature. e. The pH of a solution containing equimolar amounts of CN-(aq) and HC2H3O2(aq) is ...
... a. CN-(aq) is a stronger base than C2H3O2-(aq) b. HCN(aq) is a stronger acid than HC2H3O2(aq) c. The conjugate base of CN-(aq) is C2H3O2-(aq) d. The equilibrium constant will increase with an increase in temperature. e. The pH of a solution containing equimolar amounts of CN-(aq) and HC2H3O2(aq) is ...
Compounds Power point
... If the positive ion is a transition metal, the name of the compound MUST include ...
... If the positive ion is a transition metal, the name of the compound MUST include ...
Reaction rate and activation energy of the acidolysis
... energy activates the molecules (loosens bonds, polarisation etc.) so that they can react. The portion of molecules with this increased energy content increases with increasing temperature. The greater the portion of the molecules capable of reaction, the more molecules that will react, and so the hi ...
... energy activates the molecules (loosens bonds, polarisation etc.) so that they can react. The portion of molecules with this increased energy content increases with increasing temperature. The greater the portion of the molecules capable of reaction, the more molecules that will react, and so the hi ...
Test #1 Study Guide
... Molecular compounds are made up of two nonmetals and are easily distinguished from ionic compounds because of this. Naming of molecular ions requires you to know the prefixes that indicate the number of a certain thing. o For the first element, indicate the number that exist in the molecule with a p ...
... Molecular compounds are made up of two nonmetals and are easily distinguished from ionic compounds because of this. Naming of molecular ions requires you to know the prefixes that indicate the number of a certain thing. o For the first element, indicate the number that exist in the molecule with a p ...
432 Final Exam Study Guide
... aqueous Pb(NO3)2. You would expect one of the products of this reaction to be: a. K2S b. NaPb c. PbSO4 d. CNO3 ____2. A chemical formula written above or below the yield sign indicates: a. that a gas is formed. b. That the substance is a catalyst. c. That heat must be supplied. d. A reversible react ...
... aqueous Pb(NO3)2. You would expect one of the products of this reaction to be: a. K2S b. NaPb c. PbSO4 d. CNO3 ____2. A chemical formula written above or below the yield sign indicates: a. that a gas is formed. b. That the substance is a catalyst. c. That heat must be supplied. d. A reversible react ...
Chem 1A Final Exam – Fall 2005
... b) What is the vapor pressure at 100oC of a 2.0 molal aqueous solution of glucose, C6H12O6 ? Hint: Determine the mole fraction of glucose and use Raoult’s Law. (You don’t need to know the molar mass of glucose to solve this problem.) ...
... b) What is the vapor pressure at 100oC of a 2.0 molal aqueous solution of glucose, C6H12O6 ? Hint: Determine the mole fraction of glucose and use Raoult’s Law. (You don’t need to know the molar mass of glucose to solve this problem.) ...
Document
... Neutral: pH 7.00 Neutral solutions have equal concentrations of H+ and OH– 45. Acid-Base Concentration (pH) http://staff.jccc.net/PDECELL/chemistry/phscal e.gif 46. Neutralization Mixing acids and bases leads to displacement reactions that restore pH to neutral Acid + Base Water + Salt HCl + NaOH H2 ...
... Neutral: pH 7.00 Neutral solutions have equal concentrations of H+ and OH– 45. Acid-Base Concentration (pH) http://staff.jccc.net/PDECELL/chemistry/phscal e.gif 46. Neutralization Mixing acids and bases leads to displacement reactions that restore pH to neutral Acid + Base Water + Salt HCl + NaOH H2 ...
Questions 1-2
... Calculate the number of moles of NaOBr(s) that would have to be added to 125 mL of 0.160 M HOBr to produce a buffer solution with [H+] = 5.00 x 10-9 M. Assume that volume change is negligible. ...
... Calculate the number of moles of NaOBr(s) that would have to be added to 125 mL of 0.160 M HOBr to produce a buffer solution with [H+] = 5.00 x 10-9 M. Assume that volume change is negligible. ...
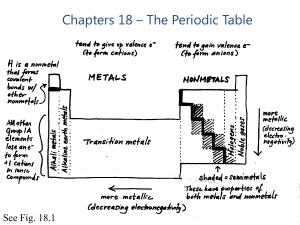
Chapters 18 – The Periodic Table
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
... element behaves like a true metal Common behavior: reaction with metal to become 2- ion in ionic compound; for most metals, most common minerals are oxides or sulfides Covalent bonds with other NMs; series of covalent hydrides (H2X) All but O have d orbitals available, so more than an octet is ...
chemistry important question i
... (C) Arrange the following compounds in increasing order of acid strength: Benzoic acid, 4-Nitrobenzoic acid, 4-Methoxybenzoic acid. 34. (a) Write the mechanism of hydration of ethene to form ethanol. (b) How are the following conversions carried out? (i) Propanol to propan-2-ol. (ii) Propanol to 1-p ...
... (C) Arrange the following compounds in increasing order of acid strength: Benzoic acid, 4-Nitrobenzoic acid, 4-Methoxybenzoic acid. 34. (a) Write the mechanism of hydration of ethene to form ethanol. (b) How are the following conversions carried out? (i) Propanol to propan-2-ol. (ii) Propanol to 1-p ...