Operate some of the instruments and perform analysis
... 1. Determination of pK value of an acid-base Indicator (Methyl red) using Colorimeter. 2. Simultaneous estimation of Mn2+ and Cr3+ in a solution of KMnO4 and K2Cr2O7. 3. Determination of concentration of ferrous ions in a given sample spectrophotometrically by Ophenanthroline method and by using KCN ...
... 1. Determination of pK value of an acid-base Indicator (Methyl red) using Colorimeter. 2. Simultaneous estimation of Mn2+ and Cr3+ in a solution of KMnO4 and K2Cr2O7. 3. Determination of concentration of ferrous ions in a given sample spectrophotometrically by Ophenanthroline method and by using KCN ...
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
... 1. OH- group consisting of carboxylic acid functional groups; 2. hydroxyl group with sp 3-hybrid oxygen; 3. N-H acidic center; 4. OH group bonded directly to the heterocycle; 39. The most strong acidic properties of the compound shown in: 1. acetic acid; 2. propanoic acid; 3. 2-methylpropanoic acid; ...
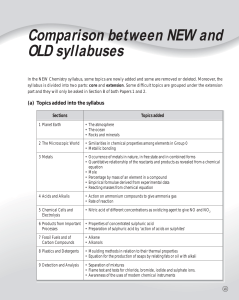
Molecular Formulas - Hatboro
... outside of the classroom. The ultimate objective, of course, is to prepare you to take the AP Chemistry test on May 4, 2015, and in order to accomplish this, topics are covered very quickly. For this reason, most students take AP Chemistry after they already completed a year of high school chemistry ...
... outside of the classroom. The ultimate objective, of course, is to prepare you to take the AP Chemistry test on May 4, 2015, and in order to accomplish this, topics are covered very quickly. For this reason, most students take AP Chemistry after they already completed a year of high school chemistry ...
Lesson 1 - Working With Chemicals
... NH4+, which acts as a metal in compounds. - The name of the cation (metal) is followed by the name of the anion (non-metal – negatively ...
... NH4+, which acts as a metal in compounds. - The name of the cation (metal) is followed by the name of the anion (non-metal – negatively ...
IGCSE Revision Guide (Double Award) | PDF
... Recognise and explain OXIDATION as loss of electrons and REDUCTION as gain of electrons: • Oil RiG Recall the charge on common ions – both metals and non-‐metals – and compound ions e.g. SO42-‐, C ...
... Recognise and explain OXIDATION as loss of electrons and REDUCTION as gain of electrons: • Oil RiG Recall the charge on common ions – both metals and non-‐metals – and compound ions e.g. SO42-‐, C ...
01.CN_Other pages/p1-9
... (b) Suggest a method by which the black solid B can be converted to a metal in the laboratory. Write an equation for the reaction ...
... (b) Suggest a method by which the black solid B can be converted to a metal in the laboratory. Write an equation for the reaction ...
1. A glucose molecule contains six carbons, twelve hydrogens and
... A sample of HCl(aq) has a volume of 25.00 mL and a concentration of 3.50 M. What is the HCl concentration if the sample is diluted to a volume of 75.00 mL? A. 1.41 M C. 1.05 M B. ...
... A sample of HCl(aq) has a volume of 25.00 mL and a concentration of 3.50 M. What is the HCl concentration if the sample is diluted to a volume of 75.00 mL? A. 1.41 M C. 1.05 M B. ...
Summer Assignment for AP Chemistry: I hope you are all ready for a
... The summer assignment is due the first day of school in August. And there will be a test on this material within the first two weeks of school. We will begin the year with a brief project that will very quickly review the material listed above, and then test on that material. However, you will want ...
... The summer assignment is due the first day of school in August. And there will be a test on this material within the first two weeks of school. We will begin the year with a brief project that will very quickly review the material listed above, and then test on that material. However, you will want ...
CfE Higher Chemistry Homework 3.5
... Write a balanced chemical equation for the production of hydrogen sulfide from aluminium sulfide and water. ...
... Write a balanced chemical equation for the production of hydrogen sulfide from aluminium sulfide and water. ...
Chapter 4 Solution Chemistry
... used to determine the concentration of ions in solution. For example, Ag+ can be added to a solution of Cl- to precipitate all of the Cl- in the form of insoluble AgCl. The concentration of the Cl- is determined by weighing the AgCl and using the stoichiometry of the reaction. To determine the conce ...
... used to determine the concentration of ions in solution. For example, Ag+ can be added to a solution of Cl- to precipitate all of the Cl- in the form of insoluble AgCl. The concentration of the Cl- is determined by weighing the AgCl and using the stoichiometry of the reaction. To determine the conce ...
JF Physical Chemistry 2010-2011. JF CH 1101: Introduction to
... which relates the change in internal energy of a system to the work done on the system and the heat absorbed by the system. Hence derive a relationship between the change in internal energy ΔU and the change in enthalpy ΔH of a system. b. A gas absorbs 300 J of heat and at the same time expands by 1 ...
... which relates the change in internal energy of a system to the work done on the system and the heat absorbed by the system. Hence derive a relationship between the change in internal energy ΔU and the change in enthalpy ΔH of a system. b. A gas absorbs 300 J of heat and at the same time expands by 1 ...
AP Chem Summer Assignment KEY
... 3. sodium sulfide + manganese (VI) acetate 3Na2S + Mn(CH3COO)6 6NaCH3COO + MnS3 4. chromium(III) bromide + sodium sulfite 2 CrBr3 + 3 Na2SO3 ...
... 3. sodium sulfide + manganese (VI) acetate 3Na2S + Mn(CH3COO)6 6NaCH3COO + MnS3 4. chromium(III) bromide + sodium sulfite 2 CrBr3 + 3 Na2SO3 ...
Acid-Base Reactions Worksheet #2 - Mro
... a spring and electrically ignite a detonator cap. This causes sodium azide, NaN3, to decompose explosively according to the following unbalanced equation: NaN3(s) ...
... a spring and electrically ignite a detonator cap. This causes sodium azide, NaN3, to decompose explosively according to the following unbalanced equation: NaN3(s) ...
lecture10
... however, it is very ill-behaved indeed. These are ions. Think about what happens when you dissolve salt (NaCl) in water. The sodium and the chloride ions separate from one another giving Na+ and Cl-. The reason this reaction goes forward is because it results in an increase in entropy as the salt cr ...
... however, it is very ill-behaved indeed. These are ions. Think about what happens when you dissolve salt (NaCl) in water. The sodium and the chloride ions separate from one another giving Na+ and Cl-. The reason this reaction goes forward is because it results in an increase in entropy as the salt cr ...
Summer Assignment Ch. 2-5
... Concept 3.3 Acidic and basic conditions affect living organisms 23. What two ions form when water dissociates? ...
... Concept 3.3 Acidic and basic conditions affect living organisms 23. What two ions form when water dissociates? ...
29.2 Chemical Bonds
... In chemical reactions you start with reactants that are combined into products. The reactants and products may include atoms, molecules, and energy. ...
... In chemical reactions you start with reactants that are combined into products. The reactants and products may include atoms, molecules, and energy. ...
voltammetric studies of vitamin k3 in acid aqueous solution
... because the second electron enters immediately after the protonation of the radical anion formed by the transfer of the first electron from the mercury cathode to the substrate. Since protonation is a fast reaction in very acidic medium, i.e., very low pH values, for the voltage scan rates used the ...
... because the second electron enters immediately after the protonation of the radical anion formed by the transfer of the first electron from the mercury cathode to the substrate. Since protonation is a fast reaction in very acidic medium, i.e., very low pH values, for the voltage scan rates used the ...
Single-Replacement Reactions
... Count the number of atoms of each type of element appearing on both sides Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
... Count the number of atoms of each type of element appearing on both sides Balance the atoms of an element one at a time by adding coefficients (the numbers in front) - save H and O until LAST! Check to make sure it is balanced. ...
Chemistry 1B General Chemistry Laboratory
... units when showing an example calculation. Since many calculations are repetitive, you can continue to work them out on a separate piece of white paper to include with your report. Observations: These are a crucial part of your scientific endeavors. Depending on the experiment, some observations can ...
... units when showing an example calculation. Since many calculations are repetitive, you can continue to work them out on a separate piece of white paper to include with your report. Observations: These are a crucial part of your scientific endeavors. Depending on the experiment, some observations can ...