chapter 5 gases
... Strategy: In order to break a redox reaction down into an oxidation half-reaction and a reduction halfreaction, you should first assign oxidation numbers to all the atoms in the reaction. In this way, you can determine which element is oxidized (loses electrons) and which element is reduced (gains e ...
... Strategy: In order to break a redox reaction down into an oxidation half-reaction and a reduction halfreaction, you should first assign oxidation numbers to all the atoms in the reaction. In this way, you can determine which element is oxidized (loses electrons) and which element is reduced (gains e ...
2014 Syllabus - Cambridge International Examinations
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Advanced Practical Skills 1 and Advanced Practical Skills 2. In other sessions only Advanced Practical Skills 1 will be available. These papers will be equivalent and each candidate wil ...
sol-gel chemistry of transition metal oxides
... author proposed a mechanism of hydrolysis in which hydroxyl groups are added to the which leads to the formation of condensed species. ...
... author proposed a mechanism of hydrolysis in which hydroxyl groups are added to the which leads to the formation of condensed species. ...
Chapter 2 1.Certain gases in the 293K and 9.97 × 104Pa when the
... 1. Try to use ionic bond theory to show a single mass of metal potassium and chlorine reaction, the formation of potassium chloride process? How to understand the ionic bond non-directional and saturation? Answer: Atoms derived from the two because of electrostatic attraction and the attraction betw ...
... 1. Try to use ionic bond theory to show a single mass of metal potassium and chlorine reaction, the formation of potassium chloride process? How to understand the ionic bond non-directional and saturation? Answer: Atoms derived from the two because of electrostatic attraction and the attraction betw ...
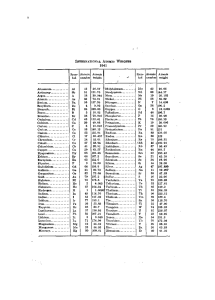
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... Mr. Milton Goff, Mr. A. E. Edwards, Mr. Lee K. Henke, and many students and others. They are indebted to Mr. Henry H. Schmalz, a studSnt in the course, for the helpful new drawings. ...
... Mr. Milton Goff, Mr. A. E. Edwards, Mr. Lee K. Henke, and many students and others. They are indebted to Mr. Henry H. Schmalz, a studSnt in the course, for the helpful new drawings. ...
File - IB CHEM NINJA
... of the equilibrium constant, Kc, because the values of the forward and reverse rate constants kf and kr respectively do not change. A change in temperature does however affect the rate constants, so that the value of Kc changes as well as the position of equilibrium. If an unreactive gas is added to ...
... of the equilibrium constant, Kc, because the values of the forward and reverse rate constants kf and kr respectively do not change. A change in temperature does however affect the rate constants, so that the value of Kc changes as well as the position of equilibrium. If an unreactive gas is added to ...
Experiments in General Chemistry: Featuring MeasureNet
... Chemists observe matter by determining, measuring, and monitoring physical and chemical properties of matter. A property is any characteristic that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and so ...
... Chemists observe matter by determining, measuring, and monitoring physical and chemical properties of matter. A property is any characteristic that can be used to describe matter (e.g., size, color, mass, density, solubility, etc.). In this experiment, we will determine the density of liquids and so ...
Reaction Kinetics Basics
... collide with a third molecular entity (e.g. a molecule or radical). In a unimolecular reaction, only one reaction partner species is changed. Examples include photochemical reactions (e.g. NO2 + hν ! NO + O, where hν represents a photon) and unimolecular decomposition such as the decomposition of fu ...
... collide with a third molecular entity (e.g. a molecule or radical). In a unimolecular reaction, only one reaction partner species is changed. Examples include photochemical reactions (e.g. NO2 + hν ! NO + O, where hν represents a photon) and unimolecular decomposition such as the decomposition of fu ...
The Free High School Science Texts
... Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation; with no Invariant Sections, no FrontCover Texts, and no Back-Cover Texts. A copy of the license is ...
... Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation; with no Invariant Sections, no FrontCover Texts, and no Back-Cover Texts. A copy of the license is ...
Signs of Reaction - Calderglen High School
... The atoms of a compound are joined together. The substances in a mixture are NOT joined and can be easily separated. Calderglen High School ...
... The atoms of a compound are joined together. The substances in a mixture are NOT joined and can be easily separated. Calderglen High School ...
Chemistry 30 June 2001 Grade 12 Diploma Examination
... (Record your three-digit answer in the numerical-response section on the answer sheet.) *You can receive marks for this question even if the previous question was answered incorrectly. ...
... (Record your three-digit answer in the numerical-response section on the answer sheet.) *You can receive marks for this question even if the previous question was answered incorrectly. ...
CHOICE BASED CREDIT SYSTEM B. Sc. WITH CHEMISTRY
... ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hydrolysis-calculation of hydrolysis constant, degree of hydrolysis and pH for different salts. Buffer solutions. Solubility and solubility product of sparingly soluble s ...
... ionization, ionization constant and ionic product of water. Ionization of weak acids and bases, pH scale, common ion effect. Salt hydrolysis-calculation of hydrolysis constant, degree of hydrolysis and pH for different salts. Buffer solutions. Solubility and solubility product of sparingly soluble s ...
Concept based notes Chemistry Lab Manual
... Q. 73. Why, H2S gas passed in presence of NH4OH? Ans. When H2S gas is passed in alkaline medium or in presence of NH4OH, the H+ ions from the dissociation of H2S gas combine with hydroxyl ions (OH-) from the dissociation of NH4OH to from nearly unionised H2O. The removal of H+ ions from the solutio ...
... Q. 73. Why, H2S gas passed in presence of NH4OH? Ans. When H2S gas is passed in alkaline medium or in presence of NH4OH, the H+ ions from the dissociation of H2S gas combine with hydroxyl ions (OH-) from the dissociation of NH4OH to from nearly unionised H2O. The removal of H+ ions from the solutio ...
MAJOR - Bijni College
... (g) Solubility of salts and molecules in water. (h) Relative acid –base strength of Lewis and protonic acids with reference to oxides, hydroxides and oxoacids. (i) Electrode potentials and redox behaviour in aqueous solutions. Unit 4.2 Chemistry of Non-transition Elements I ...
... (g) Solubility of salts and molecules in water. (h) Relative acid –base strength of Lewis and protonic acids with reference to oxides, hydroxides and oxoacids. (i) Electrode potentials and redox behaviour in aqueous solutions. Unit 4.2 Chemistry of Non-transition Elements I ...
Influence of Temperature on Electrical
... In a water-steam cycle the pH is set at an alkaline range from 8.5 to 10, by adding an alkalizing agent. This agent is usually ammonia or an amine, e.g., morpholine or ethanolamine. All these substances are weak bases which only partly dissociate into ions. In comparison to the other electrolytes, t ...
... In a water-steam cycle the pH is set at an alkaline range from 8.5 to 10, by adding an alkalizing agent. This agent is usually ammonia or an amine, e.g., morpholine or ethanolamine. All these substances are weak bases which only partly dissociate into ions. In comparison to the other electrolytes, t ...
National German Competition
... Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. Following this path you can get -hydroxy acids and then in follo-up reactions new unsaturated and saturated carboxylic acids. Adding zin ...
... Zinc organic compounds are longer known and more often used. These compounds are applied to synthesize alcohols, more exactly in the synthesis of hydroxy esters. Following this path you can get -hydroxy acids and then in follo-up reactions new unsaturated and saturated carboxylic acids. Adding zin ...
A comparison of the effects of fluoride and chloride
... alcohol dehydrogenase can catalyze either oxidation or reduction depending upon the conditions. ...
... alcohol dehydrogenase can catalyze either oxidation or reduction depending upon the conditions. ...