Chemistry.of Organic Compounds
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
... in this text considerable emphasis is placed on the explanation of physical properties and on the mechanism of organic reactions. Where such material is given, an effort has been made to keep the discussion as simple as possible even at the risk of being quantitatively inaccurate, for "except ye utt ...
Under Choice Based Credit System Proposed syllabus and Scheme of Examination
... Chemical Bonding and Molecular Structure Ionic Bonding: General characteristics of ionic bonding. Energy considerations in ionic bonding, lattice energy and solvation energy and their importance in the context of stability and solubility of ionic compounds. Statement of Born-Landé equation for calcu ...
... Chemical Bonding and Molecular Structure Ionic Bonding: General characteristics of ionic bonding. Energy considerations in ionic bonding, lattice energy and solvation energy and their importance in the context of stability and solubility of ionic compounds. Statement of Born-Landé equation for calcu ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... These activities are intended to enable candidates to develop the skills, knowledge and understanding of How Science Works. Other aspects of the skills, knowledge and understanding of How Science Works will be better developed through investigative work and it is expected that teachers will adopt a ...
... These activities are intended to enable candidates to develop the skills, knowledge and understanding of How Science Works. Other aspects of the skills, knowledge and understanding of How Science Works will be better developed through investigative work and it is expected that teachers will adopt a ...
quantitative_chemistry
... This quantity is approximately 300 billion times more than the current human population of the earth! The comparison serves as a reminder of just how tiny molecules must be if that many are required to make up half a gram. Knowing that the average molecular mass of aspirin is 180.2 amu, a chemist at ...
... This quantity is approximately 300 billion times more than the current human population of the earth! The comparison serves as a reminder of just how tiny molecules must be if that many are required to make up half a gram. Knowing that the average molecular mass of aspirin is 180.2 amu, a chemist at ...
Syllabus Cambridge International A & AS Level Chemistry Syllabus code 9701
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Paper 31 and Paper 32. In other sessions only Paper 31 will be available. Paper 31 and Paper 32 will be equivalent and each candidate will be required to take only one of them. This is ...
... In some examination sessions, two versions of the Advanced Practical Skills paper will be available, identified as Paper 31 and Paper 32. In other sessions only Paper 31 will be available. Paper 31 and Paper 32 will be equivalent and each candidate will be required to take only one of them. This is ...
BSC HS CHEMISTRY SEMESTER I to VI CBCEGS
... entropy, evaluation of absolute entropy from heat capacity data. Gibbs and Helmholtz functions; Gibbs function (G) and Helmholtz function (A) as thermodynamic quantities. A & G as criteria for thermodynamic equilibrium and spontaneity, their advantage over entropy change. Variation of G and A with P ...
... entropy, evaluation of absolute entropy from heat capacity data. Gibbs and Helmholtz functions; Gibbs function (G) and Helmholtz function (A) as thermodynamic quantities. A & G as criteria for thermodynamic equilibrium and spontaneity, their advantage over entropy change. Variation of G and A with P ...
Test bank questions
... a. If hydrogen gas is added to the above system at equilibrium, which direction will the reaction shift? b. If nitrogen is added to the system at equilibrium, what will happen to the ammonia concentration? c. If nitrogen is removed from the system at equilibrium, what will happen to the hydrogen (H2 ...
... a. If hydrogen gas is added to the above system at equilibrium, which direction will the reaction shift? b. If nitrogen is added to the system at equilibrium, what will happen to the ammonia concentration? c. If nitrogen is removed from the system at equilibrium, what will happen to the hydrogen (H2 ...
Activation of Alcohols Toward Nucleophilic Substitution: Conversion
... alcohols are converted to saturated alkyl halides.6 Because the use of HCl shows poor results for the conversion of an alcohol to an alkyl chloride, a catalyst such as the zinc used in the Lucas reagent is required. This reaction was improved by adding zinc chloride and had the advantage of milder c ...
... alcohols are converted to saturated alkyl halides.6 Because the use of HCl shows poor results for the conversion of an alcohol to an alkyl chloride, a catalyst such as the zinc used in the Lucas reagent is required. This reaction was improved by adding zinc chloride and had the advantage of milder c ...
20. Chemical Equilibrium
... are converted to products before a reaction stops. In truth, however, experiments show that the conversion of reactants into products is often incomplete in chemical reactions. This is the case no matter how long the reaction is allowed to continue. As a reaction progresses, the concentrations of th ...
... are converted to products before a reaction stops. In truth, however, experiments show that the conversion of reactants into products is often incomplete in chemical reactions. This is the case no matter how long the reaction is allowed to continue. As a reaction progresses, the concentrations of th ...
document
... We can do the same sort of thing with the mass of the elements and compounds Let’s use same chemical equation C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Instead of moles we will use mass which is more realistic If we have 44.1 g of propane, how much oxygen will it react with? ...
... We can do the same sort of thing with the mass of the elements and compounds Let’s use same chemical equation C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Instead of moles we will use mass which is more realistic If we have 44.1 g of propane, how much oxygen will it react with? ...
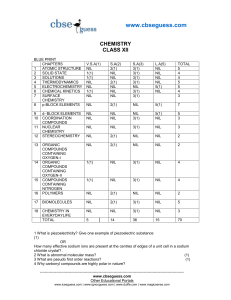
guess paper class xii
... 14 Analysis shows that nickel oxide has formula Ni0.98O1.00. What fractions of the nickel exist as Ni+2 and Ni+3 ions? ...
... 14 Analysis shows that nickel oxide has formula Ni0.98O1.00. What fractions of the nickel exist as Ni+2 and Ni+3 ions? ...
Tuning the Molecular Properties of Polybenzimidazole by
... groups which are far apart in the benzene ring because acid groups are probably unable to form the internal dimmers through hydrogen bonding interaction between carboxylic groups of the same acids or coming from two different molecules of the acid.35,36 Thus TPA (1,4-) has lower solubility than IPA ...
... groups which are far apart in the benzene ring because acid groups are probably unable to form the internal dimmers through hydrogen bonding interaction between carboxylic groups of the same acids or coming from two different molecules of the acid.35,36 Thus TPA (1,4-) has lower solubility than IPA ...
Coagulation Kinetics of Humic Aggregates in Mono- and Di
... groups, that is, carboxyl and phenolic −OH, have been measured and reported.41,42 From the titration data, the Donnan shell model with two discrete sites was applied to simulate our results.29 The pK values of carboxylic and phenolic −OH groups were estimated to be 5.08 and 7.96. The site densities ...
... groups, that is, carboxyl and phenolic −OH, have been measured and reported.41,42 From the titration data, the Donnan shell model with two discrete sites was applied to simulate our results.29 The pK values of carboxylic and phenolic −OH groups were estimated to be 5.08 and 7.96. The site densities ...
BS Chemistry - Government College University Faisalabad
... Nature and types of chemical bonding, lewis concepts, ionic, covalent, coordinate covalent bond; Valence bond theory (VBT), Molecular orbital theory (MOT). Interpretation of shapes of inorganic molecules on the basis of valence shell electron pair repulsion (VSEPR) theory and hybridization. Acid and ...
... Nature and types of chemical bonding, lewis concepts, ionic, covalent, coordinate covalent bond; Valence bond theory (VBT), Molecular orbital theory (MOT). Interpretation of shapes of inorganic molecules on the basis of valence shell electron pair repulsion (VSEPR) theory and hybridization. Acid and ...
Low temperature synthesis of layered Na CoO and K
... Layered NaxCoO2 and KxCoO2 phases were synthesized from cobalt oxalate or nitrate salts mixed with NaOH or KOH (AR grade) in the weight ratio of 1 : 10 in a recrystallized alumina crucible. For the preparation of NaxCoO2 , a typical run contains CoC2 O4 .2H2 O (3⋅6591 g) and NaOH (7⋅9798 g). The mix ...
... Layered NaxCoO2 and KxCoO2 phases were synthesized from cobalt oxalate or nitrate salts mixed with NaOH or KOH (AR grade) in the weight ratio of 1 : 10 in a recrystallized alumina crucible. For the preparation of NaxCoO2 , a typical run contains CoC2 O4 .2H2 O (3⋅6591 g) and NaOH (7⋅9798 g). The mix ...
Chemistry (Revised)
... Marks SECTION B All answers must be written clearly and legibly in ink. 1. Attempts have been made to make foods healthier by using alternatives to traditional cooking ingredients. ...
... Marks SECTION B All answers must be written clearly and legibly in ink. 1. Attempts have been made to make foods healthier by using alternatives to traditional cooking ingredients. ...
B.Sc. (Hons.) Chemistry
... 3. Indexing of a given powder diffraction pattern of a cubic crystalline system. 4. pH metry a. Study the effect on pH of addition of HCl/NaOH to solutions of acetic acid, sodium acetate and their mixtures. b. Preparation of buffer solutions of different pH i. Sodium acetate-acetic acid ii. Ammonium ...
... 3. Indexing of a given powder diffraction pattern of a cubic crystalline system. 4. pH metry a. Study the effect on pH of addition of HCl/NaOH to solutions of acetic acid, sodium acetate and their mixtures. b. Preparation of buffer solutions of different pH i. Sodium acetate-acetic acid ii. Ammonium ...
Polymers
... 3) Explain Addition polymerisation. Give examples? Addition Polymerisation (Chain Growth Polymerisation) Addition or chain growth polymerisation is the linking of molecules having mul- tiple bonds. These unsaturated monomers have extra internal bonds which are able to break and link up with other m ...
... 3) Explain Addition polymerisation. Give examples? Addition Polymerisation (Chain Growth Polymerisation) Addition or chain growth polymerisation is the linking of molecules having mul- tiple bonds. These unsaturated monomers have extra internal bonds which are able to break and link up with other m ...
chemistry - The Aga Khan University
... 12.3.4 Batteries 12.3.4.1 Primary Batteries 12.3.4.2 Secondary Batteries 12.3.4.3 Fuel Cells 12.3.5 Corrosion and its Prevention ...
... 12.3.4 Batteries 12.3.4.1 Primary Batteries 12.3.4.2 Secondary Batteries 12.3.4.3 Fuel Cells 12.3.5 Corrosion and its Prevention ...
BRIEF ANSWERS TO SELECTED PROBLEMS APPENDIX G
... Si; 14 (c) Cu; 63.55 u (d) Br; 79.90 u 2.60 Atoms of these two kinds of substances will form ionic bonds, in which one or more electrons are transferred from the metal atom to the nonmetal atom to form a cation and an anion, respectively. 2.63 Coulomb’s law states the energy of attraction in an ioni ...
... Si; 14 (c) Cu; 63.55 u (d) Br; 79.90 u 2.60 Atoms of these two kinds of substances will form ionic bonds, in which one or more electrons are transferred from the metal atom to the nonmetal atom to form a cation and an anion, respectively. 2.63 Coulomb’s law states the energy of attraction in an ioni ...