Document
... Mg(s) + HCl(aq) H2(g) + MgCl2 (aq) We need to take steps to find the simplest ratios of these materials. - Step 1 - take an inventory of individual atoms shown in skeleton equation. ...
... Mg(s) + HCl(aq) H2(g) + MgCl2 (aq) We need to take steps to find the simplest ratios of these materials. - Step 1 - take an inventory of individual atoms shown in skeleton equation. ...
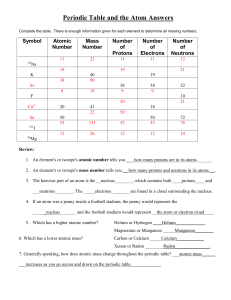
Periodic Table and the Atom Answers
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
... stoichiometry problems, I would highly suggest consulting this section of the site before answering these questions. When doing stoichiometry problems, people are frequently worried by statements such as “if you have an excess of (compound X)”. This statement shouldn’t worry you… what it really mean ...
MT 3 Practice
... 10. For the following reaction at 25°C I2(g) + Cl2(g) 2ICl(g) DH° = -26.9 kJ and DS° = 11.3 J/K. Calculate DG° for the reaction in kilojoules. [A] -50.6 kJ [B] 102 kJ [C] 50.6 kJ [D] -30.3 kJ 11. Given the following Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g) 3Fe2O3(s) + CO(g) 2Fe3O4(s) + CO2(g) calculate DG ...
... 10. For the following reaction at 25°C I2(g) + Cl2(g) 2ICl(g) DH° = -26.9 kJ and DS° = 11.3 J/K. Calculate DG° for the reaction in kilojoules. [A] -50.6 kJ [B] 102 kJ [C] 50.6 kJ [D] -30.3 kJ 11. Given the following Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g) 3Fe2O3(s) + CO(g) 2Fe3O4(s) + CO2(g) calculate DG ...
lecture1423183006
... consequence moves to the state B. The absorption of heat increase the internal energy of the ...
... consequence moves to the state B. The absorption of heat increase the internal energy of the ...
Writing Net Ionic Equations
... to go to completion. Unionized or partially ionized molecules give solutions that are known as nonelectrolytes or weak electrolytes. The best known nonelectrolyte is water formed in acid-base neutralization reactions. Acetic acid is an example of an acid that is primarily molecular (weak electrolyt ...
... to go to completion. Unionized or partially ionized molecules give solutions that are known as nonelectrolytes or weak electrolytes. The best known nonelectrolyte is water formed in acid-base neutralization reactions. Acetic acid is an example of an acid that is primarily molecular (weak electrolyt ...
Mr. Dehne AP Chem Name: ___________ Date: Per#: ___ AP
... 33. During the developing process of black-and-white film, silver bromide is removed from photographic film by the fixer. The major component of the fixer is sodium thiosulfate. The net ionic equation for the reaction is AgBr(s) + 2S2O32-(aq) → Ag(S2O3)23-(aq) + Br-(aq) What mass of AgBr can be diss ...
... 33. During the developing process of black-and-white film, silver bromide is removed from photographic film by the fixer. The major component of the fixer is sodium thiosulfate. The net ionic equation for the reaction is AgBr(s) + 2S2O32-(aq) → Ag(S2O3)23-(aq) + Br-(aq) What mass of AgBr can be diss ...
3. Chemical changes and Structure Unit Questions
... o The ionisation energy is the energy required to remove one mole of electrons from one mole of gaseous atoms o You can have second and third ionisation energies, but note that if there is a full outer shell the atom will not want to lose another electron making the energy very high. o The general f ...
... o The ionisation energy is the energy required to remove one mole of electrons from one mole of gaseous atoms o You can have second and third ionisation energies, but note that if there is a full outer shell the atom will not want to lose another electron making the energy very high. o The general f ...
Balancing Chemical Reactions
... Balancing Chemical Reactions - 100 • These are the only thing that can be used to balance chemical equations. ...
... Balancing Chemical Reactions - 100 • These are the only thing that can be used to balance chemical equations. ...
Chemistry 20 Lesson 36 – The Whole Enchilada
... Methanol is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure. a. Write the balanced chemical reaction. b. ...
... Methanol is used in the production of many chemicals. Methanol is made by reacting carbon monoxide and hydrogen at high temperature and pressure. a. Write the balanced chemical reaction. b. ...
C:\Users\mrh70950\Documents\My Files\WordPerfect
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
... i. anti-addition stereochemistry yields 1-alkenes from terminal alkynes and (E)-alkenes from internal alkynes 2. double hydrogenation: addition of 2 mol of H2 to yield alkanes a. noble metal catalyst + excess H2 3. electrophilic additions (all by very similar mechanisms) a. hydrohalogenation: addit ...
Guide to Chapter 17. Thermodynamics
... T/F b. When DH and DS are both positive, the reaction is non-spontaneous at all temperatures. T/F c. DS for vaporization is always positive. T/F d. DS for melting is always positive. T/F e. DH for vaporization is always positive. T/F f. If DH and DS are both positive, DG will increase with increasin ...
... T/F b. When DH and DS are both positive, the reaction is non-spontaneous at all temperatures. T/F c. DS for vaporization is always positive. T/F d. DS for melting is always positive. T/F e. DH for vaporization is always positive. T/F f. If DH and DS are both positive, DG will increase with increasin ...
Chapter 16
... compare Qc and Kc Ksp values are also a type of equilibrium constant, but are valid for saturated solutions only. We can use “ion product” (IP) to determine whether a precipitate will form: ...
... compare Qc and Kc Ksp values are also a type of equilibrium constant, but are valid for saturated solutions only. We can use “ion product” (IP) to determine whether a precipitate will form: ...
reactions taking place within cells
... • Incomplete combustion due to inadequate supply of O 2 leading to CO or C(indicated by deposit of soot on the bottom of calorimeter) 1st law of thermodynamics Energy can’t be created or destroyed, only converted from one form to another Hess’s law H for a reaction is independent of the route it ta ...
... • Incomplete combustion due to inadequate supply of O 2 leading to CO or C(indicated by deposit of soot on the bottom of calorimeter) 1st law of thermodynamics Energy can’t be created or destroyed, only converted from one form to another Hess’s law H for a reaction is independent of the route it ta ...
Chapter 19: Thermochemistry II: Entropy and free Energy
... FLASHBACK First Law: The internal energy of an isolated system is constant. Eisolated = constant What is internal energy ...
... FLASHBACK First Law: The internal energy of an isolated system is constant. Eisolated = constant What is internal energy ...
Strumenti tutor LIM
... A chemical transformation takes place when....................(atoms in the reactants are rearranged to form new substabces)(old bonds are broken and new bonds are formed)( at least one new substance is formed) We can realize that a chemical reaction is taking place when...........( there is a chang ...
... A chemical transformation takes place when....................(atoms in the reactants are rearranged to form new substabces)(old bonds are broken and new bonds are formed)( at least one new substance is formed) We can realize that a chemical reaction is taking place when...........( there is a chang ...
Chemical equilibrium
In a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations which have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. The reaction rates of the forward and backward reactions are generally not zero, but equal. Thus, there are no net changes in the concentrations of the reactant(s) and product(s). Such a state is known as dynamic equilibrium.