Science-M2-Basic-Che..
... At the heart of chemistry is physics—the study of attracting and repelling forces that link up atoms and molecules. A chemical bond is the physical process responsible for the attractive interactions between atoms and molecules, and which can make diatomic and polyatomic chemical compounds stable. I ...
... At the heart of chemistry is physics—the study of attracting and repelling forces that link up atoms and molecules. A chemical bond is the physical process responsible for the attractive interactions between atoms and molecules, and which can make diatomic and polyatomic chemical compounds stable. I ...
12021 14115 3 Hours / 100 Marks Seat No.
... a) Obtain an expression for intensity of electric field at a point in an electric field of charge Q coulomb, placed in a medium of dielectric constant K. b) Define electric lines of force. State its any three properties. c) A sphere of radius 20 cm is given a charge of 1600 microcoulomb and placed i ...
... a) Obtain an expression for intensity of electric field at a point in an electric field of charge Q coulomb, placed in a medium of dielectric constant K. b) Define electric lines of force. State its any three properties. c) A sphere of radius 20 cm is given a charge of 1600 microcoulomb and placed i ...
Ch 11 Chemical Reactions
... Reactants = the substances you start with, change into… Products = the substances you end up with Reactants Products ...
... Reactants = the substances you start with, change into… Products = the substances you end up with Reactants Products ...
Basic Background Review: Acid-Base , Redox, and Stable Isotopes
... methane C, CH2O CO2 and other dissolved inorganic carbon species ...
... methane C, CH2O CO2 and other dissolved inorganic carbon species ...
AP CHEMISTRY - An Incomplete List of Topics
... The total loss of electrons by the oxidation reaction(s) must equal the total gain of electrons by the reduction reaction(s). First, adjust the coefficients of the reactants and products that contain the oxidized and reduced elements, then balance any other elements by inspection. ...
... The total loss of electrons by the oxidation reaction(s) must equal the total gain of electrons by the reduction reaction(s). First, adjust the coefficients of the reactants and products that contain the oxidized and reduced elements, then balance any other elements by inspection. ...
Too Hot to Handle Lab
... heat, and the prefix – “exo” which means out of. Heat comes out of, or is released from, a reacting substance during an exothermic reaction. A reaction that involves burning, or a combustion reaction, is an example of an exothermic reaction. The combustion of methane gas, which occurs in a gas stove ...
... heat, and the prefix – “exo” which means out of. Heat comes out of, or is released from, a reacting substance during an exothermic reaction. A reaction that involves burning, or a combustion reaction, is an example of an exothermic reaction. The combustion of methane gas, which occurs in a gas stove ...
1 - M*W
... gas. Which of the following represents the reactants in this reaction? a) Magnesium and magnesium chloride b) Hydrochloric acid and hydrogen gas c) Magnesium and hydrochloric acid d) Magnesium chloride and hydrogen gas 50) The equation E=mc2 shows that a) Chemical reactions are either exothermic or ...
... gas. Which of the following represents the reactants in this reaction? a) Magnesium and magnesium chloride b) Hydrochloric acid and hydrogen gas c) Magnesium and hydrochloric acid d) Magnesium chloride and hydrogen gas 50) The equation E=mc2 shows that a) Chemical reactions are either exothermic or ...
Chapter 20 Electrochemistry
... acidic solution. (a) Cu(s) + NO3(aq) Cu2+(aq) + NO2(g) (b) Mn2+(aq) + NaBiO3(s) Bi3+(aq) + MnO4(aq) Electrochemistry ...
... acidic solution. (a) Cu(s) + NO3(aq) Cu2+(aq) + NO2(g) (b) Mn2+(aq) + NaBiO3(s) Bi3+(aq) + MnO4(aq) Electrochemistry ...
CHEMISTRY EXAM 2 REVIEW
... My child completed this review and studied for at least 30 minutes. Define the following chemistry terms: [Chemistry Dictionary] 1. alloy a mixture of metals 2. brittleness the property of matter that is how easily the substance breaks or shatters when force is applied to it. 3. compound a substance ...
... My child completed this review and studied for at least 30 minutes. Define the following chemistry terms: [Chemistry Dictionary] 1. alloy a mixture of metals 2. brittleness the property of matter that is how easily the substance breaks or shatters when force is applied to it. 3. compound a substance ...
Matter, Mass and Weight
... element is represented by a symbol: H for hydrogen, O for oxygen, C for carbon, Cl for chlorine. Some elements are not stable as a single atoms but form stable units called molecules. Symbols for hydrogen and oxygen are H and O but their molecular formulas are H2 and O2 A compound is a pure substanc ...
... element is represented by a symbol: H for hydrogen, O for oxygen, C for carbon, Cl for chlorine. Some elements are not stable as a single atoms but form stable units called molecules. Symbols for hydrogen and oxygen are H and O but their molecular formulas are H2 and O2 A compound is a pure substanc ...
Grade 11 Chemistry Exam Review
... Element X consists of 30.00% of an isotope with mass 24.02 u and 70.00% of an isotope with mass 26.10 u. The average atomic mass of X is a) 24.64 u. b) 25.06 u. c) 25.48 u. d) 50.12 u ...
... Element X consists of 30.00% of an isotope with mass 24.02 u and 70.00% of an isotope with mass 26.10 u. The average atomic mass of X is a) 24.64 u. b) 25.06 u. c) 25.48 u. d) 50.12 u ...
Balancing Chemical Equations
... You cannot add a subscript number in a reaction to change the amount of atoms, you can only change the amount of compounds. Therefore you add another to the front of the compound. Since the compounds cannot be separated, you cannot just add a two in front of the oxygen atom only, it has to be in fro ...
... You cannot add a subscript number in a reaction to change the amount of atoms, you can only change the amount of compounds. Therefore you add another to the front of the compound. Since the compounds cannot be separated, you cannot just add a two in front of the oxygen atom only, it has to be in fro ...
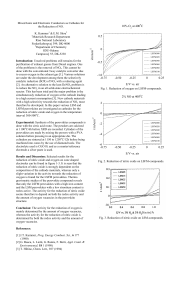
-6 -4 -2 0 2 -0.75 -0.50 -0.25 0 0.25 E/V vs. air i/m A cm 2% NO at
... done with the conventional 3way catalytic converter due to excess oxygen in the exhaust gas [1]. Various solutions are under the development among them the selectively catalytic reduction (SCR) of NOx with a reducing agent [2]. An alternative solution to the lean DeNOx problem is to reduce the NOx i ...
... done with the conventional 3way catalytic converter due to excess oxygen in the exhaust gas [1]. Various solutions are under the development among them the selectively catalytic reduction (SCR) of NOx with a reducing agent [2]. An alternative solution to the lean DeNOx problem is to reduce the NOx i ...
Subject:
... Big Idea/Theme: Understanding how effective collisions encourage chemical reactions and how pressure, temperature and concentration affects the progression of equilibrium in a reversible reaction Learning Targets: (“I can” or “I will” statements) I will understand the factors that affect the rate of ...
... Big Idea/Theme: Understanding how effective collisions encourage chemical reactions and how pressure, temperature and concentration affects the progression of equilibrium in a reversible reaction Learning Targets: (“I can” or “I will” statements) I will understand the factors that affect the rate of ...
Types of Reactions notes 02 Types of chemical reactions
... When you see a chemical formula, often the formula is followed by a symbol in parentheses. For example: H2O(l) - the water is liquid H2O(s) - the water is solid (ice) H2O(g)- the water is a gas (steam) NaCl(aq) – means that the chemical is disolved in water. In this case it would be salt dis ...
... When you see a chemical formula, often the formula is followed by a symbol in parentheses. For example: H2O(l) - the water is liquid H2O(s) - the water is solid (ice) H2O(g)- the water is a gas (steam) NaCl(aq) – means that the chemical is disolved in water. In this case it would be salt dis ...
1 ChE 505 WORKSHOP 1 1. Why are chemical reactions important
... What is the relationship between the initial moles of reactants and products, the moles for each of the above after some reaction time, the stoichiometric coefficients and reaction extent? ...
... What is the relationship between the initial moles of reactants and products, the moles for each of the above after some reaction time, the stoichiometric coefficients and reaction extent? ...
Chemical Reactions Notes-1a-1
... The positive ions have the surrounding oxygen atoms of water pointing towards the ion, negative ions have the surrounding hydrogen atoms of water pointing towards the ion. The transport of ions through the solution causes electric current to flow through the solution. ...
... The positive ions have the surrounding oxygen atoms of water pointing towards the ion, negative ions have the surrounding hydrogen atoms of water pointing towards the ion. The transport of ions through the solution causes electric current to flow through the solution. ...
Electricity Notes
... A. Electricity begins at the atomic level where protons and electrons have electric charge. 1. Protons carry a positive change. 2. Electrons carry a negative charge. 3. Ions form when atoms lose or gain electrons and become positively or negatively charged. 4. Electrons can move from object to obje ...
... A. Electricity begins at the atomic level where protons and electrons have electric charge. 1. Protons carry a positive change. 2. Electrons carry a negative charge. 3. Ions form when atoms lose or gain electrons and become positively or negatively charged. 4. Electrons can move from object to obje ...
Oxidation numbers
... · Loses electrons Reduction takes place when an element in a reaction · Decreases its oxidation number · Gains electrons To help you remember this, you can use “OIL RIG” – Oxidation is Loss Reduction is Gain (of electrons) ...
... · Loses electrons Reduction takes place when an element in a reaction · Decreases its oxidation number · Gains electrons To help you remember this, you can use “OIL RIG” – Oxidation is Loss Reduction is Gain (of electrons) ...
Question to answer… - Rochester Century High School
... reactions using symbols and formulas Instead of writing: “When you add solid silver to hydrogen sulfide gas, you get solid silver (I) sulfide and diatomic hydrogen gas.” You can just write: Ag(s) + H2S(g) --> Ag2S(s) + H2 (g) ...
... reactions using symbols and formulas Instead of writing: “When you add solid silver to hydrogen sulfide gas, you get solid silver (I) sulfide and diatomic hydrogen gas.” You can just write: Ag(s) + H2S(g) --> Ag2S(s) + H2 (g) ...
Exam #2
... mass of the nucleus is concentrated in a very small volume. The electron diffraction experiment demonstrated Heisenberg’s hypothesis that matter and energy are interconvertable. The solution to the Schrodinger wave equation for the hydrogen atom does not provide a detailed description of the electro ...
... mass of the nucleus is concentrated in a very small volume. The electron diffraction experiment demonstrated Heisenberg’s hypothesis that matter and energy are interconvertable. The solution to the Schrodinger wave equation for the hydrogen atom does not provide a detailed description of the electro ...
Electrochemistry
Electrochemistry is the branch of physical chemistry that studies chemical reactions which take place at the interface of an electrode, usually a solid metal or a semiconductor, and an ionic conductor, the electrolyte. These reactions involve electric charges moving between the electrodes and the electrolyte (or ionic species in a solution). Thus electrochemistry deals with the interaction between electrical energy and chemical change.When a chemical reaction is caused by an externally supplied current, as in electrolysis, or if an electric current is produced by a spontaneous chemical reaction as in a battery, it is called an electrochemical reaction. Chemical reactions where electrons are transferred directly between molecules and/or atoms are called oxidation-reduction or (redox) reactions. In general, electrochemistry describes the overall reactions when individual redox reactions are separate but connected by an external electric circuit and an intervening electrolyte.