Slide 1 - s3.amazonaws.com
... 7.4 Quantum Mechanics Physicists were both mystified and intrigued by Bohr’s theory. They questioned why the energies of hydrogen electron are quantized, or, why is the electron in a Bohr atom restricted or orbiting the nucleus at certain fixed distance? For a decade there is no logical explanation ...
... 7.4 Quantum Mechanics Physicists were both mystified and intrigued by Bohr’s theory. They questioned why the energies of hydrogen electron are quantized, or, why is the electron in a Bohr atom restricted or orbiting the nucleus at certain fixed distance? For a decade there is no logical explanation ...
e-the-quantum-numberssv-2
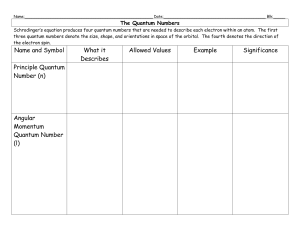
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
... The Quantum Numbers Schrodinger’s equation produces four quantum numbers that are needed to describe each electron within an atom. The first three quantum numbers denote the size, shape, and orientations in space of the orbital. The fourth denotes the direction of the electron spin. ...
High Efficiency Quantum- well Quantum-dot Solar Cells
... High Efficiency Quantumwell Quantum-dot Solar Cells Presented by: ...
... High Efficiency Quantumwell Quantum-dot Solar Cells Presented by: ...