Stoichiometry notes 1
... Reminder - The five steps of stoichiometry 1. Write a balanced chemical equation. 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the m ...
... Reminder - The five steps of stoichiometry 1. Write a balanced chemical equation. 2. Label your given and target substances. 3. Convert your given unit(s) to moles of given substance using the appropriate conversion factor. 4. Convert moles of given substance to moles of target substance using the m ...
Chapter 21 The Electric Field I: Discrete Charge Distributions
... (c) False. Electric field lines intersect any point in space occupied by a point charge. (d) True. An electric field partially polarizes the molecules; resulting in the separation of their charges and the creation of electric dipole moments. 17 ••• [SSM] Two molecules have dipole moments of equal ma ...
... (c) False. Electric field lines intersect any point in space occupied by a point charge. (d) True. An electric field partially polarizes the molecules; resulting in the separation of their charges and the creation of electric dipole moments. 17 ••• [SSM] Two molecules have dipole moments of equal ma ...
topic: chemical formula, chemical equations and stoichiometry
... The relative atomic mass of an element = Mass of 1 atom of the element 1/12 x mass of 1 atom of carbon-12 The relative molecular mass, RMM, of a compound is defined as, The number of times the mass of one molecule of a compound is heavier than 1/12 times the mass of one carbon-12 atom. The relative ...
... The relative atomic mass of an element = Mass of 1 atom of the element 1/12 x mass of 1 atom of carbon-12 The relative molecular mass, RMM, of a compound is defined as, The number of times the mass of one molecule of a compound is heavier than 1/12 times the mass of one carbon-12 atom. The relative ...
Conceptual Questions - Colorado Mesa University
... 25.1. An insulator can be charged. Plastic is an insulator. A plastic rod can be charged by rubbing it with wool. 25.2. A conductor can be charged. A conductor can be charged by touching it with another charged object. 25.3. B and D are both neutral because they have no effect on each other and neut ...
... 25.1. An insulator can be charged. Plastic is an insulator. A plastic rod can be charged by rubbing it with wool. 25.2. A conductor can be charged. A conductor can be charged by touching it with another charged object. 25.3. B and D are both neutral because they have no effect on each other and neut ...
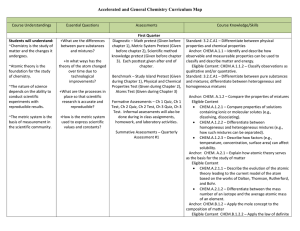
Chemistry Curriculum Map - Belle Vernon Area School District
... the rate of chemical reactions. Standard: 3.2.C.A1 – Explain the relationship of an elements position on the periodic table to its atomic number, ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHEM.A.2.1 – Explain how atomic theory serves as the basis for ...
... the rate of chemical reactions. Standard: 3.2.C.A1 – Explain the relationship of an elements position on the periodic table to its atomic number, ionization energy, electro-negativity, atomic size, and classification of elements. Anchor: CHEM.A.2.1 – Explain how atomic theory serves as the basis for ...
Experimental Test of Wave-Particle Duality by Single Neutral Atoms
... Various tests of BCP with single photons have been performed [2–10]. However, the low detection efficiency associated with fast-moving, massless photons makes the results less persuasive and quite untenable. Here we use a well-controlled, massive, single trapped Cesium atom in a Ramsey interferomete ...
... Various tests of BCP with single photons have been performed [2–10]. However, the low detection efficiency associated with fast-moving, massless photons makes the results less persuasive and quite untenable. Here we use a well-controlled, massive, single trapped Cesium atom in a Ramsey interferomete ...
JOURNAL OF CONDENSED MATTER NUCLEAR SCIENCE Experiments and Methods in Cold Fusion
... track [17,18]. When the detector is etched, the tracks are enlarged until they are visible with the aid of a microscope. However, neutrons only leave tracks under certain conditions. The neutron must either elastically scatter off or undergo an inelastic nuclear reaction with, the hydrogen, carbon, ...
... track [17,18]. When the detector is etched, the tracks are enlarged until they are visible with the aid of a microscope. However, neutrons only leave tracks under certain conditions. The neutron must either elastically scatter off or undergo an inelastic nuclear reaction with, the hydrogen, carbon, ...
ElectroGravitics_01
... suggested to test one or several of these theoretical views. This is an area where the experimental procedure is workable and the outcome could have direct results in the area of inertia forces. "During the late 60s, William J. Hooper put forth an interesting theory involving the v x B terms dynamic ...
... suggested to test one or several of these theoretical views. This is an area where the experimental procedure is workable and the outcome could have direct results in the area of inertia forces. "During the late 60s, William J. Hooper put forth an interesting theory involving the v x B terms dynamic ...
Electrogravitic References
... suggested to test one or several of these theoretical views. This is an area where the experimental procedure is workable and the outcome could have direct results in the area of inertia forces. "During the late 60s, William J. Hooper put forth an interesting theory involving the v x B terms dynamic ...
... suggested to test one or several of these theoretical views. This is an area where the experimental procedure is workable and the outcome could have direct results in the area of inertia forces. "During the late 60s, William J. Hooper put forth an interesting theory involving the v x B terms dynamic ...
Light, Matter, and Geometry: The Cornerstones of
... is the gap and the missing bridge. If there had been a book closing the gap, or providing the bridge, this thesis would have been entirely different. As it is, ...
... is the gap and the missing bridge. If there had been a book closing the gap, or providing the bridge, this thesis would have been entirely different. As it is, ...
Lattice QCD and String Theory Lattice 2005 Julius Kuti Confining Force
... Polchinski and Strominger: we need a new effective string description For strings emerging from field theory we would like to require D-2 oscillators, Poincare invariance, and dim invariance Goldstone theorem does not protect longitudinal mode from acquiring a mass (breathing mode of Z(2) vortex is ...
... Polchinski and Strominger: we need a new effective string description For strings emerging from field theory we would like to require D-2 oscillators, Poincare invariance, and dim invariance Goldstone theorem does not protect longitudinal mode from acquiring a mass (breathing mode of Z(2) vortex is ...
Search for anomalous production of prompt same-sign
... 8 Results and interpretation 8.1 Signal region 8.2 Fiducial cross-section limits 8.3 Cross-section and mass limits for pair-produced doubly charged Higgs bosons ...
... 8 Results and interpretation 8.1 Signal region 8.2 Fiducial cross-section limits 8.3 Cross-section and mass limits for pair-produced doubly charged Higgs bosons ...
DISCRETE 2016: Fifth Symposium on Prospects in the Physics of
... We study CPT and Lorentz violation (LV) in the electroweak gauge sector of the Standard Model in the context of the Standard-Model Extension (SME). In particular, we show that any non-zero value of a certain relevant LV parameter that is thus far unbounded by experiment would imply that for sufficie ...
... We study CPT and Lorentz violation (LV) in the electroweak gauge sector of the Standard Model in the context of the Standard-Model Extension (SME). In particular, we show that any non-zero value of a certain relevant LV parameter that is thus far unbounded by experiment would imply that for sufficie ...
HCSS-June09-partA - Indico
... opposing beam at the collision point, it senses the fields of the opposing beam. Due to the typically Gaussian shape of the beams in the transverse direction, the field (force) on this particle is non-linear, in particular at large amplitudes ! The effect of the non-linear fields can become so s ...
... opposing beam at the collision point, it senses the fields of the opposing beam. Due to the typically Gaussian shape of the beams in the transverse direction, the field (force) on this particle is non-linear, in particular at large amplitudes ! The effect of the non-linear fields can become so s ...
Three particle Hyper Entanglement: Teleportation and Quantum Key
... state [1], super dense coding of information [2] and secure communication [3]. An arbitrary qubit can be teleported from one particle to another with the use of an entangled pair of particles, which had been experimentally verified in different quantum systems [4, 5]. However, distinguishing all the ...
... state [1], super dense coding of information [2] and secure communication [3]. An arbitrary qubit can be teleported from one particle to another with the use of an entangled pair of particles, which had been experimentally verified in different quantum systems [4, 5]. However, distinguishing all the ...
Elementary particle
In particle physics, an elementary particle or fundamental particle is a particle whose substructure is unknown, thus it is unknown whether it is composed of other particles. Known elementary particles include the fundamental fermions (quarks, leptons, antiquarks, and antileptons), which generally are ""matter particles"" and ""antimatter particles"", as well as the fundamental bosons (gauge bosons and Higgs boson), which generally are ""force particles"" that mediate interactions among fermions. A particle containing two or more elementary particles is a composite particle.Everyday matter is composed of atoms, once presumed to be matter's elementary particles—atom meaning ""indivisible"" in Greek—although the atom's existence remained controversial until about 1910, as some leading physicists regarded molecules as mathematical illusions, and matter as ultimately composed of energy. Soon, subatomic constituents of the atom were identified. As the 1930s opened, the electron and the proton had been observed, along with the photon, the particle of electromagnetic radiation. At that time, the recent advent of quantum mechanics was radically altering the conception of particles, as a single particle could seemingly span a field as would a wave, a paradox still eluding satisfactory explanation.Via quantum theory, protons and neutrons were found to contain quarks—up quarks and down quarks—now considered elementary particles. And within a molecule, the electron's three degrees of freedom (charge, spin, orbital) can separate via wavefunction into three quasiparticles (holon, spinon, orbiton). Yet a free electron—which, not orbiting an atomic nucleus, lacks orbital motion—appears unsplittable and remains regarded as an elementary particle.Around 1980, an elementary particle's status as indeed elementary—an ultimate constituent of substance—was mostly discarded for a more practical outlook, embodied in particle physics' Standard Model, science's most experimentally successful theory. Many elaborations upon and theories beyond the Standard Model, including the extremely popular supersymmetry, double the number of elementary particles by hypothesizing that each known particle associates with a ""shadow"" partner far more massive, although all such superpartners remain undiscovered. Meanwhile, an elementary boson mediating gravitation—the graviton—remains hypothetical.