Physphax Review
... 26. A force F is a push or pull. Forces are vectors: F = magnitude (strength of force) + direction. 27. Forces measured in newtons, N (derived). 1 N = 1 kg·m/s2 = weight of a stick of butter or small apple 28. Two basic types: a/ contact: normal, tension, friction. b/ at a distance: weight & other f ...
... 26. A force F is a push or pull. Forces are vectors: F = magnitude (strength of force) + direction. 27. Forces measured in newtons, N (derived). 1 N = 1 kg·m/s2 = weight of a stick of butter or small apple 28. Two basic types: a/ contact: normal, tension, friction. b/ at a distance: weight & other f ...
Objective of the course Aim of the course is to introduce the basic
... Objective of the course Aim of the course is to introduce the basic notions of non-relativistic quantum mechanics and its interpretation. At the end of the course the students should: 1) have understood the definition of physical state and the superposition principle in quantum mechanics, the defini ...
... Objective of the course Aim of the course is to introduce the basic notions of non-relativistic quantum mechanics and its interpretation. At the end of the course the students should: 1) have understood the definition of physical state and the superposition principle in quantum mechanics, the defini ...
the constancy of for an ideal gas undergoing an adiabatic
... Could this equation be a basis to build a bridge between the law of gases (mainly characterized by the Boltzmann Constant), and quantum mechanics (which will evidently involve the energy quantity E )? Here though, while the equality pV kT points to the law of gases, the next equality kT (2 / 3) ...
... Could this equation be a basis to build a bridge between the law of gases (mainly characterized by the Boltzmann Constant), and quantum mechanics (which will evidently involve the energy quantity E )? Here though, while the equality pV kT points to the law of gases, the next equality kT (2 / 3) ...
Investigating Matter Notes
... 1.2 Investigating Matter Notes There are 2 types of changes in matter: Chemical Change A chemical change is a _____________________ change in matter that occurs when _____________ combine to form ______________ substances. We say a _____________________________ has been formed because the __________ ...
... 1.2 Investigating Matter Notes There are 2 types of changes in matter: Chemical Change A chemical change is a _____________________ change in matter that occurs when _____________ combine to form ______________ substances. We say a _____________________________ has been formed because the __________ ...
MS-word - Table of Contents
... profound meaning to this? 4. Origin of the Natural Laws. The two combinations contain all electron-positron properties. Charge depends on whether there is a + or amplitude of the IN wave at the center. If a resonance is superimposed upon an anti-resonance, they annihilate. The amplitude at the cente ...
... profound meaning to this? 4. Origin of the Natural Laws. The two combinations contain all electron-positron properties. Charge depends on whether there is a + or amplitude of the IN wave at the center. If a resonance is superimposed upon an anti-resonance, they annihilate. The amplitude at the cente ...
Physical Quantities and Units
... which produces a magnetic force per unit length of 2 x 10-7 Nm-1 on each wire Charge – The property of some elementary particles which give rise to an interaction between them and consequently a host of material phenomena described as electrical. Charge, Q : The quantity of electricity passing a ...
... which produces a magnetic force per unit length of 2 x 10-7 Nm-1 on each wire Charge – The property of some elementary particles which give rise to an interaction between them and consequently a host of material phenomena described as electrical. Charge, Q : The quantity of electricity passing a ...
Notes - SFA Physics and Astronomy
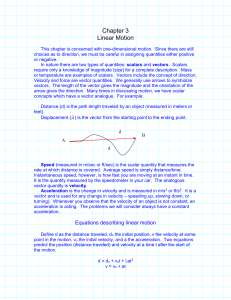
... rate at which distance is covered. Average speed is simply distance/time. Instantaneous speed, however, is how fast you are moving at an instant in time. It is the quantity measured by the speedometer in your car. The analogous vector quantity is velocity. Acceleration is the change in velocity and ...
... rate at which distance is covered. Average speed is simply distance/time. Instantaneous speed, however, is how fast you are moving at an instant in time. It is the quantity measured by the speedometer in your car. The analogous vector quantity is velocity. Acceleration is the change in velocity and ...
Quantum Theory
... rules that apply to the world we see are only approximations of the rules that govern the unseen world of light and subatomic particles. ...
... rules that apply to the world we see are only approximations of the rules that govern the unseen world of light and subatomic particles. ...
Lecture13
... by restoring forces and the system do not oscillate indefinitely. The friction reduces the mechanical energy of the system as time passes, and the motion is said to be damped. ...
... by restoring forces and the system do not oscillate indefinitely. The friction reduces the mechanical energy of the system as time passes, and the motion is said to be damped. ...
Bohr`s Model of the Atom - Mr. Walsh`s AP Chemistry
... nucleus as in Rutherford’s model, but that these electrons had only certain allowed quantum values of energy, which could be described by a quantum number (n). The value of that quantum number was the same n as in Rydberg’s equation, and that using quantum numbers in Rydberg’s equation could predict ...
... nucleus as in Rutherford’s model, but that these electrons had only certain allowed quantum values of energy, which could be described by a quantum number (n). The value of that quantum number was the same n as in Rydberg’s equation, and that using quantum numbers in Rydberg’s equation could predict ...
kinetic energy of photoelectrons (eV)
... discrete amounts of energy (or whole number multiples) •He called the bundles quanta (singular: quantum) ...
... discrete amounts of energy (or whole number multiples) •He called the bundles quanta (singular: quantum) ...
Chapter 2 - UCF Chemistry
... • Bohr’s theory correctly explains the H emission spectrum and those of hydrogenlike ions (He+, Li2+ … 1e− species) • The theory fails for atoms of all other elements because it is not an adequate theory: it doesn’t take into account the fact that the (very small) electron can be thought as having w ...
... • Bohr’s theory correctly explains the H emission spectrum and those of hydrogenlike ions (He+, Li2+ … 1e− species) • The theory fails for atoms of all other elements because it is not an adequate theory: it doesn’t take into account the fact that the (very small) electron can be thought as having w ...
Qualifying Exam for Graduate Students – Fall 2008
... 1. In 1883, the volcano on the island of Krakatoa erupted with a force 13,000 times the atomic bomb dropped on Hiroshima, Japan. According to Wikipedia, it was measured to be 180 dB 100 miles away and was clearly audible 2000 miles away. Somewhere along the line, it was designated as the “loudest so ...
... 1. In 1883, the volcano on the island of Krakatoa erupted with a force 13,000 times the atomic bomb dropped on Hiroshima, Japan. According to Wikipedia, it was measured to be 180 dB 100 miles away and was clearly audible 2000 miles away. Somewhere along the line, it was designated as the “loudest so ...
Chapter 2 - UCF Chemistry
... • Bohr’s theory correctly explains the H emission spectrum and those of hydrogenlike ions (He+, Li2+ … 1e− species) • The theory fails for atoms of all other elements because it is not an adequate theory: it doesn’t take into account the fact that the (very small) electron can be thought as having w ...
... • Bohr’s theory correctly explains the H emission spectrum and those of hydrogenlike ions (He+, Li2+ … 1e− species) • The theory fails for atoms of all other elements because it is not an adequate theory: it doesn’t take into account the fact that the (very small) electron can be thought as having w ...
Fractional Quantum Hall effect in a Curved Space
... - Particle density is local, coordinate invariant quantity => composed entirely of curvature invariants: Scalar curvature and Laplace-Beltrami. Functional form is FIXED, only coefficients may vary. True for any state in ...
... - Particle density is local, coordinate invariant quantity => composed entirely of curvature invariants: Scalar curvature and Laplace-Beltrami. Functional form is FIXED, only coefficients may vary. True for any state in ...
1-QM Foundations
... Bohr also concluded that electron waves must be standing waves, waves that appear fixed in place and simply oscillate vertically up to a maximum positive amplitude and down to a minimum negative amplitude. Standing waves are often found when a wave of a specific frequency moves in one direction whil ...
... Bohr also concluded that electron waves must be standing waves, waves that appear fixed in place and simply oscillate vertically up to a maximum positive amplitude and down to a minimum negative amplitude. Standing waves are often found when a wave of a specific frequency moves in one direction whil ...
Red-Electrostatics Protons have what type of charge? Electrons
... A wave that vibrates perpendicular to the direction of the wave motion is what type of wave? The high region area of a longitudinal wave is called what? The type of wave produced by two identical waves traveling in the opposite direction. A(n) ____ is produced during constructive interference of wav ...
... A wave that vibrates perpendicular to the direction of the wave motion is what type of wave? The high region area of a longitudinal wave is called what? The type of wave produced by two identical waves traveling in the opposite direction. A(n) ____ is produced during constructive interference of wav ...