Physics 214b-2008 Walter F
... IMPORTANT: This exam will be truly cumulative, i.e. it will cover material from the entire semester. For example, it will cover material such as the quantum nature of light that we discussed back in chapter 1. However, there will be some extra emphasis on the material since exam 2, since you’ve not ...
... IMPORTANT: This exam will be truly cumulative, i.e. it will cover material from the entire semester. For example, it will cover material such as the quantum nature of light that we discussed back in chapter 1. However, there will be some extra emphasis on the material since exam 2, since you’ve not ...
exploiting the superposition principle foundations and applications
... Clocks and matter wave interferometer test two aspects of the Einstein principle of equivalence: "2 sides of the same medal" Matter wave interferometry extend the range of test parameters (Holger Müller and Mike Hohensee) Matter-wave tests are yet orders of magnitude off from ...
... Clocks and matter wave interferometer test two aspects of the Einstein principle of equivalence: "2 sides of the same medal" Matter wave interferometry extend the range of test parameters (Holger Müller and Mike Hohensee) Matter-wave tests are yet orders of magnitude off from ...
PPT
... F is the minimum energy needed to strip an electron from the metal. F is defined as positive. Not all electrons will leave with the maximum kinetic energy (due to losses). ...
... F is the minimum energy needed to strip an electron from the metal. F is defined as positive. Not all electrons will leave with the maximum kinetic energy (due to losses). ...
On the Linkage between Planck`s Quantum and
... How can an atom serve as one-wavelength dipole for any wavelength emitted? As assumed by both Wilhelm Wien and Max Planck, blackbody radiation is emitted by the atoms or molecules at the blackbody surface - not least due to the linkage of the energy of a quantum of radiation to the average thermal e ...
... How can an atom serve as one-wavelength dipole for any wavelength emitted? As assumed by both Wilhelm Wien and Max Planck, blackbody radiation is emitted by the atoms or molecules at the blackbody surface - not least due to the linkage of the energy of a quantum of radiation to the average thermal e ...
2 t ) a
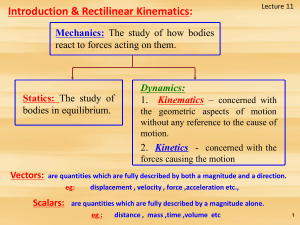
... The three kinematic equations can be integrated for the special case when acceleration is constant ( a = ac ) to obtain very useful equations. A common example of constant acceleration is gravity; i.e., a body freely falling toward earth. In this case, ( ac = g = 9.81 m/s2 = 32.2 ft/s2 ) downward. T ...
... The three kinematic equations can be integrated for the special case when acceleration is constant ( a = ac ) to obtain very useful equations. A common example of constant acceleration is gravity; i.e., a body freely falling toward earth. In this case, ( ac = g = 9.81 m/s2 = 32.2 ft/s2 ) downward. T ...
Review
... Bohr got energy right, but he said angular momentum L=nħ, and thought the electron was a point particle orbiting around nucleus (like planetary system). ...
... Bohr got energy right, but he said angular momentum L=nħ, and thought the electron was a point particle orbiting around nucleus (like planetary system). ...
CHAPTER 7: The Hydrogen Atom
... The Dutch physicist Pieter Zeeman showed the spectral lines emitted by atoms in a magnetic field split into multiple energy levels. It is called the Zeeman effect. A spectral line is split into three lines. Consider the atom to behave like a small magnet. Think of an electron as an orbiting circular ...
... The Dutch physicist Pieter Zeeman showed the spectral lines emitted by atoms in a magnetic field split into multiple energy levels. It is called the Zeeman effect. A spectral line is split into three lines. Consider the atom to behave like a small magnet. Think of an electron as an orbiting circular ...
The Heisenberg Uncertainty derivations
... it’s -value is known with zero uncertainty), then it is also automatically in an eigenstate of , and hence its -value is also known precisely (with no uncertainty). Consider instead the case where the two observables don’t commute (and hence have different eigenvectors). Suppose the system is in a s ...
... it’s -value is known with zero uncertainty), then it is also automatically in an eigenstate of , and hence its -value is also known precisely (with no uncertainty). Consider instead the case where the two observables don’t commute (and hence have different eigenvectors). Suppose the system is in a s ...
Chapter 6 Electronic Structure of Atoms
... 3. Energy is only absorbed or emitted in such a way as to move an electron from one “allowed” energy state to another; the energy is defined by E = h Electronic Structure of Atoms ...
... 3. Energy is only absorbed or emitted in such a way as to move an electron from one “allowed” energy state to another; the energy is defined by E = h Electronic Structure of Atoms ...
Tutorial 8 Angular Momentum and Planar Kinematics
... apogee C. (b) Use conservation of energy to determine the magnitude of the velocity at C. (c) To determine the magnitudes of the radial velocity vr and transverse velocity vθ. at B. ...
... apogee C. (b) Use conservation of energy to determine the magnitude of the velocity at C. (c) To determine the magnitudes of the radial velocity vr and transverse velocity vθ. at B. ...
A Suggested Interpretation of the Quantum Theory in Terms of
... the particle position, but can say only that the particle must be somewhere in the region in which ~f~ is appreciable. Similarly, the momentum of a particle that happens to be at the point, x, is given by p= Vs(x), so that since x is not known, the precise value of p is also not, in general, inferra ...
... the particle position, but can say only that the particle must be somewhere in the region in which ~f~ is appreciable. Similarly, the momentum of a particle that happens to be at the point, x, is given by p= Vs(x), so that since x is not known, the precise value of p is also not, in general, inferra ...
Classical Particles Having Complex Energy Exhibit Quantum
... particle drift through the lattice in one direction only (see Fig. 6). After running continuously on a computer for several months to determine which complex energies give rise to tunneling (hopping) behavior and which complex energies produce conduction-like behavior, we have found that conduction ...
... particle drift through the lattice in one direction only (see Fig. 6). After running continuously on a computer for several months to determine which complex energies give rise to tunneling (hopping) behavior and which complex energies produce conduction-like behavior, we have found that conduction ...
Quantum Physics Notes
... Newton's theory - light consists of particles called corpuscles; this theory only explained reflection Wave theory of light (Maxwell's theory) - light behaves like a wave; this explained all the properties of light such as reflection, refraction, diffraction, and interference; it did not explain the ...
... Newton's theory - light consists of particles called corpuscles; this theory only explained reflection Wave theory of light (Maxwell's theory) - light behaves like a wave; this explained all the properties of light such as reflection, refraction, diffraction, and interference; it did not explain the ...
All the 5`s - The Physics Teacher
... For a fixed mass of gas at constant temperature, pressure is inversely proportional to volume. b) The moon orbits the earth. What is the relationship between the period of the moon and the radius of its orbit? The period squared is proportional to the radius cubed c) Why is it necessary to have a st ...
... For a fixed mass of gas at constant temperature, pressure is inversely proportional to volume. b) The moon orbits the earth. What is the relationship between the period of the moon and the radius of its orbit? The period squared is proportional to the radius cubed c) Why is it necessary to have a st ...
Name - MIT
... intensity of emitted light decreases. C) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. D) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light decreases. E) the emitted power per squar ...
... intensity of emitted light decreases. C) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. D) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light decreases. E) the emitted power per squar ...
Name
... B) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. C) the emitted power per square meter increases and the wavelength of maximum intensity of emitted light decreases. D) the emitted power per square meter decreases and the wavelength o ...
... B) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. C) the emitted power per square meter increases and the wavelength of maximum intensity of emitted light decreases. D) the emitted power per square meter decreases and the wavelength o ...
Name
... B) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. C) the emitted power per square meter increases and the wavelength of maximum intensity of emitted light decreases. D) the emitted power per square meter decreases and the wavelength o ...
... B) the emitted power per square meter decreases and the wavelength of maximum intensity of emitted light increases. C) the emitted power per square meter increases and the wavelength of maximum intensity of emitted light decreases. D) the emitted power per square meter decreases and the wavelength o ...