The Relation Between Classical and Quantum Mechanical Rigid
... 3 - LYmodel of 12C, however; the internal coordinates are concentrated at a definite position, and could be fairly approximated by oscillators. A second example of this correspondence occurs in an m-particle model of 160 [4]. The four a-particles are assumed to lie on two equilateral triangles with ...
... 3 - LYmodel of 12C, however; the internal coordinates are concentrated at a definite position, and could be fairly approximated by oscillators. A second example of this correspondence occurs in an m-particle model of 160 [4]. The four a-particles are assumed to lie on two equilateral triangles with ...
Chapter 3 Stoichiometry
... Stoichiometry: The study of quantities of materials consumed and produced in chemical reactions Atomic Masses: Are determined by comparing with 12C (carbon-12 scale). By definition, carbon-12 is assigned a mass of exactly 12 atomic mass units (amu) and the masses of all other atoms are given relativ ...
... Stoichiometry: The study of quantities of materials consumed and produced in chemical reactions Atomic Masses: Are determined by comparing with 12C (carbon-12 scale). By definition, carbon-12 is assigned a mass of exactly 12 atomic mass units (amu) and the masses of all other atoms are given relativ ...
Spherical Tensors
... Notice the reversal of m, m compared with the usual operation of a matrix on a vector: this is because we are rotating the basis here, rather than rotating relative to the basis. The basic idea is that these irreducible subgroups into which Cartesian tensors apparently decompose under rotation (gen ...
... Notice the reversal of m, m compared with the usual operation of a matrix on a vector: this is because we are rotating the basis here, rather than rotating relative to the basis. The basic idea is that these irreducible subgroups into which Cartesian tensors apparently decompose under rotation (gen ...
Stoichiometry w RICE
... ~change both initial masses to moles ~compare ratio, I need a NaOH : H2SO4 of 2 : 1, so ask: “Do I have 2x as much NaOH as H2SO4?” No, b/c 2(0.612) = 1.22 and I only have 1.00, therefore NaOH is my limiting reactant and H2SO4 is excess. ...
... ~change both initial masses to moles ~compare ratio, I need a NaOH : H2SO4 of 2 : 1, so ask: “Do I have 2x as much NaOH as H2SO4?” No, b/c 2(0.612) = 1.22 and I only have 1.00, therefore NaOH is my limiting reactant and H2SO4 is excess. ...
Rotational spectrum of SO3 and theoretical evidence for the
... TROVE procedure is used, for which the computational details are described extensively elsewhere.46–49 This procedure ...
... TROVE procedure is used, for which the computational details are described extensively elsewhere.46–49 This procedure ...
Calculations with Chemical Formulas and Equations Chapter 3
... Chemistry requires a method for determining the numbers of molecules in a given mass of a substance. This allows the chemist to carry out “recipes” for compounds based on the relative numbers of atoms involved. The calculation involving the quantities of reactants and products in a chemical equation ...
... Chemistry requires a method for determining the numbers of molecules in a given mass of a substance. This allows the chemist to carry out “recipes” for compounds based on the relative numbers of atoms involved. The calculation involving the quantities of reactants and products in a chemical equation ...
Scattering Matrix Formulation of the Total Photoionization of Two
... doubly-excited states could be labelled by approximate quantum numbers that could be understood in terms of group-theoretical quantities [4,5]. Further progress in both experiment and numerical methods made it possible to observe irregular fluctuations in the photoionization spectrum beyond the regi ...
... doubly-excited states could be labelled by approximate quantum numbers that could be understood in terms of group-theoretical quantities [4,5]. Further progress in both experiment and numerical methods made it possible to observe irregular fluctuations in the photoionization spectrum beyond the regi ...
1 The Mole 6.02 X 10 23
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
... • Molecular Mass/Molecular Weight: If you have a single molecule, mass is measured in amu’s instead of grams. But, the molecular mass/weight is the same numerical value as 1 mole of molecules. Only the units are different. (This is the beauty of Avogadro’s Number!) ...
The Vibrational-Rotational Spectrum of HCl
... First, we must recognize that Hydrogen Chloride is a diatomic gaseous substance. Because it is a diatomic molecule, it will have one vibrational mode and one rotational mode of motion. Thus, we can model HCl molecules as vibrating rotors. ...
... First, we must recognize that Hydrogen Chloride is a diatomic gaseous substance. Because it is a diatomic molecule, it will have one vibrational mode and one rotational mode of motion. Thus, we can model HCl molecules as vibrating rotors. ...
Document
... (in amu): One H2O molecule weighs 18.0 amu1 mol of H2O weighs 18.0g. One NO3– ion weighs 62.0 amu 1 mol of NO3– weighs 62.0 g One NaCl unit weighs 58.5 amu 1 mol of NaCl weighs 58.5 g. Interconverting Masses, Moles, and Numbers of Particles Conversion of mass to moles and moles to mass are made ...
... (in amu): One H2O molecule weighs 18.0 amu1 mol of H2O weighs 18.0g. One NO3– ion weighs 62.0 amu 1 mol of NO3– weighs 62.0 g One NaCl unit weighs 58.5 amu 1 mol of NaCl weighs 58.5 g. Interconverting Masses, Moles, and Numbers of Particles Conversion of mass to moles and moles to mass are made ...
O - Free Exam Papers
... • However, the same atoms are present before and after reaction. They have just joined up in different ways. • Because of this the total mass of reactants is always equal to the total mass of products. • This idea is known as the Law of Conservation of Mass. ...
... • However, the same atoms are present before and after reaction. They have just joined up in different ways. • Because of this the total mass of reactants is always equal to the total mass of products. • This idea is known as the Law of Conservation of Mass. ...
Chapter 3 Chemical Reactions and Reaction Stoichiometry
... Click here to view a tutorial on calculating the amount of excess reactant remaining after a reaction is 100% complete. Limiting reactant and theoretical yield calculations are also included. Click here to view the solution to a simple percent-yield problem. The video also reviews the concepts of th ...
... Click here to view a tutorial on calculating the amount of excess reactant remaining after a reaction is 100% complete. Limiting reactant and theoretical yield calculations are also included. Click here to view the solution to a simple percent-yield problem. The video also reviews the concepts of th ...
The Rotation-vibration Hamiltonian
... and i = 1, … , N ) and corresponding velocities that enter the equation for the Hamiltonian (see equation (3.26)). On the other hand, it is well known that a N-body harmonic oscillator has only 3N − 6 degrees of freedom ( 3N − 5 for a linear oscillator), which are the quantities normally used to exp ...
... and i = 1, … , N ) and corresponding velocities that enter the equation for the Hamiltonian (see equation (3.26)). On the other hand, it is well known that a N-body harmonic oscillator has only 3N − 6 degrees of freedom ( 3N − 5 for a linear oscillator), which are the quantities normally used to exp ...
Document
... (in amu): One H2O molecule weighs 18.0 amu1 mol of H2O weighs 18.0g. One NO3– ion weighs 62.0 amu 1 mol of NO3– weighs 62.0 g One NaCl unit weighs 58.5 amu 1 mol of NaCl weighs 58.5 g. Interconverting Masses, Moles, and Numbers of Particles Conversion of mass to moles and moles to mass are made ...
... (in amu): One H2O molecule weighs 18.0 amu1 mol of H2O weighs 18.0g. One NO3– ion weighs 62.0 amu 1 mol of NO3– weighs 62.0 g One NaCl unit weighs 58.5 amu 1 mol of NaCl weighs 58.5 g. Interconverting Masses, Moles, and Numbers of Particles Conversion of mass to moles and moles to mass are made ...
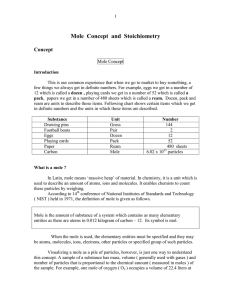
Mole Concept and Stoichiometry
... standard temperature and pressure ( STP = O0 C and 1 atm ), has a mass of 31.998 grams and contains about 6.022 x 1023 molecules of oxygen. Measuring one of these quantities allows the calculation of the others and this is frequently done in stoichiometry. One interpretation : A specific number of p ...
... standard temperature and pressure ( STP = O0 C and 1 atm ), has a mass of 31.998 grams and contains about 6.022 x 1023 molecules of oxygen. Measuring one of these quantities allows the calculation of the others and this is frequently done in stoichiometry. One interpretation : A specific number of p ...
solution
... are developing vehicles that run on hydrogen. These cars are environmentally friendly because their only emission is water vapor. One way to obtain hydrogen for fuel is to use an emission-free energy source such as wind power to split hydrogen from water. What mass of hydrogen (in grams) does 1.00 g ...
... are developing vehicles that run on hydrogen. These cars are environmentally friendly because their only emission is water vapor. One way to obtain hydrogen for fuel is to use an emission-free energy source such as wind power to split hydrogen from water. What mass of hydrogen (in grams) does 1.00 g ...
Binnie Chapter 3
... Steps for Combustion Analysis Problems 1. Calculate the moles of CO2 produced (same as mols of C) 2. Calculate the moles of H2O produced Multiply this number by two to calculate the number of moles of H atoms present in the original compound. ...
... Steps for Combustion Analysis Problems 1. Calculate the moles of CO2 produced (same as mols of C) 2. Calculate the moles of H2O produced Multiply this number by two to calculate the number of moles of H atoms present in the original compound. ...
Lecture 10 Example: Particle in a box
... essentially the same for all energies, but is just “stretched” to larger amplitudes for higher energies. For n = 5, shown in the lower left panel of Figure 4, we see the continued trend that the peaks near x = 0 are smaller than the peaks near the edges. Note that the probability densities continue ...
... essentially the same for all energies, but is just “stretched” to larger amplitudes for higher energies. For n = 5, shown in the lower left panel of Figure 4, we see the continued trend that the peaks near x = 0 are smaller than the peaks near the edges. Note that the probability densities continue ...
Topic 3
... maximum amount of product that can be obtained from given amounts of reactants. The percentage yield is the actual yield (experimentally determined) expressed as a percentage of the theoretical yield (calculated like in ...
... maximum amount of product that can be obtained from given amounts of reactants. The percentage yield is the actual yield (experimentally determined) expressed as a percentage of the theoretical yield (calculated like in ...
Lecture 5
... 2 H2 + O2 → 2 H2O The chemical substances on the left of the equation are the ‘reactants’. The chemical substances on the right of the equation are the ‘products’. The numbers in front of the formulas are the coefficients. ...
... 2 H2 + O2 → 2 H2O The chemical substances on the left of the equation are the ‘reactants’. The chemical substances on the right of the equation are the ‘products’. The numbers in front of the formulas are the coefficients. ...