chapter twenty-one transition metals and coordination chemistry
... See Figure 21.27 for the tetrahedral crystal field diagram. Notice that the orbitals are reverse of that in the octahedral crystal field diagram. The degenerate d z 2 and d x 2 − y 2 are at a lower energy than the degenerate dxy, dxz, and dyz orbitals. Again, the reason for this is that tetrahedral ...
... See Figure 21.27 for the tetrahedral crystal field diagram. Notice that the orbitals are reverse of that in the octahedral crystal field diagram. The degenerate d z 2 and d x 2 − y 2 are at a lower energy than the degenerate dxy, dxz, and dyz orbitals. Again, the reason for this is that tetrahedral ...
TOPIC 7. CHEMICAL CALCULATIONS I
... Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually no neutrons in its nucleus with one electron orbiting outside that nucleus, through to very large atoms such as uranium for example which contains 92 protons and ...
... Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually no neutrons in its nucleus with one electron orbiting outside that nucleus, through to very large atoms such as uranium for example which contains 92 protons and ...
SyllAbuS - Cambridge International Examinations
... affects its physical and chemical properties, and influences how substances react chemically. • Experiments and evidence Chemists use evidence gained from observations and experiments to build models and theories of the structure and reactivity of materials. • Patterns in chemical behaviour and re ...
... affects its physical and chemical properties, and influences how substances react chemically. • Experiments and evidence Chemists use evidence gained from observations and experiments to build models and theories of the structure and reactivity of materials. • Patterns in chemical behaviour and re ...
TOPIC 7. CHEMICAL CALCULATIONS I
... Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually no neutrons in its nucleus with one electron orbiting outside that nucleus, through to very large atoms such as uranium for example which contains 92 protons and ...
... Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually no neutrons in its nucleus with one electron orbiting outside that nucleus, through to very large atoms such as uranium for example which contains 92 protons and ...
unit iv – stoichiometry 1
... * Stoichiometry - the study of quantitative relationships in chemical formulas and equations * Chemical Formulas - a series of symbols and numbers used to represent the composition of an element or a compound I. Formulas for Compounds A. Ionic Compounds - represents ratio of cations to anions * reme ...
... * Stoichiometry - the study of quantitative relationships in chemical formulas and equations * Chemical Formulas - a series of symbols and numbers used to represent the composition of an element or a compound I. Formulas for Compounds A. Ionic Compounds - represents ratio of cations to anions * reme ...
Holt Modern Chemistry Workbook: intro - ch 5
... The goal of basic research is to increase knowledge. In chemistry, basic research includes the study of the properties of a chemical. It also includes the study of what happens when two chemicals are mixed. Sometimes, scientists do basic r esearch simply to satisfy their curiosity about a chemical ...
... The goal of basic research is to increase knowledge. In chemistry, basic research includes the study of the properties of a chemical. It also includes the study of what happens when two chemicals are mixed. Sometimes, scientists do basic r esearch simply to satisfy their curiosity about a chemical ...
ChemConnections
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
... same types of atoms in the same physical state, the more atoms per molecule, the more types of motion available to it and, thus, the higher its entropy. (d) 4 mol S2. The two samples contain the same number of sulfur atoms, but different numbers of molecules. Despite the greater complexity of S8 , t ...
Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the
... tropicity: ions at the beginning of the series are called kosmotropic, ions at the end are chaotropic (cf. text). chemicals. They can easily introduce chirality into a given proAnions that stabilize the native structure of water are called cess, either as a solvent or—much more frequently by reason ...
... tropicity: ions at the beginning of the series are called kosmotropic, ions at the end are chaotropic (cf. text). chemicals. They can easily introduce chirality into a given proAnions that stabilize the native structure of water are called cess, either as a solvent or—much more frequently by reason ...
PC_Chemistry_Macomb_April08
... Potential energy is stored whenever work must be done to change the distance between two objects. The attraction between the two objects may be gravitational, electrostatic, magnetic, or strong force. Chemical potential energy is the result of electrostatic attractions between atoms. Explain the cha ...
... Potential energy is stored whenever work must be done to change the distance between two objects. The attraction between the two objects may be gravitational, electrostatic, magnetic, or strong force. Chemical potential energy is the result of electrostatic attractions between atoms. Explain the cha ...
Part II - American Chemical Society
... b. i. Adding Ba2+ ion will decrease the molar solubility of BaF2 due to the common ion effect. ii. Adding H+ will increase the molar solubility of BaF2 as the F– ion will react with H+ to form HF, thereby causing more BaF2 to dissolve by Le Chatelier’s Principle. c. The calculated Ksp will be too la ...
... b. i. Adding Ba2+ ion will decrease the molar solubility of BaF2 due to the common ion effect. ii. Adding H+ will increase the molar solubility of BaF2 as the F– ion will react with H+ to form HF, thereby causing more BaF2 to dissolve by Le Chatelier’s Principle. c. The calculated Ksp will be too la ...
Organic Chemistry Curriculum Map - Belle Vernon Area School District
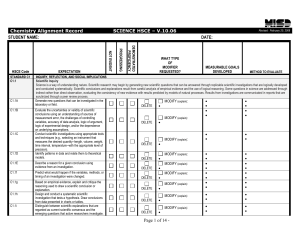
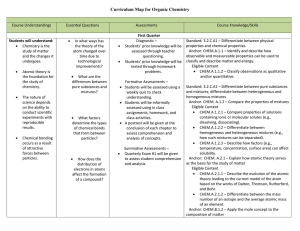
... Standard: 3.2.C.A2 – Predict chemical formulas based on the number of valence electrons. Anchor: CHEM.A.1.1 – Identify and describe how observable and measureable properties can be used to classify and describe matter and energy. Eligible Content CHEM.A.1.1.5 – Apply systematic set of rules (IUPAC ...
... Standard: 3.2.C.A2 – Predict chemical formulas based on the number of valence electrons. Anchor: CHEM.A.1.1 – Identify and describe how observable and measureable properties can be used to classify and describe matter and energy. Eligible Content CHEM.A.1.1.5 – Apply systematic set of rules (IUPAC ...
CB document - mvhs
... All of the reactants and products in the above equation, along with hydrating water molecules, would be considered the “system.” Everything else—nonhydrating water molecules, the beaker, the air, etc.—would be considered the “surroundings.” Most commonly, energy is exchanged between a system and its ...
... All of the reactants and products in the above equation, along with hydrating water molecules, would be considered the “system.” Everything else—nonhydrating water molecules, the beaker, the air, etc.—would be considered the “surroundings.” Most commonly, energy is exchanged between a system and its ...
The Effect of Using Computer Simulations in Teaching Chemical
... findings of Menn (1993) who stated that learners retained 90% of what they had learned using computer simulations. The concretisation of objects, atoms, molecules and bacteria, for example, makes learning meaningful and appealing to real life situations. Learners can test theories by developing alte ...
... findings of Menn (1993) who stated that learners retained 90% of what they had learned using computer simulations. The concretisation of objects, atoms, molecules and bacteria, for example, makes learning meaningful and appealing to real life situations. Learners can test theories by developing alte ...
sample
... different gases. Based on these measurements, he concluded that “the pressure of a fixed amount of gas, measured at constant volume, is directly proportional to its absolute temperature.” This statement is best described as a A) theory. B) hypothesis. C) law. D) experiment. E) definition. Ans: C Dif ...
... different gases. Based on these measurements, he concluded that “the pressure of a fixed amount of gas, measured at constant volume, is directly proportional to its absolute temperature.” This statement is best described as a A) theory. B) hypothesis. C) law. D) experiment. E) definition. Ans: C Dif ...
Concept Development Studies in Chemistry
... development studies will enhance your development of critical, analytical thinking, a skill which is most important to success in Science. As a note, these studies are not intended to be historical developments, although the experiments presented are the ones which led to the concepts discussed. Onl ...
... development studies will enhance your development of critical, analytical thinking, a skill which is most important to success in Science. As a note, these studies are not intended to be historical developments, although the experiments presented are the ones which led to the concepts discussed. Onl ...
PART 6-ICHO-26-30
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
2003 AP Chemistry Form B Scoring Guidelines - AP Central
... (d) Of the compounds NaOH , CuS , and NaNO3 , which one is appropriate to use in a salt bridge? Briefly explain your answer, and for each of the other compounds, include a reason why it is not appropriate. NaOH is not appropriate. The anion, OH−, would migrate towards the anode. The OH− would react ...
... (d) Of the compounds NaOH , CuS , and NaNO3 , which one is appropriate to use in a salt bridge? Briefly explain your answer, and for each of the other compounds, include a reason why it is not appropriate. NaOH is not appropriate. The anion, OH−, would migrate towards the anode. The OH− would react ...
Hydrolases as Catalysts for Green Chemistry and
... synthesis of a chiral pharmaceutical compound, S-clopidogrel, by selective hydrolysis of the racemic precursor. Current production of the pure S- clopidogrel isomer involves the use of a resolving agent, L-camphorsulfonic acid, and organic solvents. Screening of different hydrolases revealed that cr ...
... synthesis of a chiral pharmaceutical compound, S-clopidogrel, by selective hydrolysis of the racemic precursor. Current production of the pure S- clopidogrel isomer involves the use of a resolving agent, L-camphorsulfonic acid, and organic solvents. Screening of different hydrolases revealed that cr ...
Chapter 1 Chirality in clinical analysis 1.1. Introduction
... The term “Chirality” (from the Greek kheir for hand), means handedness, which is the existence of left/right opposites. The concept of “Chirality” has been known in chemistry since the 1870’s although a hundred years passed before chemists began using this term. In 1962, the first edition of Eliel’s ...
... The term “Chirality” (from the Greek kheir for hand), means handedness, which is the existence of left/right opposites. The concept of “Chirality” has been known in chemistry since the 1870’s although a hundred years passed before chemists began using this term. In 1962, the first edition of Eliel’s ...
Mass Relationships in Chemical Reactions
... To convert to whole numbers we divide each mole by the smaller subscript (2.174). After rounding off, we obtain NO2 as the empirical formula. The Molecular formula must have a Molar Mass that is a multiple of the Empirical formula’s Molar Mass (NO2, N2O4, N3O6, N4O8…) ...
... To convert to whole numbers we divide each mole by the smaller subscript (2.174). After rounding off, we obtain NO2 as the empirical formula. The Molecular formula must have a Molar Mass that is a multiple of the Empirical formula’s Molar Mass (NO2, N2O4, N3O6, N4O8…) ...
Part 2-ICHO-26-30
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
chemical bonding i: basic concepts
... atoms and molecules. Yet the shape of a molecule—that is, the arrangement of its atoms in space—often defines its chemistry. If water had a different shape, its properties would be significantly different, and life as we know it would not be possible. In this chapter, we will describe the interactio ...
... atoms and molecules. Yet the shape of a molecule—that is, the arrangement of its atoms in space—often defines its chemistry. If water had a different shape, its properties would be significantly different, and life as we know it would not be possible. In this chapter, we will describe the interactio ...