Document
... All matter is made of atoms. Atoms are the smallest form of elements. About 100 elements • Hydrogen is an element that accounts for about 90% of total mass of the universe. ...
... All matter is made of atoms. Atoms are the smallest form of elements. About 100 elements • Hydrogen is an element that accounts for about 90% of total mass of the universe. ...
AP CHEMISTRY SUMMER ASSIGNMENT
... a. SiO2 _______________________________ b. N2O4 _______________________________ c. PO3 _______________________________ d. SCl6 _______________________________ e. CO _______________________________ Solution: a. silicon dioxide, ...
... a. SiO2 _______________________________ b. N2O4 _______________________________ c. PO3 _______________________________ d. SCl6 _______________________________ e. CO _______________________________ Solution: a. silicon dioxide, ...
Document
... Gas 3. Elements Basic substances that make up matter Cannot be broken down by ordinary chemical means http://library.thinkquest.org/3616/chem/Periodic. htm 4. Common Elements in the Human Body 5. Other Elements Lesser elements Iodine (I) Iron (Fe) Trace elements Often part of enzymes or required for ...
... Gas 3. Elements Basic substances that make up matter Cannot be broken down by ordinary chemical means http://library.thinkquest.org/3616/chem/Periodic. htm 4. Common Elements in the Human Body 5. Other Elements Lesser elements Iodine (I) Iron (Fe) Trace elements Often part of enzymes or required for ...
AHSGE Review
... Families are arranged because of similar properties. Each element in a group will react and appear much the same. ...
... Families are arranged because of similar properties. Each element in a group will react and appear much the same. ...
Solutions - Seattle Central
... No Tests Today: Nucleic acids (DNA and RNA) made of nucleotides • 5 carbon sugar (Deoxyribose – DNA, ribose – RNA) ...
... No Tests Today: Nucleic acids (DNA and RNA) made of nucleotides • 5 carbon sugar (Deoxyribose – DNA, ribose – RNA) ...
Properties of Metals vs. Nonmetals vs. Metalloids
... Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share electrons. Identify the following pairs of atoms as potentially forming an ionic or molecular compound: Mg and C ...
... Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share electrons. Identify the following pairs of atoms as potentially forming an ionic or molecular compound: Mg and C ...
Properties of Metals vs. Nonmetals vs. Metalloids
... Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share electrons. Identify the following pairs of atoms as potentially forming an ionic or molecular compound: Mg and C ...
... Metals lose electrons and form _____________ while nonmetals gain and electrons form __________. Molecular compounds form when a ______________ and a _______________ combine as they share electrons. Identify the following pairs of atoms as potentially forming an ionic or molecular compound: Mg and C ...
Chemistry Mid-Term Review: 2015-2016
... 11. Classify the following compounds as ionic or covalent. a. MgCl2 b. Na2S c. H2O d. H2S 12. How many electrons do two atoms in a double covalent bond share? How many in a triple covalent bond? 13. Characterize a coordinate covalent bond and give an example. 14. Using electron dot structure, draw a ...
... 11. Classify the following compounds as ionic or covalent. a. MgCl2 b. Na2S c. H2O d. H2S 12. How many electrons do two atoms in a double covalent bond share? How many in a triple covalent bond? 13. Characterize a coordinate covalent bond and give an example. 14. Using electron dot structure, draw a ...
PART 2 – CHEMISTRY
... (negative ion), i.e. BaBr2 is named barium bromide. Cations formed from metal atoms have the same name as the metal, i.e. Na + is named “sodium ion”. If the metal can form cations of differing charges, the charge is given using Roman numerals, i.e. Cu+ is named “copper (I) ion”. Cu2+ is named “coppe ...
... (negative ion), i.e. BaBr2 is named barium bromide. Cations formed from metal atoms have the same name as the metal, i.e. Na + is named “sodium ion”. If the metal can form cations of differing charges, the charge is given using Roman numerals, i.e. Cu+ is named “copper (I) ion”. Cu2+ is named “coppe ...
Unit 1. Materials: Formulating Matter A. How do chemists describe
... A. How do chemists describe matter? The U.S. Mint is promoting the use of dollar coins instead of dollar bills. In what ways might coins be superior to bills? How do the properties of each form of currency depend upon their composition? In this unit, you will consider the benefits and drawbacks of u ...
... A. How do chemists describe matter? The U.S. Mint is promoting the use of dollar coins instead of dollar bills. In what ways might coins be superior to bills? How do the properties of each form of currency depend upon their composition? In this unit, you will consider the benefits and drawbacks of u ...
2015-2016 AP CHEMISTRY MIDTERM EXAM Review
... 4. Answer all three portions in this part. Give the formulas to show the reactants and the products for the three following chemical reactions. Each reaction occurs in aqueous solution unless otherwise indicated. Represent the substances in solution as ions if the substance is extensively ionized. ...
... 4. Answer all three portions in this part. Give the formulas to show the reactants and the products for the three following chemical reactions. Each reaction occurs in aqueous solution unless otherwise indicated. Represent the substances in solution as ions if the substance is extensively ionized. ...
October 23, 2013
... Remember that a chemical formula tells us the mole ratio between the elements in a compound. For example, there are two moles of hydrogen for every one mole of oxygen in water. ...
... Remember that a chemical formula tells us the mole ratio between the elements in a compound. For example, there are two moles of hydrogen for every one mole of oxygen in water. ...
Chapter 6 Chemical Reactions: An Introduction
... – Burning in air means reacting with O2 – Metals are solids, except for Hg, which is liquid. ...
... – Burning in air means reacting with O2 – Metals are solids, except for Hg, which is liquid. ...
chem – mixtures elements compounds for ib 1 10-10
... There are 90 naturally occurring elements Element symbols come from the Latin names for elements Examples: Carbon (C), Iron (Fe) Forging Elements The Elements Song ...
... There are 90 naturally occurring elements Element symbols come from the Latin names for elements Examples: Carbon (C), Iron (Fe) Forging Elements The Elements Song ...
Balancing Equations
... Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in order t ...
... Find the number of atoms for each element on the left side. Compare those against the number of the atoms of the same element on the right side. Determine where to place coefficients in front of formulas so that the left side has the same number of atoms as the right side for EACH element in order t ...
Midterm Review Sample Content Questions
... 22. Identify the following reactions as alpha, beta, and/or gamma radiation. A. B. ...
... 22. Identify the following reactions as alpha, beta, and/or gamma radiation. A. B. ...
QUIZ: History of Atomic Structure
... B) deflection of cathode rays by electric and magnetic fields C) The oil drop experiment D) the periodic table E) the mass spectrometer 5. Which of the following is NOT part of Dalton's atomic theory? A) Atoms are the smallest, indivisible particles in nature. B) Chemical reactions are simple rearra ...
... B) deflection of cathode rays by electric and magnetic fields C) The oil drop experiment D) the periodic table E) the mass spectrometer 5. Which of the following is NOT part of Dalton's atomic theory? A) Atoms are the smallest, indivisible particles in nature. B) Chemical reactions are simple rearra ...
Atomic combinations: Electronegativity and ionic
... Another example of ionic bonding takes place between magnesium (Mg) and oxygen (O) to form magnesium oxide (MgO). Magnesium has two valence electrons and an electronegativity of 1.2, while oxygen has six valence electrons and an electronegativity of 3.5. Since oxygen has a higher electronegativity, ...
... Another example of ionic bonding takes place between magnesium (Mg) and oxygen (O) to form magnesium oxide (MgO). Magnesium has two valence electrons and an electronegativity of 1.2, while oxygen has six valence electrons and an electronegativity of 3.5. Since oxygen has a higher electronegativity, ...
Definitions - Loreto Science
... The first ionisation energy of an atom • is the minimum energy required to completely remove the most loosely bound electron from a neutral gaseous atom in the ground state.****** ...
... The first ionisation energy of an atom • is the minimum energy required to completely remove the most loosely bound electron from a neutral gaseous atom in the ground state.****** ...
The Chemical Context of Life Chapter 2 Notes
... The different states of potential energy that electrons have in an atom are called energy levels or electron shells. - the first shell has the lowest energy. The second shell has more than the first, etc. Valence electrons: those in the outermost shell ...
... The different states of potential energy that electrons have in an atom are called energy levels or electron shells. - the first shell has the lowest energy. The second shell has more than the first, etc. Valence electrons: those in the outermost shell ...
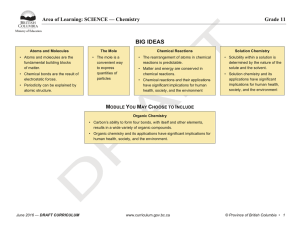
BIG IDEAS - BC Curriculum - Province of British Columbia
... • Carbon’s ability to form four bonds, with itself and other elements, results in a wide variety of organic compounds. • Organic chemistry and its applications have significant implications for human health, society, and the environment. ...
... • Carbon’s ability to form four bonds, with itself and other elements, results in a wide variety of organic compounds. • Organic chemistry and its applications have significant implications for human health, society, and the environment. ...
Unit 2 Review Questions Fill in the blank In a(n) change, a new
... The mass number is the sum of electrons and protons in the atom. l. A Bohr diagram shows electrons in orbits about the nucleus. m. A row of the periodic table is called a period. n. The size of atoms increase down a column of the periodic table. o. Alkali metals include fluorine, chlorine, and iodin ...
... The mass number is the sum of electrons and protons in the atom. l. A Bohr diagram shows electrons in orbits about the nucleus. m. A row of the periodic table is called a period. n. The size of atoms increase down a column of the periodic table. o. Alkali metals include fluorine, chlorine, and iodin ...
SCI 3101 Test IV MULTIPLE CHOICE. 1) The sky is blue because air
... 15) Why are the atomic masses listed in the periodic table not whole numbers? A) The atomic masses are average atomic masses. B) Scientists have yet to make the precise measurements. C) That would be too much of a coincidence. D) Today's instruments are able to measure the atomic masses to many deci ...
... 15) Why are the atomic masses listed in the periodic table not whole numbers? A) The atomic masses are average atomic masses. B) Scientists have yet to make the precise measurements. C) That would be too much of a coincidence. D) Today's instruments are able to measure the atomic masses to many deci ...
How to balance chemical equations File
... This equation shows you what happens in the reaction, but it doesn’t show you how much of each element you need to produce the ammonia. To find out how much of each element you need, you have to balance the equation — make sure that the number of atoms on the left side of the equation equals the num ...
... This equation shows you what happens in the reaction, but it doesn’t show you how much of each element you need to produce the ammonia. To find out how much of each element you need, you have to balance the equation — make sure that the number of atoms on the left side of the equation equals the num ...