Optimizing OFETs properties for spintronics applications
... pure. Ces faibles valeurs de résistance de contact, ainsi que les résultats de la caractérisation des propriétés physico-chimiques, démontrent clairement que notre couche de carbone ultra-mince agit efficacement comme barrière imperméable contre l'oxydation de la surface de Ni, améliorant la capacit ...
... pure. Ces faibles valeurs de résistance de contact, ainsi que les résultats de la caractérisation des propriétés physico-chimiques, démontrent clairement que notre couche de carbone ultra-mince agit efficacement comme barrière imperméable contre l'oxydation de la surface de Ni, améliorant la capacit ...
DC Biasing using a S..
... If we ASSUME that the BJT is in active mode, then we ENFORCE the proper equalities and ANALYZE this circuit to find collector current IC: ...
... If we ASSUME that the BJT is in active mode, then we ENFORCE the proper equalities and ANALYZE this circuit to find collector current IC: ...
6 theoretical problems 2 practical problems
... 3.2 Write molecular formulas for all possible ionic compounds which comply with the following conditions: each compound has 1) empirical formula Pt(NH3)2Cl2, 2) an anion and a cation and is composed of discrete, monomeric square planar platinum(II) complex, 3) only one type of cation and one type of ...
... 3.2 Write molecular formulas for all possible ionic compounds which comply with the following conditions: each compound has 1) empirical formula Pt(NH3)2Cl2, 2) an anion and a cation and is composed of discrete, monomeric square planar platinum(II) complex, 3) only one type of cation and one type of ...
LTC1732-4
... an LED. When the battery current drops to 10% of the fullscale current (C/10), the N-channel MOSFET is turned off and a weak 35µA current source to ground is connected to the CHRG pin. A 15ms time delay is included to help prevent false triggering due to transient currents. The end-of-charge compara ...
... an LED. When the battery current drops to 10% of the fullscale current (C/10), the N-channel MOSFET is turned off and a weak 35µA current source to ground is connected to the CHRG pin. A 15ms time delay is included to help prevent false triggering due to transient currents. The end-of-charge compara ...
Exercise 5
... change in v leads to an exponential change in i, it is much better to use the setup with the variable current source. However, variable voltage sources are much more common in a lab than variable current sources. To convert a variable voltage source into a ...
... change in v leads to an exponential change in i, it is much better to use the setup with the variable current source. However, variable voltage sources are much more common in a lab than variable current sources. To convert a variable voltage source into a ...
Exercise 6: LEDs and MatLab
... change in v leads to an exponential change in i, it is much better to use the setup with the variable current source. However, variable voltage sources are much more common in a lab than variable current sources. To convert a variable voltage source into a ...
... change in v leads to an exponential change in i, it is much better to use the setup with the variable current source. However, variable voltage sources are much more common in a lab than variable current sources. To convert a variable voltage source into a ...
BSP75N 60V self-protected low-side Intellifet MOSFET switch Summary
... 60V self-protected low-side IntellifetTM MOSFET switch Summary Continuous drain source voltage ...
... 60V self-protected low-side IntellifetTM MOSFET switch Summary Continuous drain source voltage ...
Use the following answers for questions 1
... A 72. How many moles of solid Ba(NO3)2 should be added to 300. milliliters of 0.20molar Fe(NO3)3 to increase the concentration of the NO3¯ ion to 1.0-molar? (Assume that the volume of the solution remains constant.) (A) 0.060 mole (B) 0.12 mole (C) 0.24 mole (D) 0.30 mole (E) 0.40 mole E 28. Given t ...
... A 72. How many moles of solid Ba(NO3)2 should be added to 300. milliliters of 0.20molar Fe(NO3)3 to increase the concentration of the NO3¯ ion to 1.0-molar? (Assume that the volume of the solution remains constant.) (A) 0.060 mole (B) 0.12 mole (C) 0.24 mole (D) 0.30 mole (E) 0.40 mole E 28. Given t ...
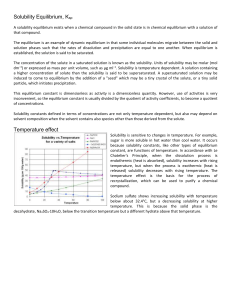
ksp - lozon.ca
... The solubility of a sparingly soluble salt is reduced in a solution that contains an ion in common with that salt. For instance, the solubility of silver chloride in water is reduced if a solution of sodium chloride is added to a suspension of silver chloride in water. A practical example used very ...
... The solubility of a sparingly soluble salt is reduced in a solution that contains an ion in common with that salt. For instance, the solubility of silver chloride in water is reduced if a solution of sodium chloride is added to a suspension of silver chloride in water. A practical example used very ...
Mnemonic Devices - Free WonderKids-e
... Ini this method, the oxidation and reduction processes are divided into separate equations called half-reactions that are balanced separately. Example: Balance the net ionic equation for the reaction of iron (III) chloride with tin (II) chloride. Solution: ...
... Ini this method, the oxidation and reduction processes are divided into separate equations called half-reactions that are balanced separately. Example: Balance the net ionic equation for the reaction of iron (III) chloride with tin (II) chloride. Solution: ...
Electrical Principals (3 exam questions)
... G5B - The Decibel; current and voltage dividers; electrical power calculations; sine wave rootmean-square (RMS) values; PEP calculations G5C – Resistors, capacitors and inductors in series and parallel; transformers ...
... G5B - The Decibel; current and voltage dividers; electrical power calculations; sine wave rootmean-square (RMS) values; PEP calculations G5C – Resistors, capacitors and inductors in series and parallel; transformers ...
General Chemistry - Bioinorganic and Solution Chemistry Group
... Solids and liquids are only a problem when you ingest them. This may happen unconsciously, e.g. by contaminated hands. There is currently a strong impulse to wear gloves even during the most harmless operations in the laboratory. Unfortunately in most cases this is not a solution but rather a reloca ...
... Solids and liquids are only a problem when you ingest them. This may happen unconsciously, e.g. by contaminated hands. There is currently a strong impulse to wear gloves even during the most harmless operations in the laboratory. Unfortunately in most cases this is not a solution but rather a reloca ...
APV25S0 - Calibration Circuit
... The function of the calibration edge generators is to transform the trigger issued by the calibration processor into a voltage step of programmable amplitude. Since the output of the each edge generator is AC coupled to the input of the analog channels, the voltage step ∆V causes a charge injection ...
... The function of the calibration edge generators is to transform the trigger issued by the calibration processor into a voltage step of programmable amplitude. Since the output of the each edge generator is AC coupled to the input of the analog channels, the voltage step ∆V causes a charge injection ...
Nanofluidic circuitry
Nanofluidic circuitry is a nanotechnology aiming for control of fluids in nanometer scale. Due to the effect of an electrical double layer within the fluid channel, the behavior of nanofluid is observed to be significantly different compared with its microfluidic counterparts. Its typical characteristic dimensions fall within the range of 1–100 nm. At least one dimension of the structure is in nanoscopic scale. Phenomena of fluids in nano-scale structure are discovered to be of different properties in electrochemistry and fluid dynamics.