Increased Functional Half-life of Fibroblast Growth Factor
... Fibroblast growth factors (FGFs) are a family of polypeptide exhibiting diverse biological roles in multiple developmental and metabolic processes. The 22 members of the human FGF family have a conserved ~120 amino acid core, however, they exhibit significant difference in size (17-25kDa) and sequen ...
... Fibroblast growth factors (FGFs) are a family of polypeptide exhibiting diverse biological roles in multiple developmental and metabolic processes. The 22 members of the human FGF family have a conserved ~120 amino acid core, however, they exhibit significant difference in size (17-25kDa) and sequen ...
Potential Role of Sulfur-Containing Antioxidant Systems in Highly
... cytosol, mitochondria and endoplasmic reticulum of eukaryotic cells; however most of the GSH (80%–85%) is present in the cytosol [42]. The GSH system is one of the major thiol-dependant antioxidant systems in mammalian cells while GSH specifically is the most abundant non-protein thiol in mammalian ...
... cytosol, mitochondria and endoplasmic reticulum of eukaryotic cells; however most of the GSH (80%–85%) is present in the cytosol [42]. The GSH system is one of the major thiol-dependant antioxidant systems in mammalian cells while GSH specifically is the most abundant non-protein thiol in mammalian ...
Chapter 3 Carbon and the Molecular Diversity of Life The element
... A molecule with the chemical formula C6H12O6 is probably a A) carbohydrate. B) lipid. C) monosaccharide D) carbohydrate and lipid only. E) carbohydrate and monosaccharide only. Answer: E Topic: Concept 3.3 Why are hydrocarbons insoluble in water? A) The majority of their bonds are polar covalent ca ...
... A molecule with the chemical formula C6H12O6 is probably a A) carbohydrate. B) lipid. C) monosaccharide D) carbohydrate and lipid only. E) carbohydrate and monosaccharide only. Answer: E Topic: Concept 3.3 Why are hydrocarbons insoluble in water? A) The majority of their bonds are polar covalent ca ...
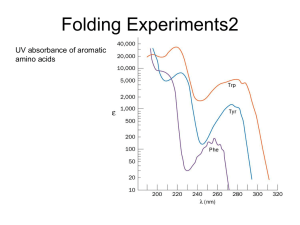
6. Protein Folding
... • When refolding occurs, you can get the proline‐peptide bond in the wrong form. From a kinetic standpoint, cis‐trans proline isomerization is a very slow process that can prevent the progress of protein folding by trapping one or more proline residues crucial for folding in the non‐native isomer, e ...
... • When refolding occurs, you can get the proline‐peptide bond in the wrong form. From a kinetic standpoint, cis‐trans proline isomerization is a very slow process that can prevent the progress of protein folding by trapping one or more proline residues crucial for folding in the non‐native isomer, e ...
Chapter 10
... Due to this increased reactivity LAH will not work in alcoholic or aqueous solvents ...
... Due to this increased reactivity LAH will not work in alcoholic or aqueous solvents ...
Model Description Sheet
... RP: Zcorp with plaster Description: An estimated 900,000 people a year develop venous blood clots in the United States and 60,000 to 100,000 die as a result. Anticoagulants are used to prevent and treat thrombosis. New oral anticoagulants (NOAC’s), like apixaban (Eliquis) are designed to specificall ...
... RP: Zcorp with plaster Description: An estimated 900,000 people a year develop venous blood clots in the United States and 60,000 to 100,000 die as a result. Anticoagulants are used to prevent and treat thrombosis. New oral anticoagulants (NOAC’s), like apixaban (Eliquis) are designed to specificall ...
Click Here to download this tutorial as a PDF
... identify which two atoms are connected by the strut. Then select only those two atoms using the select atomno= command. Then use the connect strut command to add the strut. Finally, use the strut 1.0 command to add a strut or the connect strut delete command to remove the bond between the two select ...
... identify which two atoms are connected by the strut. Then select only those two atoms using the select atomno= command. Then use the connect strut command to add the strut. Finally, use the strut 1.0 command to add a strut or the connect strut delete command to remove the bond between the two select ...
Bonds and Structural Supports - MSOE Center for BioMolecular
... If the first amino acid connected by the triangle bond is residue number N, then the second amino acid connected by the hydrogen bond is residue number N + 2. So to remove a triangle bond, you will need to select two amino acids that have just one amino acid in between them. To remove a triangle bon ...
... If the first amino acid connected by the triangle bond is residue number N, then the second amino acid connected by the hydrogen bond is residue number N + 2. So to remove a triangle bond, you will need to select two amino acids that have just one amino acid in between them. To remove a triangle bon ...
1 keratin, fibrous structural protein of hair, nails, horn, hoofs, wool
... into coils called alpha helices and contain many disulfide bonds (bonds between pairs of sulfur ions). Disulphide bonds are particularly stable and can resist the action of proteolytic enzymes, which specialize in breaking apart proteins. Keratin is also insoluble in water. When human hair is straig ...
... into coils called alpha helices and contain many disulfide bonds (bonds between pairs of sulfur ions). Disulphide bonds are particularly stable and can resist the action of proteolytic enzymes, which specialize in breaking apart proteins. Keratin is also insoluble in water. When human hair is straig ...
Protein Sulfenylation in Mitochondria: Biochemistry and
... therefore remain reduced or whether their redox state is controlled by reducing enzymes such as thioredoxins or glutaredoxins. Thiol oxidation in the matrix of mitochondria In inner membrane of mitochondria separates the glutathione pool of the cytosol/IMS from that of the matrix (Kojer et al., 201 ...
... therefore remain reduced or whether their redox state is controlled by reducing enzymes such as thioredoxins or glutaredoxins. Thiol oxidation in the matrix of mitochondria In inner membrane of mitochondria separates the glutathione pool of the cytosol/IMS from that of the matrix (Kojer et al., 201 ...
Protein core - Acsu.buffalo.edu
... Thermal stability influences the average lifespan of a protein molecules since proteolysis likely occurs while the protein is in an unfolded state ...
... Thermal stability influences the average lifespan of a protein molecules since proteolysis likely occurs while the protein is in an unfolded state ...
Seminar 7 – Theoretical part is in the exercise 4. Seminar 7
... 11. For amino acids with neutral R groups, at any pH below the pI of the amino acid, the population of amino acids in solution will have: A. a net negative charge. B. a net positive charge. C. no charged groups. D. no net charge. E. positive and negative charges in equal concentration. ...
... 11. For amino acids with neutral R groups, at any pH below the pI of the amino acid, the population of amino acids in solution will have: A. a net negative charge. B. a net positive charge. C. no charged groups. D. no net charge. E. positive and negative charges in equal concentration. ...
Redox rescues virus from eR trap - Institute of Molecular Life Sciences
... exceedingly simple, yet its mechanisms hold many secrets. Upon engulfment of a virus by the host cell, uncoating of the viral genome from the protective capsid is required for the activation of viral genes, using the transcriptional machinery of the host cell to drive synthesis of viral progeny. New ...
... exceedingly simple, yet its mechanisms hold many secrets. Upon engulfment of a virus by the host cell, uncoating of the viral genome from the protective capsid is required for the activation of viral genes, using the transcriptional machinery of the host cell to drive synthesis of viral progeny. New ...
Chapter 4 Problem Set
... connections between them. A motif can be an elaborate structure involving multiple protein segments folded together, such as a large ß barrel. A domain is a part of a polypeptide chain that is independently stable or could undergo movements as a single entity with respect to the entire protein. Obvi ...
... connections between them. A motif can be an elaborate structure involving multiple protein segments folded together, such as a large ß barrel. A domain is a part of a polypeptide chain that is independently stable or could undergo movements as a single entity with respect to the entire protein. Obvi ...
Complex Protein Structure
... broken and rearrangement occurs) B) denatured proteins have a different chemistry (raw versus cooked egg) C) denatured proteins may lead to sickness or death (loss of enzyme function) D) denatured proteins cannot usually revert to their original state ...
... broken and rearrangement occurs) B) denatured proteins have a different chemistry (raw versus cooked egg) C) denatured proteins may lead to sickness or death (loss of enzyme function) D) denatured proteins cannot usually revert to their original state ...
Powerpoint slides
... involves the relative movement of its two lobes to each other in a cooperative manner ...
... involves the relative movement of its two lobes to each other in a cooperative manner ...
1. Inter-chain disulfide bonds
... It is the movement of charged molecules in an electrical field towards the oppositely charged electrode. It is used for separation of proteins for dignosis of some diseases such as immunity disease , genetic diseases ( such as Hb S and Hb C diseases. The rate of migration of charged molecules depend ...
... It is the movement of charged molecules in an electrical field towards the oppositely charged electrode. It is used for separation of proteins for dignosis of some diseases such as immunity disease , genetic diseases ( such as Hb S and Hb C diseases. The rate of migration of charged molecules depend ...
presentation source
... Inter-chain interactions • Quanternary structure of proteins – interaction between 2 or more polypeptides – Disulfide bridges • cysteine -S-S- cysteine • --CH2-S-S-CH2-• see Fig. 5-22 ...
... Inter-chain interactions • Quanternary structure of proteins – interaction between 2 or more polypeptides – Disulfide bridges • cysteine -S-S- cysteine • --CH2-S-S-CH2-• see Fig. 5-22 ...
Organic Notes for Chapter 4 and 5
... Highly polar; therefore water soluble Acts as a weak acid (releases H+) Found in amino acids and fatty acids ...
... Highly polar; therefore water soluble Acts as a weak acid (releases H+) Found in amino acids and fatty acids ...
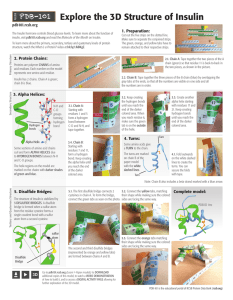
Explore the 3D Structure of Insulin
... residues 7 and 11, form a hydrogen bond. Keep creating the alpha helix until you reach the end of the darker ...
... residues 7 and 11, form a hydrogen bond. Keep creating the alpha helix until you reach the end of the darker ...
Imunoglobulins
... are linked together by disulfide bond. In addition, the two heavy chains are held together by inter-chain disulfide bonds and by non-covalent interactions. • 2. Intra-chain disulfide bonds - Within each of the polypeptide chains (within H and L chains) ...
... are linked together by disulfide bond. In addition, the two heavy chains are held together by inter-chain disulfide bonds and by non-covalent interactions. • 2. Intra-chain disulfide bonds - Within each of the polypeptide chains (within H and L chains) ...
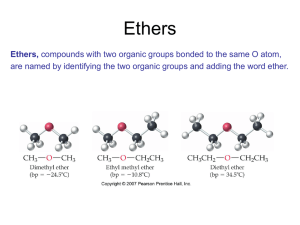
Ethers
... primarily as chlorofluorocarbon (CFC) substitutes, as the ozone depleting effects are only about 10% of the CFCs. Hydrofluoro compounds (HFCs) : They contain no chlorine. Lower global warming potential than the HCFC’s because of no known effects on the ...
... primarily as chlorofluorocarbon (CFC) substitutes, as the ozone depleting effects are only about 10% of the CFCs. Hydrofluoro compounds (HFCs) : They contain no chlorine. Lower global warming potential than the HCFC’s because of no known effects on the ...
Amino Acids 2
... 2. Gel-filtration chromatography separates a mixture of proteins on the basis of: A) size B) charge C) affinity for ligands in the column matrix D) density 3. What is the purpose of treating a protein with 2-mercaptoethanol? A) To hydrolyze the protein into its amino acids. B) To derivatize any free ...
... 2. Gel-filtration chromatography separates a mixture of proteins on the basis of: A) size B) charge C) affinity for ligands in the column matrix D) density 3. What is the purpose of treating a protein with 2-mercaptoethanol? A) To hydrolyze the protein into its amino acids. B) To derivatize any free ...
6th semester-2006 Project Proposal
... disulfide bridge. This results in the generation of highly reactive charged cysteine residue which immediately cross-link to free sulfhydryl groups. The method is currently used to generate nano-arrays of technologically relevant proteins such as immunoglobulins, for medical diagnosis. The main draw ...
... disulfide bridge. This results in the generation of highly reactive charged cysteine residue which immediately cross-link to free sulfhydryl groups. The method is currently used to generate nano-arrays of technologically relevant proteins such as immunoglobulins, for medical diagnosis. The main draw ...
Disulfide

In chemistry and biology a disulfide refers to a functional group with the general structure R–S–S–R. The linkage is also called an SS-bond or a disulfide bridge and is usually derived by the coupling of two thiol groups. In formal terms, the connection is a persulfide, in analogy to its congener, peroxide (R–O–O–R), but this terminology is rarely used, except in reference to hydrodisulfides (R–S–S–H or H–S–S–H compounds).In inorganic chemistry disulfide usually refers to the corresponding anion S22−, or −S–S−, for example in disulfur dichloride.