File
... Assume you have 100 g of the compound. Change “%” to “g” Convert grams to moles for each element Divide each mole amount by the smallest mole amount, these numbers are the coefficients for the E.F. If the numbers from step 4 are not all whole numbers, multiply the step 4 numbers by a whole number so ...
... Assume you have 100 g of the compound. Change “%” to “g” Convert grams to moles for each element Divide each mole amount by the smallest mole amount, these numbers are the coefficients for the E.F. If the numbers from step 4 are not all whole numbers, multiply the step 4 numbers by a whole number so ...
Chemistry 201 - Department of Chemistry | Oregon State University
... Instructions: You should have with you several number two pencils, an eraser, your 3" x 5" note card, a calculator, and your University ID Card. If you have notes with you, place them in a sealed backpack and place the backpack OUT OF SIGHT or place the notes directly on the table at the front of th ...
... Instructions: You should have with you several number two pencils, an eraser, your 3" x 5" note card, a calculator, and your University ID Card. If you have notes with you, place them in a sealed backpack and place the backpack OUT OF SIGHT or place the notes directly on the table at the front of th ...
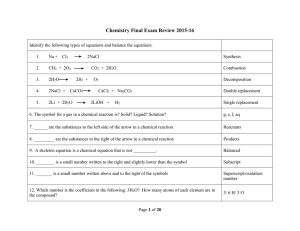
xmas review questions 01516 with hints
... 30. A 27.0–gram sample of an unknown hydrocarbon was burned in excess oxygen to form 88.0 grams of carbon dioxide and 27.0 grams of water. What is a possible molecular formula of the hydrocarbon? (A) CH4 (C) C4H3 (E) C4H10 (B) C2H2 (D) C4H6 Did you see the significance of the 88 g CO2 and 27 g H2O? ...
... 30. A 27.0–gram sample of an unknown hydrocarbon was burned in excess oxygen to form 88.0 grams of carbon dioxide and 27.0 grams of water. What is a possible molecular formula of the hydrocarbon? (A) CH4 (C) C4H3 (E) C4H10 (B) C2H2 (D) C4H6 Did you see the significance of the 88 g CO2 and 27 g H2O? ...
A = 27
... #33 The excited state must have the same # of electrons as the neutral atom, however one or more must be at a higher energy level (outermost shell) that the ground state of the periodic table ( for Al it is 2-8-3), 13 electrons.The ans is 1) 2-7-4 (13 e-), one electron promoted from shell 2 to shell ...
... #33 The excited state must have the same # of electrons as the neutral atom, however one or more must be at a higher energy level (outermost shell) that the ground state of the periodic table ( for Al it is 2-8-3), 13 electrons.The ans is 1) 2-7-4 (13 e-), one electron promoted from shell 2 to shell ...
Slide 1
... Gram formula mass • Gram formula mass (gfm) (a.k.a. “molecular mass” or “formula mass”) • the sum total mass using grams (g) instead of a.m.u. • (gfm) always equals 1 mole (mol) of a compound **(gfm = 1 mole) *** Q.1. How many molecules are in 1 mole? ...
... Gram formula mass • Gram formula mass (gfm) (a.k.a. “molecular mass” or “formula mass”) • the sum total mass using grams (g) instead of a.m.u. • (gfm) always equals 1 mole (mol) of a compound **(gfm = 1 mole) *** Q.1. How many molecules are in 1 mole? ...
Percent Composition
... pure the sample was that was used in the reaction. • We can also use the known formula to compare percent composition values to see if the experiment was done with accuracy • Percent composition is used in fertilizers. If the percent composition of nitrogen is determined, then the amount that should ...
... pure the sample was that was used in the reaction. • We can also use the known formula to compare percent composition values to see if the experiment was done with accuracy • Percent composition is used in fertilizers. If the percent composition of nitrogen is determined, then the amount that should ...
Type Of Chemical Reaction
... grams of NH3 gas would bubble out of the solution if you raise the temperature to 90 °C? approx 60 grams 70g-10g=60g h. What is the independent variable on this graph? Temperature i. What is the dependent variable on this graph? Solubility j. Which substance is the most soluble at all temperatures? ...
... grams of NH3 gas would bubble out of the solution if you raise the temperature to 90 °C? approx 60 grams 70g-10g=60g h. What is the independent variable on this graph? Temperature i. What is the dependent variable on this graph? Solubility j. Which substance is the most soluble at all temperatures? ...
Characteristics of Gases Pressure Gas Laws The Ideal
... Some nitrogen gas is in a 2.00-L tank at a pressure of 3.00 atm. The tank is connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No tempera ...
... Some nitrogen gas is in a 2.00-L tank at a pressure of 3.00 atm. The tank is connected to a 5.00-L tank that is completely empty (evacuated), and a valve connects the two tanks., If the valve is opened, determine the total pressure in this two-tank system after the nitrogen stops flowing. No tempera ...
Full answers
... An Ag electrode immersed in a saturated aqueous solution of AgBr has a reduction potential of 0.437 V at 25 C with respect to the standard hydrogen electrode. Calculate the solubility product of AgBr at 25 C. The standard potential for the reduction Ag+(aq) + e- Ag(s), E° = +0.80 V. The aqueou ...
... An Ag electrode immersed in a saturated aqueous solution of AgBr has a reduction potential of 0.437 V at 25 C with respect to the standard hydrogen electrode. Calculate the solubility product of AgBr at 25 C. The standard potential for the reduction Ag+(aq) + e- Ag(s), E° = +0.80 V. The aqueou ...
StudyGuide_Biochemistry
... 1. Carbon atoms can share _______ electrons. 2. What are monomers and polymers? 3. What are the four types of organic compounds that are necessary for life? 4. What three elements must all carbohydrates contain? 5. What are the three types of carbohydrates? How are they different? Give an example of ...
... 1. Carbon atoms can share _______ electrons. 2. What are monomers and polymers? 3. What are the four types of organic compounds that are necessary for life? 4. What three elements must all carbohydrates contain? 5. What are the three types of carbohydrates? How are they different? Give an example of ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Sample Exercise 3.4 Writing Balanced Equations for Combustion Reactions Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the u ...
... Sample Exercise 3.4 Writing Balanced Equations for Combustion Reactions Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the u ...
Experiment #5 WHERE`S THE EVIDENCE
... A physical property is a characteristic of a substance that can be observed without changing the substance into another substance. The temperature at which a solid melts is a physical property. Color, hardness, and texture are other physical properties. A chemical property is a characteristic of a s ...
... A physical property is a characteristic of a substance that can be observed without changing the substance into another substance. The temperature at which a solid melts is a physical property. Color, hardness, and texture are other physical properties. A chemical property is a characteristic of a s ...
SLE133 – “Chemistry in Our World” Summary Notes Week 1
... All the elements in the periodic table are categorized as Metals (good conductors of heat and electricity), Nonmetals (poor conductors of heat and electricity), and Metalloids (have both metallic and non-metallic characteristics). ...
... All the elements in the periodic table are categorized as Metals (good conductors of heat and electricity), Nonmetals (poor conductors of heat and electricity), and Metalloids (have both metallic and non-metallic characteristics). ...
rate of chemical reaction and chemical equilibrium
... Magnitude of equilibrium constant, K, indicates how far a chemical reaction can go. In other words, it is an indicator of the extent of a chemical reaction. Larger the value of K, higher will be the equilibrium concentration of products on the right hand side of the reaction. Smaller value of K indi ...
... Magnitude of equilibrium constant, K, indicates how far a chemical reaction can go. In other words, it is an indicator of the extent of a chemical reaction. Larger the value of K, higher will be the equilibrium concentration of products on the right hand side of the reaction. Smaller value of K indi ...
Inorganic Chemistry Lesson 3
... As you probably have noticed, some molecules contain lower case indexes (“coefficients”). That means the number of atoms can be different in different molecules. For example, “H2 O” ...
... As you probably have noticed, some molecules contain lower case indexes (“coefficients”). That means the number of atoms can be different in different molecules. For example, “H2 O” ...
Chemistry EOC Review Name
... c. nitrogen dioxide 64. List two differences between ionic and covalent compounds. Unit 10 (Chapter 11): 65. What do the following symbols mean: a. ...
... c. nitrogen dioxide 64. List two differences between ionic and covalent compounds. Unit 10 (Chapter 11): 65. What do the following symbols mean: a. ...
Rxn Pred students
... Salts are generally formed from a cation (on the far left side of the periodic table) and an anion (on the far right side of the table) ...
... Salts are generally formed from a cation (on the far left side of the periodic table) and an anion (on the far right side of the table) ...
Chem 2A Final Review
... Solubility: the phenomena is Likes Dissolve Likes, the fact is molecules dissolve if they have the same solubility Melting and Boiling Points Mole Amu, Molar Mass Empirical and Molecular Formula Balancing Chemical Formulas Limiting Reagents Theoretical and Percent Yield 64. The answer that is closes ...
... Solubility: the phenomena is Likes Dissolve Likes, the fact is molecules dissolve if they have the same solubility Melting and Boiling Points Mole Amu, Molar Mass Empirical and Molecular Formula Balancing Chemical Formulas Limiting Reagents Theoretical and Percent Yield 64. The answer that is closes ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.