Fe(H2O)63+ + H2O → ← H3O+ + Fe(H2O)5(OH)2+
... changing the size of the container introducing more I2 into the container measuring the molar concentrations instead of pressures changing the temperature none of these, it is always constant ...
... changing the size of the container introducing more I2 into the container measuring the molar concentrations instead of pressures changing the temperature none of these, it is always constant ...
end of year review
... A. Sodium atoms become positive ions when they lose electrons. B. Each element emits a unique bright-line spectrum when it falls from an excited state to a ground state. C. Beryllium atoms bombarded with alpha particles produce beams that are not influenced by magnetic fields. D. Each element has ph ...
... A. Sodium atoms become positive ions when they lose electrons. B. Each element emits a unique bright-line spectrum when it falls from an excited state to a ground state. C. Beryllium atoms bombarded with alpha particles produce beams that are not influenced by magnetic fields. D. Each element has ph ...
Atomic Structure - s3.amazonaws.com
... Early Models of the Atom Dalton’s Atomic Theory (Between 1766-1844) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions when atoms are separated, joined, or rearranged. Atoms of one element are neve ...
... Early Models of the Atom Dalton’s Atomic Theory (Between 1766-1844) Atoms of different elements can physically mix together or can chemically combine in simple whole-number ratios to form compounds. Chemical reactions when atoms are separated, joined, or rearranged. Atoms of one element are neve ...
Chemistry
... form a substance with qualities different from the elements alone • Chemical bonding- 2+ elements joining together to form a compound • Molecule- the smallest amount of a substance that can exist independently and keep the properties of the substance • Ex) Water= 2 hydrogen atoms & 1 oxygen atom ...
... form a substance with qualities different from the elements alone • Chemical bonding- 2+ elements joining together to form a compound • Molecule- the smallest amount of a substance that can exist independently and keep the properties of the substance • Ex) Water= 2 hydrogen atoms & 1 oxygen atom ...
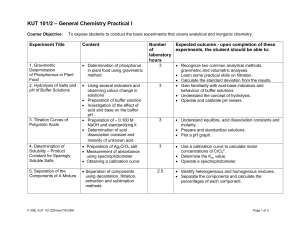
KUT 101/2 – General Chemistry Practical I
... • Calculate the standard deviation from the results. • Gain familiarity with acid-base indicators and behaviour of buffer solutions. • Understand the concept of hydrolysis. • Operate and calibrate pH meters. ...
... • Calculate the standard deviation from the results. • Gain familiarity with acid-base indicators and behaviour of buffer solutions. • Understand the concept of hydrolysis. • Operate and calibrate pH meters. ...
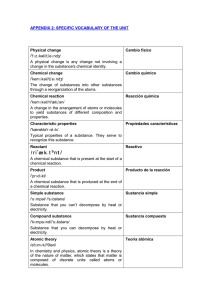
specific vocabulary of the unit
... statistics of weather over durations ranging from decades to millions of years. It can be manifested manifest in changes to averages, extremes, remes, or other statistical measures, and may occur in a specific region or for the Earth as a whole. ...
... statistics of weather over durations ranging from decades to millions of years. It can be manifested manifest in changes to averages, extremes, remes, or other statistical measures, and may occur in a specific region or for the Earth as a whole. ...
Chemistry - StudyTime NZ
... Both atoms have the same atomic number. Because different elements are defined by their atomic number, we can say that both isotopes are the element Carbon. This means they have the same number of p ...
... Both atoms have the same atomic number. Because different elements are defined by their atomic number, we can say that both isotopes are the element Carbon. This means they have the same number of p ...
No Slide Title
... the tendency of the reactants to form products is balanced by the tendency of the products to form reactants. • Could also be defined as a system in which the rates of the forward and reverse reactions are the same. No observable changes occur at equilibrium. ...
... the tendency of the reactants to form products is balanced by the tendency of the products to form reactants. • Could also be defined as a system in which the rates of the forward and reverse reactions are the same. No observable changes occur at equilibrium. ...
end of year review
... _____ 6. Block X and Block Y have the same mass. Both blocks are placed into a container of pure water. Block X floats in the water, and Block Y sinks to the bottom of the container. Which of the following statements is an accurate conclusion from this demonstration? a. Block Y is heavier than Bloc ...
... _____ 6. Block X and Block Y have the same mass. Both blocks are placed into a container of pure water. Block X floats in the water, and Block Y sinks to the bottom of the container. Which of the following statements is an accurate conclusion from this demonstration? a. Block Y is heavier than Bloc ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... concise way by chemical equations. ...
... concise way by chemical equations. ...
File
... 68. A compound has a molar mass of 90. grams per mole and the empirical formula CH2O. What is the molecular formula of this compound? A) CH 2O B) C2H4O2 C) C3H6O3 D) C4H8O4 69. Which equation shows a conservation of mass? A) Na + Cl2 ® NaCl B) Al + Br 2 ® AlBr 3 C) H2O ® H2 + O2 D) PCl 5 ® PCl 3 + C ...
... 68. A compound has a molar mass of 90. grams per mole and the empirical formula CH2O. What is the molecular formula of this compound? A) CH 2O B) C2H4O2 C) C3H6O3 D) C4H8O4 69. Which equation shows a conservation of mass? A) Na + Cl2 ® NaCl B) Al + Br 2 ® AlBr 3 C) H2O ® H2 + O2 D) PCl 5 ® PCl 3 + C ...
Household Items That May Contain Mercury
... Note: These four types of laboratories do not necessarily exist independently of each other. It is quite probable, for instance, that an extraction laboratory will also function as a conversion laboratory to transform the product into a refined form. Hazardous materials responders would have little ...
... Note: These four types of laboratories do not necessarily exist independently of each other. It is quite probable, for instance, that an extraction laboratory will also function as a conversion laboratory to transform the product into a refined form. Hazardous materials responders would have little ...
How to Use Reaction Stoichiometry
... Figure 4.6 (a) When an octane molecule undergoes complete combustion, it forms carbon dioxide and water: one CO2 molecule is formed for each carbon atom present (yellow arrows). (b) However, in a limited supply of oxygen, some of the carbon atoms end up as carbon monoxide molecules, CO, so the yiel ...
... Figure 4.6 (a) When an octane molecule undergoes complete combustion, it forms carbon dioxide and water: one CO2 molecule is formed for each carbon atom present (yellow arrows). (b) However, in a limited supply of oxygen, some of the carbon atoms end up as carbon monoxide molecules, CO, so the yiel ...
Powerpoint
... Double replacement: AB + CD AD + BC – AgNO3(aq) + KCl(aq) KNO3(aq) + AgCl(s) Check Table F for insoluble compounds that form precipitates when solutions are mixed. J Deutsch 2003 ...
... Double replacement: AB + CD AD + BC – AgNO3(aq) + KCl(aq) KNO3(aq) + AgCl(s) Check Table F for insoluble compounds that form precipitates when solutions are mixed. J Deutsch 2003 ...
Matter and Measurement
... We know that at the equivalence point of the titration enough H2SO4 has been added so that all the NaOH has reacted. ...
... We know that at the equivalence point of the titration enough H2SO4 has been added so that all the NaOH has reacted. ...
Catalyst Notes - University of Idaho
... Surface reaction: Generally, species are highly mobile on surfaces. Adsorbed species ‘hop’ to adjacent sites by overcoming a small energy barrier. A good catalyst for a species would be one where these energy barriers are small. A poor catalyst may be such a strong absorber for a species that the sp ...
... Surface reaction: Generally, species are highly mobile on surfaces. Adsorbed species ‘hop’ to adjacent sites by overcoming a small energy barrier. A good catalyst for a species would be one where these energy barriers are small. A poor catalyst may be such a strong absorber for a species that the sp ...
Unit 6 Study Guide - Dorman High School
... Which of the following statments is not true of balancing a chemical equation? A) Subscripts in the reactants must be conserved in the products. B) Coefficients are used to balance the atoms on both sides. C) The law of conservation of matter must be followed. D) Phases are often shown for each comp ...
... Which of the following statments is not true of balancing a chemical equation? A) Subscripts in the reactants must be conserved in the products. B) Coefficients are used to balance the atoms on both sides. C) The law of conservation of matter must be followed. D) Phases are often shown for each comp ...
Chemistry SOL Review Test
... 37) The element iron is made up of the isotopes 58Fe (2.45%), 56Fe (80.3%), 54Fe (10.2%), and 57Fe (7.05%). Calculate the average atomic mass (weight) of iron. Answer: 58 (0.0245)+56(0.803)+54(0.102)+57(0.0705) = 55.92 Unit 4: Electrons in atoms (Ch. 5) 38) Describe the following prop 39) erties and ...
... 37) The element iron is made up of the isotopes 58Fe (2.45%), 56Fe (80.3%), 54Fe (10.2%), and 57Fe (7.05%). Calculate the average atomic mass (weight) of iron. Answer: 58 (0.0245)+56(0.803)+54(0.102)+57(0.0705) = 55.92 Unit 4: Electrons in atoms (Ch. 5) 38) Describe the following prop 39) erties and ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.