Principles of Chemistry 1 and 2 Notes
... two or more bonds of the central atom and the surrounding terminal atoms. - The repulsion between electrons in different bonding pairs causes them to remain as far apart as possible. - The geometry that the molecule finally assumes leads to minimize the repulsion (the molecule assumes more stability ...
... two or more bonds of the central atom and the surrounding terminal atoms. - The repulsion between electrons in different bonding pairs causes them to remain as far apart as possible. - The geometry that the molecule finally assumes leads to minimize the repulsion (the molecule assumes more stability ...
Chapter 14 - Moore Public Schools
... The Effect of Concentration Changes on Equilibrium • Adding a reactant will decrease the amounts of the other reactants and increase the amount of the products until a new position of equilibrium is found that has the same K. • Removing a product will increase the amounts of the other products and ...
... The Effect of Concentration Changes on Equilibrium • Adding a reactant will decrease the amounts of the other reactants and increase the amount of the products until a new position of equilibrium is found that has the same K. • Removing a product will increase the amounts of the other products and ...
essential-biology-03-chemistry-of-life
... ESSENTIAL BIOLOGY 03: THE CHEMISTRY OF LIFE 126. A limiting factor is the one factor which is in shortest supply or is preventing the rate of a reaction from increasing. If this factor were increased, rate of reaction would increase until another plateau was reached. Explain HOW the following facto ...
... ESSENTIAL BIOLOGY 03: THE CHEMISTRY OF LIFE 126. A limiting factor is the one factor which is in shortest supply or is preventing the rate of a reaction from increasing. If this factor were increased, rate of reaction would increase until another plateau was reached. Explain HOW the following facto ...
File - Mr. Arnold`s Classes
... extra carbon is released as carbon dioxide. Acetyl CoA can also be produced from fatty acids. When the fatty acid chain contains an even number of carbons, no CO 2 is released. How many Acetyl CoA molecules can be produced with the following fatty acids? a. 23C b. 18C c. 31C ...
... extra carbon is released as carbon dioxide. Acetyl CoA can also be produced from fatty acids. When the fatty acid chain contains an even number of carbons, no CO 2 is released. How many Acetyl CoA molecules can be produced with the following fatty acids? a. 23C b. 18C c. 31C ...
Chemistry 100 Second Homework
... a. Draw the structure of a short segment of polytetrafluoroethylene. b. What type of intermolecular attractions are present between polytetrafluoroethylene molecules? (Be careful! Think about the shape of the molecule.) c. The C-F bond in polytetrafluoroethylene has a bond energy of about 504 kJ/mol ...
... a. Draw the structure of a short segment of polytetrafluoroethylene. b. What type of intermolecular attractions are present between polytetrafluoroethylene molecules? (Be careful! Think about the shape of the molecule.) c. The C-F bond in polytetrafluoroethylene has a bond energy of about 504 kJ/mol ...
CHAPTER 14
... structure is stabilized by intramolecular hydrogen bonds between the NH and CO groups of the main chain, giving rise to an overall rodlike shape. The CO group of each amino acid is hydrogen-bonded to the NH group of the amino acid that is four residues away in the sequence. In this manner all the ma ...
... structure is stabilized by intramolecular hydrogen bonds between the NH and CO groups of the main chain, giving rise to an overall rodlike shape. The CO group of each amino acid is hydrogen-bonded to the NH group of the amino acid that is four residues away in the sequence. In this manner all the ma ...
21. glycolysis
... on some other common hexoses such as fructose and mannose. The activity of hexokinase is inhibited by the product of the raction (i.e., glucose 6-phosphate) which binds the enzyme at an allosteric site. Hexokinase has a high affinity (i.e., low Km value of about 1.0 mM) for Fig. 21–5. Computer graph ...
... on some other common hexoses such as fructose and mannose. The activity of hexokinase is inhibited by the product of the raction (i.e., glucose 6-phosphate) which binds the enzyme at an allosteric site. Hexokinase has a high affinity (i.e., low Km value of about 1.0 mM) for Fig. 21–5. Computer graph ...
Urea–urethane nanocomposites obtained from modified methylalumoxane oligomers A. B
... molar ratio of substrates. Urea–urethane elastomers are characterized by such advantageous properties like high tear strength and tensile modulus. Therefore they have found many applications for a wide range of products like for instance sieves for mining industry. We have developed a new method of ...
... molar ratio of substrates. Urea–urethane elastomers are characterized by such advantageous properties like high tear strength and tensile modulus. Therefore they have found many applications for a wide range of products like for instance sieves for mining industry. We have developed a new method of ...
Autocatalytic Sets in E. coli Metabolism
... reactions within the set of catalyzed reactions. Moreover, by grouping these reactions with the same generic catalyst (Protein or RNA), we are simplifying the network’s catalyst space without losing biological information. iv) Reactions for which the E. coli enzyme is unknown were assigned to anothe ...
... reactions within the set of catalyzed reactions. Moreover, by grouping these reactions with the same generic catalyst (Protein or RNA), we are simplifying the network’s catalyst space without losing biological information. iv) Reactions for which the E. coli enzyme is unknown were assigned to anothe ...
Life 9e - Garvness
... a. Electrons are received from NADH and FADH2. b. Electrons are passed from donor to recipient carrier molecules in a series of oxidation– reduction reactions. c. Usually the terminal electron acceptor is oxygen. d. Most of the enzymes are part of the inner mitochondrial membrane. e. All of the abov ...
... a. Electrons are received from NADH and FADH2. b. Electrons are passed from donor to recipient carrier molecules in a series of oxidation– reduction reactions. c. Usually the terminal electron acceptor is oxygen. d. Most of the enzymes are part of the inner mitochondrial membrane. e. All of the abov ...
chemical reactions and stoichiometry chemical reactions and
... R4.1.1 Although gasoline is a complex mixture of molecules, the chemical reaction that takes place in an automobile engine can be represented by combustion of one of its components, octane (C8H18). Such burning of fossil fuels releases millions of tonnes of carbon dioxide into Earth’s atmosphere eac ...
... R4.1.1 Although gasoline is a complex mixture of molecules, the chemical reaction that takes place in an automobile engine can be represented by combustion of one of its components, octane (C8H18). Such burning of fossil fuels releases millions of tonnes of carbon dioxide into Earth’s atmosphere eac ...
Ch 10 Practice Problems 1. Consider the process A(l) A(s). Which
... For the reaction A + B C + D, H° = +40 kJ and S° = +50 J/K. Therefore, the reaction under standard conditions is A) spontaneous at temperatures less than 10 K. B) spontaneous at temperatures greater than 800 K. C) spontaneous only at temperatures between 10 K and 800 K. D) spontaneous at all tem ...
... For the reaction A + B C + D, H° = +40 kJ and S° = +50 J/K. Therefore, the reaction under standard conditions is A) spontaneous at temperatures less than 10 K. B) spontaneous at temperatures greater than 800 K. C) spontaneous only at temperatures between 10 K and 800 K. D) spontaneous at all tem ...
Oxidative decarboxylation of pyruvate
... Only about 7 % of the total potential energy present in glucose is released in glycolysis. ...
... Only about 7 % of the total potential energy present in glucose is released in glycolysis. ...
Leaf orientation, photorespiration and xanthophyll cycle protect
... During leaf development, the newly initiating leaves are often exposed to full sunlight at the topmost canopy, indicating that those young leaves have to endure extremely high irradiance. However, young leaves have lower photosynthesis activity per unit area compared with fully developed leaves (Kra ...
... During leaf development, the newly initiating leaves are often exposed to full sunlight at the topmost canopy, indicating that those young leaves have to endure extremely high irradiance. However, young leaves have lower photosynthesis activity per unit area compared with fully developed leaves (Kra ...
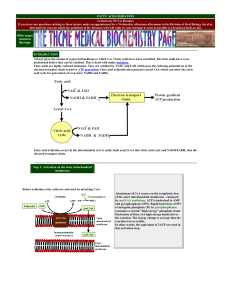
Tetracycline Antibiotics
... Aminoglycosides are so named because their structures consist of amino sugars linked glycosidically. All have at least one aminohexose, and some have a pentose lacking an amino group. ...
... Aminoglycosides are so named because their structures consist of amino sugars linked glycosidically. All have at least one aminohexose, and some have a pentose lacking an amino group. ...
CHANNELING OF SUBSTRATES AND INTERMEDIATES IN
... In the original investigation of tryptophan synthase, the electron density corresponding to a surface loop in the α-subunit, namely that delineated by Leu-177 to Ala-190, was disordered. Subsequent X-ray crystallographic studies of a sitedirected mutant protein (βK87T) with various combinations of l ...
... In the original investigation of tryptophan synthase, the electron density corresponding to a surface loop in the α-subunit, namely that delineated by Leu-177 to Ala-190, was disordered. Subsequent X-ray crystallographic studies of a sitedirected mutant protein (βK87T) with various combinations of l ...
CHAPTER 6
... Digestive breakdown is unregulated - nearly 100% • But tissue glycogen is an important energy reservoir - its breakdown is carefully controlled • Glycogen consists of "granules" of high MW range from 6 x 106 ~ 1600 x 106 • Glycogen phosphorylase cleaves glucose from the nonreducing ends of glycogen ...
... Digestive breakdown is unregulated - nearly 100% • But tissue glycogen is an important energy reservoir - its breakdown is carefully controlled • Glycogen consists of "granules" of high MW range from 6 x 106 ~ 1600 x 106 • Glycogen phosphorylase cleaves glucose from the nonreducing ends of glycogen ...
CHAPTER 14 CHEMICAL KINETICS
... there are 8 A molecules and 8 B molecules. At t 20 s, the concentration of A will decrease to half of its concentration at t 10 s. There will be 4 A molecules at t 20 s. Because the mole ratio between A and B is 1:1, four more B molecules will be produced and there will be 12 B molecules prese ...
... there are 8 A molecules and 8 B molecules. At t 20 s, the concentration of A will decrease to half of its concentration at t 10 s. There will be 4 A molecules at t 20 s. Because the mole ratio between A and B is 1:1, four more B molecules will be produced and there will be 12 B molecules prese ...
chapter 21
... there are 8 A molecules and 8 B molecules. At t 20 s, the concentration of A will decrease to half of its concentration at t 10 s. There will be 4 A molecules at t 20 s. Because the mole ratio between A and B is 1:1, four more B molecules will be produced and there will be 12 B molecules prese ...
... there are 8 A molecules and 8 B molecules. At t 20 s, the concentration of A will decrease to half of its concentration at t 10 s. There will be 4 A molecules at t 20 s. Because the mole ratio between A and B is 1:1, four more B molecules will be produced and there will be 12 B molecules prese ...
PDF - MD Body and Med spa
... Alternatively you can email any problems to Dr. Brookes who will reply to your message as soon as possible ([email protected]). Other pages related to this topic ...
... Alternatively you can email any problems to Dr. Brookes who will reply to your message as soon as possible ([email protected]). Other pages related to this topic ...
Mole-Volume Conversion Assignment
... This means that 1 mole of carbon dioxide gas and one mole of hydrogen gas will take up the same volume if they have the same temperature and pressure. At standard temperature and pressure, this volume is 22.4L. Standard temperature and pressure is (273K (0oC) and 1 atm). ...
... This means that 1 mole of carbon dioxide gas and one mole of hydrogen gas will take up the same volume if they have the same temperature and pressure. At standard temperature and pressure, this volume is 22.4L. Standard temperature and pressure is (273K (0oC) and 1 atm). ...
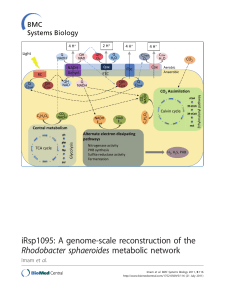
iRsp1095: A genome-scale reconstruction of the Rhodobacter
... To fill this knowledge gap, we are modeling the flow of carbon and reducing power in the well-studied photosynthetic bacterium Rhodobacter sphaeroides. This facultative bacterium is capable of either aerobic or anaerobic respiration, depending on the availability of oxygen (O2) or alternative electr ...
... To fill this knowledge gap, we are modeling the flow of carbon and reducing power in the well-studied photosynthetic bacterium Rhodobacter sphaeroides. This facultative bacterium is capable of either aerobic or anaerobic respiration, depending on the availability of oxygen (O2) or alternative electr ...