CHEM_01A_ExptD_Copper_Cycle_F14
... In this experiment, you will run a series of reactions on a piece of copper wire that transforms copper into several different chemical compounds and, eventually, into an amount of copper powder. ...
... In this experiment, you will run a series of reactions on a piece of copper wire that transforms copper into several different chemical compounds and, eventually, into an amount of copper powder. ...
Nickel plating
... 1. Fill a 50x100mm crystallizing dish with the nickel plating solution to within an 1/8inch from the top (≅210mL). This solution contains hydrochloric acid, is corrosive, and must be handled carefully. Clean up any spills immediately 2. Place the dish on a hotplate and warm the solution to about 60- ...
... 1. Fill a 50x100mm crystallizing dish with the nickel plating solution to within an 1/8inch from the top (≅210mL). This solution contains hydrochloric acid, is corrosive, and must be handled carefully. Clean up any spills immediately 2. Place the dish on a hotplate and warm the solution to about 60- ...
Chemistry - Separation techniques
... accordance with their employer’s requirements, making use of up-to-date information and taking account of their own particular circumstances. Any local rules or restrictions issued by the employer must always be followed. CLEAPSS resources are useful for carrying out risk-assessments: (http://scienc ...
... accordance with their employer’s requirements, making use of up-to-date information and taking account of their own particular circumstances. Any local rules or restrictions issued by the employer must always be followed. CLEAPSS resources are useful for carrying out risk-assessments: (http://scienc ...
Redox Reactions and Electrochemistry
... • We would choose the production of O2(g) and Cu(s). • But the voltage for producing O2(g) from solution is considerably higher than the standard potential, because of the high activation energy needed to form O2(g). • The voltage for this half cell seems to be closer to –1.5 V in reality. • The ...
... • We would choose the production of O2(g) and Cu(s). • But the voltage for producing O2(g) from solution is considerably higher than the standard potential, because of the high activation energy needed to form O2(g). • The voltage for this half cell seems to be closer to –1.5 V in reality. • The ...
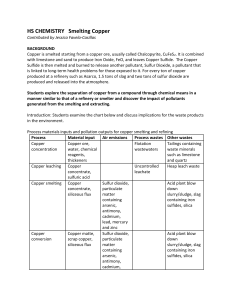
Chemical Reactions of Copper and Percent Recovery
... 4. Twist the copper wire into a flat, tight coil (the size of a penny) and place it in the bottom of your beaker. 5. Place your beaker in the hood. 6. Either you or your instructor will place ~ 5 mL of concentrated nitric acid (conc HNO3 is very CAUSTIC – Avoid Contact!! Use gloves!!) in the beaker. ...
... 4. Twist the copper wire into a flat, tight coil (the size of a penny) and place it in the bottom of your beaker. 5. Place your beaker in the hood. 6. Either you or your instructor will place ~ 5 mL of concentrated nitric acid (conc HNO3 is very CAUSTIC – Avoid Contact!! Use gloves!!) in the beaker. ...
Copper Chemistry
... Usage of copper compounds also dates back to before 4000 BC. Copper sulphate for example was an especially important compound in early times. Ancient Egyptians used it as a mordant in their dyeing process. The compound was also used to make ointments and other such preparations. Later, medicinal use ...
... Usage of copper compounds also dates back to before 4000 BC. Copper sulphate for example was an especially important compound in early times. Ancient Egyptians used it as a mordant in their dyeing process. The compound was also used to make ointments and other such preparations. Later, medicinal use ...
Sec 5.8 - 5.11 notes
... 9) Electrons do not travel through the ______ _______, only through the _______. 10) Ions (cations & anions) do not travel through the wire but only through the _____________. 11) The salt bridge can contain any ______________. 12) The anode will ________(gains/loses) mass as it is __________(oxidiz ...
... 9) Electrons do not travel through the ______ _______, only through the _______. 10) Ions (cations & anions) do not travel through the wire but only through the _____________. 11) The salt bridge can contain any ______________. 12) The anode will ________(gains/loses) mass as it is __________(oxidiz ...
Aluminum and Copper
... gas produced? (b) Write an equation for the formation of the gas from aluminum and water. (c) How would this side reaction affect the experimental mole ratio for the amounts of aluminum and copper? 3. The copper metal produced in the reaction usually turns light green upon drying overnight. (a) W ...
... gas produced? (b) Write an equation for the formation of the gas from aluminum and water. (c) How would this side reaction affect the experimental mole ratio for the amounts of aluminum and copper? 3. The copper metal produced in the reaction usually turns light green upon drying overnight. (a) W ...
AP Ch. 20 Notes (2005)
... has a lower electrical potential energy than the anode. • Potential difference: difference in electrical potential. • The potential difference is measured in volts… One volt (V) is the potential difference required to impart one joule (J) of energy to a charge of one coulomb (C). ...
... has a lower electrical potential energy than the anode. • Potential difference: difference in electrical potential. • The potential difference is measured in volts… One volt (V) is the potential difference required to impart one joule (J) of energy to a charge of one coulomb (C). ...
A) Sn4+ → Sn2+ + 2e
... E) Anode: Cl 2 + 2e – ® 2 Cl – Cathode: Zn ® Zn 2+ + 2e – 69. What are the anode and cathode half–reactions for the electrolysis of fused (melted) NaCl? A) Anode: 2 Cl– ® Cl 2 + 2e – Cathode: 2 Na+ + 2e – ® 2 Na B) Anode: 2 Cl – ® Cl 2 + 2e – Cathode: 2 H 2O + 2e ...
... E) Anode: Cl 2 + 2e – ® 2 Cl – Cathode: Zn ® Zn 2+ + 2e – 69. What are the anode and cathode half–reactions for the electrolysis of fused (melted) NaCl? A) Anode: 2 Cl– ® Cl 2 + 2e – Cathode: 2 Na+ + 2e – ® 2 Na B) Anode: 2 Cl – ® Cl 2 + 2e – Cathode: 2 H 2O + 2e ...
ch19 MSJ jlm
... 2. Use “sacrificial metal” such as Mg – (this is also cathodic protection). 3. Cover it (paint). 4. Create rust-resistant alloys, e.g., stainless steel (Fe/Ni/Cr), or nickel steels (Fe/Ni). ...
... 2. Use “sacrificial metal” such as Mg – (this is also cathodic protection). 3. Cover it (paint). 4. Create rust-resistant alloys, e.g., stainless steel (Fe/Ni/Cr), or nickel steels (Fe/Ni). ...
35. Number of reactions - Royal Society of Chemistry
... (Cu(OH)2.CuCO3) is formed initially. On adding excess A this dissolves to form a deep blue solution of Cu(NH3)42+(aq). ...
... (Cu(OH)2.CuCO3) is formed initially. On adding excess A this dissolves to form a deep blue solution of Cu(NH3)42+(aq). ...
The Synthesis and Analysis of Copper (II) Carboxylates
... Carolyn S. Yoder, and Elizabeth R. Bullock Millersville University, Millersville, PA 17551 Synthesis is unique to chemistry. Chemists make compounds, separate them from their reaction mixtures, and then determine their purity. These synthetic procedures had their origins in alchemy and, in their inc ...
... Carolyn S. Yoder, and Elizabeth R. Bullock Millersville University, Millersville, PA 17551 Synthesis is unique to chemistry. Chemists make compounds, separate them from their reaction mixtures, and then determine their purity. These synthetic procedures had their origins in alchemy and, in their inc ...
Electrolytic Etching Copper and Silver Using Copper Nitrate, a
... etching a line or area deep enough to hold ink, we often want a deeper etch with a surface that is a finish in itself. The caustic nature of nitric, hydrochloric and sulfuric acids as well as the mess and limitations of ferric chloride can be replaced with self-sustaining systems that can be operate ...
... etching a line or area deep enough to hold ink, we often want a deeper etch with a surface that is a finish in itself. The caustic nature of nitric, hydrochloric and sulfuric acids as well as the mess and limitations of ferric chloride can be replaced with self-sustaining systems that can be operate ...
CHEMISTRY SEMESTER ONE LAB 1 Lab 1: Stoichiometry and
... 3. Measure 30 mL of 1.0 M CuSO4 solution into a graduated cylinder. Pour it into an erlenmeyer flask, and heat gently to almost boiling. 4. Slowly add the hot CuSO4 solution to the beaker containing the iron powder. 5. Swirl the flask to insure the reaction goes to completion. When the reaction is c ...
... 3. Measure 30 mL of 1.0 M CuSO4 solution into a graduated cylinder. Pour it into an erlenmeyer flask, and heat gently to almost boiling. 4. Slowly add the hot CuSO4 solution to the beaker containing the iron powder. 5. Swirl the flask to insure the reaction goes to completion. When the reaction is c ...
Wet Corrosion Conditions for Wet Corrosion Just as we live in an
... depends on the molecular weight of the active chemical specie. Electrochemical attack, the scientific name for wet corrosion, depends on three circumstances occurring simultaneously. This might seem restrictive, but the evidence of wet corrosion that we see all around us proves that these conditions ...
... depends on the molecular weight of the active chemical specie. Electrochemical attack, the scientific name for wet corrosion, depends on three circumstances occurring simultaneously. This might seem restrictive, but the evidence of wet corrosion that we see all around us proves that these conditions ...
Electrolysis answers - Barnard Castle School
... (copper) lost during washing owtte allow different washing of electrodes ...
... (copper) lost during washing owtte allow different washing of electrodes ...
Unit IV: Nature of Matter
... variety of cathodes (different metals) were tested and all produced same results. Magnetic fields deflected the rays. The rays produced some chemical reactions similar to those produced by light. ...
... variety of cathodes (different metals) were tested and all produced same results. Magnetic fields deflected the rays. The rays produced some chemical reactions similar to those produced by light. ...
Early Atomic Models
... wide variety of cathodes (different metals) were tested and all produced same results. Magnetic fields deflected the rays. The rays produced some chemical reactions similar to those produced by light. ...
... wide variety of cathodes (different metals) were tested and all produced same results. Magnetic fields deflected the rays. The rays produced some chemical reactions similar to those produced by light. ...
IsaKidd refining technology

The IsaKidd Technology is a copper electrorefining and electrowinning technology that was developed independently by Copper Refineries Proprietary Limited (“CRL”), a Townsville, Queensland subsidiary of MIM Holdings Limited (which is now part of the Glencore Xstrata group of companies), and at the Falconbridge Limited (“Falconbridge”) now-dismantled Kidd Creek refinery that was at Timmins, Ontario. It is based around the use of reusable cathode starter sheets for copper electrorefining and the automated stripping of the deposited “cathode copper” from them.