Elementary Principles of Chemical Processes, 3rd Update Edition
... FEATURES Industrial Process Case Studies An important feature of the book is a set of industrial process case studies that demonstrate the role of single-unit calculations in the analysis of multiple-unit processes. We have designed the case studies to be worked on as term projects by individuals or ...
... FEATURES Industrial Process Case Studies An important feature of the book is a set of industrial process case studies that demonstrate the role of single-unit calculations in the analysis of multiple-unit processes. We have designed the case studies to be worked on as term projects by individuals or ...
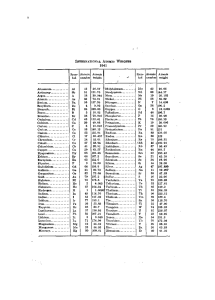
INTEKNATIONAL ATOMIC WEIGHTS Aluminum... Antimony..., Argon
... This revision introduces many new experiments and revises others in an attempt to keep abreast of the rapid developments in physical chemistry. Some of the former experiments have been eliminated or expanded because they have found their way into earlier courses and are already known to students, wh ...
... This revision introduces many new experiments and revises others in an attempt to keep abreast of the rapid developments in physical chemistry. Some of the former experiments have been eliminated or expanded because they have found their way into earlier courses and are already known to students, wh ...
006 Thermochemistry
... 56. The enthalpy change when a strong acid is neutralized by strong base is -56.1 kJ/mol. If 12.0 mL of 6.00 M HBr at 21.30C is mixed with 300. mL of 0.250 M NaOH, also at 21.30C, what will the maximum temperature reached by the resulting solution? [Assume that there is no heat loss to the contain ...
... 56. The enthalpy change when a strong acid is neutralized by strong base is -56.1 kJ/mol. If 12.0 mL of 6.00 M HBr at 21.30C is mixed with 300. mL of 0.250 M NaOH, also at 21.30C, what will the maximum temperature reached by the resulting solution? [Assume that there is no heat loss to the contain ...
Chapter 8: Balances on Nonreactive Processes
... *The flow rates were obtained by multiplying the molar fraction of each component by the total flow rate of the corresponding stream. *The enthalpy of the liquid water exiting the system was set to zero since it is leaving the system at the reference conditions. This also occurs for the nitrogen and ...
... *The flow rates were obtained by multiplying the molar fraction of each component by the total flow rate of the corresponding stream. *The enthalpy of the liquid water exiting the system was set to zero since it is leaving the system at the reference conditions. This also occurs for the nitrogen and ...
Information on measuring ammonia in water
... For coolant monitoring, the plant operator is more concerned with the fast detection of an ammonia leak in the primary circuit via a qualitative ammonia detection in the secondary circuit. The measurement of the exact concentration in the secondary circuit is of minor importance, since the main obje ...
... For coolant monitoring, the plant operator is more concerned with the fast detection of an ammonia leak in the primary circuit via a qualitative ammonia detection in the secondary circuit. The measurement of the exact concentration in the secondary circuit is of minor importance, since the main obje ...
the chemical and physical properties of condensed
... as phase diagram entities are of this type3032. Yet another type of transformation occurs during the dehydration of a crystalline hydrate. In most systems the phosphate anion is dehydrated without degradation, while in other systems the phosphate anion is destroyed33. Examples of salts which dehydra ...
... as phase diagram entities are of this type3032. Yet another type of transformation occurs during the dehydration of a crystalline hydrate. In most systems the phosphate anion is dehydrated without degradation, while in other systems the phosphate anion is destroyed33. Examples of salts which dehydra ...
Thermochemistry - Pearson Canada
... Heat is energy transferred between a system and its surroundings as a result of a temperature difference. Energy that passes from a warmer body (with a higher temperature) to a colder body (with a lower temperature) is transferred as heat. At the molecular level, molecules of the warmer body, throug ...
... Heat is energy transferred between a system and its surroundings as a result of a temperature difference. Energy that passes from a warmer body (with a higher temperature) to a colder body (with a lower temperature) is transferred as heat. At the molecular level, molecules of the warmer body, throug ...
Lecture notes
... added to the saturated solution, what would be the effect on the solubility of lead(II) chloride? (Hint: consider how the equilibrium written above would shift, according to Le Châtelier’s ...
... added to the saturated solution, what would be the effect on the solubility of lead(II) chloride? (Hint: consider how the equilibrium written above would shift, according to Le Châtelier’s ...
aq - Valencia College
... g. If the equation is not available, to calculate the slope, two points that fall on the line will be selected and a calculation of the density will be the change in y over change in x via the formula: ∆y/∆x = (y2 – y1)/ (x2 – x1). h. The value for the y-intercept, or the point where the line inters ...
... g. If the equation is not available, to calculate the slope, two points that fall on the line will be selected and a calculation of the density will be the change in y over change in x via the formula: ∆y/∆x = (y2 – y1)/ (x2 – x1). h. The value for the y-intercept, or the point where the line inters ...
ioan stamatin
... (chemical thermodynamics), one for biology and one for students at physics. For century that is truth when the interdisciplinary sciences still were not born. Departments from Faculty of Physics, University of Bucharest, have developed in the last decade large interdisciplinary fields such as Medica ...
... (chemical thermodynamics), one for biology and one for students at physics. For century that is truth when the interdisciplinary sciences still were not born. Departments from Faculty of Physics, University of Bucharest, have developed in the last decade large interdisciplinary fields such as Medica ...
Chemical Vapor Deposition (CVD)
... 1960: Introduction of the terms CVD and PVD to distinguish “chemical vapour deposition” from “physical vapour deposition.” 1960: Introduction of CVD in semiconductor fabrication. 1960: CVD TiC coating on cemented carbide tools introduced and development of CVD tungsten. 1963: Introduction of ...
... 1960: Introduction of the terms CVD and PVD to distinguish “chemical vapour deposition” from “physical vapour deposition.” 1960: Introduction of CVD in semiconductor fabrication. 1960: CVD TiC coating on cemented carbide tools introduced and development of CVD tungsten. 1963: Introduction of ...
File - IB CHEM NINJA
... consequence, macroscopic properties of the system (that is those that can be observed or measured, such as its colour, density, pH) are constant, even though on a molecular scale there is continual interconversion of reactants and products. The concentrations of the species at equilibrium will refle ...
... consequence, macroscopic properties of the system (that is those that can be observed or measured, such as its colour, density, pH) are constant, even though on a molecular scale there is continual interconversion of reactants and products. The concentrations of the species at equilibrium will refle ...
H/ t = W/m
... same thermometer was used during the experiment). Both the thermometer and thermocouple were placed in the beaker of water. The hot plate was used to slowly raise the temperature as the stirring rod allowed the water to maintain a uniform temperature. The Virtual Bench software was used to read volt ...
... same thermometer was used during the experiment). Both the thermometer and thermocouple were placed in the beaker of water. The hot plate was used to slowly raise the temperature as the stirring rod allowed the water to maintain a uniform temperature. The Virtual Bench software was used to read volt ...
WA AP Chem gas law IMF MC Set C
... B. The intermolecular forces, such as hydrogen bonds, between the H2O (l) molecules require energy to be overcome, and the molecules can only separate to become a gas once these intermolecular forces are overcome. C. Water vapor is hotter than or equal in temperature to water at constant pressure, a ...
... B. The intermolecular forces, such as hydrogen bonds, between the H2O (l) molecules require energy to be overcome, and the molecules can only separate to become a gas once these intermolecular forces are overcome. C. Water vapor is hotter than or equal in temperature to water at constant pressure, a ...
Dialysis Guide_scienova
... concentration gradient, which is highest at the start of dialysis. Through the dialysis process the concentration gradient between the sample and dialysis buffer will be reduced. In order to ...
... concentration gradient, which is highest at the start of dialysis. Through the dialysis process the concentration gradient between the sample and dialysis buffer will be reduced. In order to ...
Novel Methods and Materials in Development of Liquid Carrier
... done. There were proposals on microreactors, on membrane reactor and two on molecular modelling - one in the field of crystallisation and another in the field of mass transport modelling in zeolite membranes. At least the last topic, molecular modelling, proved to be a real advantage for my own work ...
... done. There were proposals on microreactors, on membrane reactor and two on molecular modelling - one in the field of crystallisation and another in the field of mass transport modelling in zeolite membranes. At least the last topic, molecular modelling, proved to be a real advantage for my own work ...
Oxygen diffusion through perovskite membranes
... Perovskites (ABO3) are a prominent topic of research in materials science (high-Tcelectric conductor [1, 2], ferroelectric [3, 4] or high k dielectric [5], or magnetoresistance [6] material). Doping of these perovskites with multivalent cations on the A- and B- sites can lead to the simultaneous occ ...
... Perovskites (ABO3) are a prominent topic of research in materials science (high-Tcelectric conductor [1, 2], ferroelectric [3, 4] or high k dielectric [5], or magnetoresistance [6] material). Doping of these perovskites with multivalent cations on the A- and B- sites can lead to the simultaneous occ ...
Heat transfer mechanisms of laminar flames of hydrogen+ oxygen
... importance in the lighting industry. In this paper a laminar flame of hydrogen + oxygen is used as an impinging jet in a stagnation-flow-like configuration to investigate the heating of a glass product. The research was intended to analyze the crucial phenomena determining the heat transfer rate. Th ...
... importance in the lighting industry. In this paper a laminar flame of hydrogen + oxygen is used as an impinging jet in a stagnation-flow-like configuration to investigate the heating of a glass product. The research was intended to analyze the crucial phenomena determining the heat transfer rate. Th ...
What is a solution
... The other concentration units are less frequently used:Ideal Solution: A solution of two or more constituents is said to be ideal if it obeys Raoult’s law under all conditions of temperature and concentration. We are considering a solution composed of a volatile solvent and one or more involatile so ...
... The other concentration units are less frequently used:Ideal Solution: A solution of two or more constituents is said to be ideal if it obeys Raoult’s law under all conditions of temperature and concentration. We are considering a solution composed of a volatile solvent and one or more involatile so ...
3 CO 2 (g) + 4 H 2 O(l)
... ΔH = 1.0 × 102 g × 4.18 J g-1 °C-1 × (27.4 – 15.6) °C = + 4.9 × 103 J i.e., our system has gained 4.9 × 103 J of enthalpy If we now take our 1.0 × 102 g of water at 15.6 °C and heat it to 94.6 °C and then cool it to 27.4 °C, then the overall energy change can be calculated as ΔH (15.6 °C 94.6 °C) ...
... ΔH = 1.0 × 102 g × 4.18 J g-1 °C-1 × (27.4 – 15.6) °C = + 4.9 × 103 J i.e., our system has gained 4.9 × 103 J of enthalpy If we now take our 1.0 × 102 g of water at 15.6 °C and heat it to 94.6 °C and then cool it to 27.4 °C, then the overall energy change can be calculated as ΔH (15.6 °C 94.6 °C) ...
heat
... stored potential energy it contains. The controlled explosions of the gasoline in a car’s engine transform the potential energy into useful work, which can be used to propel the car. ...
... stored potential energy it contains. The controlled explosions of the gasoline in a car’s engine transform the potential energy into useful work, which can be used to propel the car. ...
comparison of candidate materials for seasonal storage of solar heat
... Loss of reactive material due to hystereses in reaction temperature and reaction pressure • Cooling down of hot materials leaving the reactors. This can be divided into sensible heat loss, associated with a decrease in temperature, and latent heat loss, associated with a phase change at constant tem ...
... Loss of reactive material due to hystereses in reaction temperature and reaction pressure • Cooling down of hot materials leaving the reactors. This can be divided into sensible heat loss, associated with a decrease in temperature, and latent heat loss, associated with a phase change at constant tem ...
Transport Processes: Momentum, Heat, and Mass
... The separation processes deal mainly with the transfer and change of energy and the transfer and change of materials, primarily by physical means but also by physical– chemical means. The important separation processes, which can be combined in various sequences in a process and which are covered in ...
... The separation processes deal mainly with the transfer and change of energy and the transfer and change of materials, primarily by physical means but also by physical– chemical means. The important separation processes, which can be combined in various sequences in a process and which are covered in ...
Membrane distillation

Membrane distillation is a thermally driven separational programm in which separation is enabled due to phase change. A hydrophobic membrane displays a barrier for the liquid phase, allowing the vapour phase (e.g. water vapour) pass through the membrane's pores. The driving force of the process is given by a partial vapour pressure difference commonly triggered by a temperature difference.