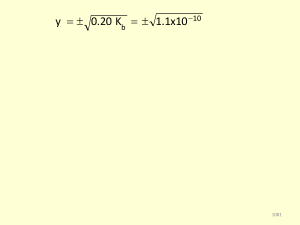Strecker Degradation Products of Aspartic and Glutamic Acids and
... acid. A recombination of free radicals leads to dicarboxylic acids (succinic acid from aspartic acid, succinic, glutaric and adipic acids from glutamic acid). The major volatile products (besides the aldehydes) are lower carboxylic acids (acetic acid from aspartic acid and propionic acid acid from g ...
... acid. A recombination of free radicals leads to dicarboxylic acids (succinic acid from aspartic acid, succinic, glutaric and adipic acids from glutamic acid). The major volatile products (besides the aldehydes) are lower carboxylic acids (acetic acid from aspartic acid and propionic acid acid from g ...
Acid‒base reaction
... The hydrogen requirement of Arrhenius and Brønsted–Lowry was removed by the Lewis definition of acid–base reactions, devised by Gilbert N. Lewis in 1923,[12] in the same year as Brønsted–Lowry, but it was not elaborated by him until 1938.[2] Instead of defining acid–base reactions in terms of proton ...
... The hydrogen requirement of Arrhenius and Brønsted–Lowry was removed by the Lewis definition of acid–base reactions, devised by Gilbert N. Lewis in 1923,[12] in the same year as Brønsted–Lowry, but it was not elaborated by him until 1938.[2] Instead of defining acid–base reactions in terms of proton ...
Topic 8: ACIDS and BASES
... H3O+ or with a another base to form water. Other examples include CH3+ and Br+ (Hal+) which you will study in organic chemistry. molecules containing positive centres (mostly organic molecules) as a result of polar bonds within the molecule e.g. C in CO2 or in halogenalkanes and S in SO2. All Br ...
... H3O+ or with a another base to form water. Other examples include CH3+ and Br+ (Hal+) which you will study in organic chemistry. molecules containing positive centres (mostly organic molecules) as a result of polar bonds within the molecule e.g. C in CO2 or in halogenalkanes and S in SO2. All Br ...
Topic 8: ACIDS and BASES
... aqueous solutions, and therefore a higher pH, than strong acids of the same concentration. Strong acids: HCl, HNO3, H2SO4 , HBr, HI, H3PO4. Weak acids: CH3COOH (=ethanoic acid), H2CO3 (carbonic acid), HCOOH, citric acid, all carboxylic/organic acids. Exercise: For each of the above acids write an eq ...
... aqueous solutions, and therefore a higher pH, than strong acids of the same concentration. Strong acids: HCl, HNO3, H2SO4 , HBr, HI, H3PO4. Weak acids: CH3COOH (=ethanoic acid), H2CO3 (carbonic acid), HCOOH, citric acid, all carboxylic/organic acids. Exercise: For each of the above acids write an eq ...
chemistry 103 - chem.uwec.edu
... Acid-base titrations: The impact of hydrolysis Salt hydrolysis has an important effect on the pH profile of acid-base titrations. The equivalence point may be above or below neutral conditions (i.e. pH = ...
... Acid-base titrations: The impact of hydrolysis Salt hydrolysis has an important effect on the pH profile of acid-base titrations. The equivalence point may be above or below neutral conditions (i.e. pH = ...
Thin-Layer Chromatography: Applying TLC as a
... interactions of the salicylic molecules with each other in the reaction mixture. This may be because the hydrogen on the phenolic substituent of salicylic acid interacts with the double bonded oxygen of the other substituent, carboxylic acid, through hydrogen bonding thus decreasing its polarity sig ...
... interactions of the salicylic molecules with each other in the reaction mixture. This may be because the hydrogen on the phenolic substituent of salicylic acid interacts with the double bonded oxygen of the other substituent, carboxylic acid, through hydrogen bonding thus decreasing its polarity sig ...
Supplementary Information
... explained (44). For instance, NH2COH has been produced in the past by reaction of ammonia and formic acid. While ammonia is generally accepted to be a major component of the primeval atmosphere (45), formic acid is the most abundant product formed in the classical Miller–Urey experiment (46). At dif ...
... explained (44). For instance, NH2COH has been produced in the past by reaction of ammonia and formic acid. While ammonia is generally accepted to be a major component of the primeval atmosphere (45), formic acid is the most abundant product formed in the classical Miller–Urey experiment (46). At dif ...
Kjeldahl Method for Determination of Nitrogen
... of acid by organic material and vaporization, salt/acid ratio, digestion length, and physical design of the Kjeldahl flask, are all interrelated. Each has an effect on the final solution temperature. A second precaution is that if the salt/acid ratio is too high, a considerable amount of material wi ...
... of acid by organic material and vaporization, salt/acid ratio, digestion length, and physical design of the Kjeldahl flask, are all interrelated. Each has an effect on the final solution temperature. A second precaution is that if the salt/acid ratio is too high, a considerable amount of material wi ...
Molecular Modeling of Hydrophobic Organic Contaminants
... • Most of the scientific and technological challenges associated with the production and processing of heavy oils is directly related to their high content of non-volatile and refractory compounds such as asphaltenes. ...
... • Most of the scientific and technological challenges associated with the production and processing of heavy oils is directly related to their high content of non-volatile and refractory compounds such as asphaltenes. ...
Introduction - Bulgarian Chemical Communications
... reaction to a reference one and their values are not confined in the limits from zero to unity. The means of calibrating ρ in terms of a Leffler equation have been discussed by A. Williams [7]. Seventy years after its conception as a quantitative description of polar effects, the Hammett equation an ...
... reaction to a reference one and their values are not confined in the limits from zero to unity. The means of calibrating ρ in terms of a Leffler equation have been discussed by A. Williams [7]. Seventy years after its conception as a quantitative description of polar effects, the Hammett equation an ...
Chapter 15 Acids and Bases
... does not explain why molecular substances, like NH3, dissolve in water to form basic solutions – even though they do not contain OH– ions does not explain how some ionic compounds, like Na2CO3 or Na2O, dissolve in water to form basic solutions – even though they do not contain OH– ions does no ...
... does not explain why molecular substances, like NH3, dissolve in water to form basic solutions – even though they do not contain OH– ions does not explain how some ionic compounds, like Na2CO3 or Na2O, dissolve in water to form basic solutions – even though they do not contain OH– ions does no ...
1. (a) Propan-1ol, C2H5CH2OH can be oxidised to propanoic acid
... to alleviate sunburn. Benzocaine reacts with dilute acids to form the ion C9H12O2N+ and with ethanoyl chloride to form C11H13O3N. When benzocaine is heated under reflux with aqueous sodium hydroxide and the solution obtained is neutralised, two compounds X and Y are formed. X has a formula of ...
... to alleviate sunburn. Benzocaine reacts with dilute acids to form the ion C9H12O2N+ and with ethanoyl chloride to form C11H13O3N. When benzocaine is heated under reflux with aqueous sodium hydroxide and the solution obtained is neutralised, two compounds X and Y are formed. X has a formula of ...
p Block Elements General Configuration: ns2 np1
... Phosphine is weakly basic and gives phosphonium compounds with acids e.g., ...
... Phosphine is weakly basic and gives phosphonium compounds with acids e.g., ...
Acids, Bases, and pH
... health (Vitamin C is a compound called ascorbic acid), and many other aspects of chemistry. In this mini-chapter you will learn the basics of acids and bases and how they are related to pH. I. Acids An Acid is a substance that donates one or more H+ ions (protons) to another substance (called a base ...
... health (Vitamin C is a compound called ascorbic acid), and many other aspects of chemistry. In this mini-chapter you will learn the basics of acids and bases and how they are related to pH. I. Acids An Acid is a substance that donates one or more H+ ions (protons) to another substance (called a base ...
chemistry advanced may 2010 marking scheme
... (d) Consider the following hydrides: CH4, NH3 and H2S. (i) State and explain briefly the acid–base properties (if any) of the substances. CH4 is neutral since it does not accept or donate protons. (0.5) NH3 is a base since it accepts protons to form NH4+. (0.5) H2S is an acid donating a proton to f ...
... (d) Consider the following hydrides: CH4, NH3 and H2S. (i) State and explain briefly the acid–base properties (if any) of the substances. CH4 is neutral since it does not accept or donate protons. (0.5) NH3 is a base since it accepts protons to form NH4+. (0.5) H2S is an acid donating a proton to f ...
File - Fidaa`s Level 2 Portfolio
... topic, but in reality it is huge. Chemicals are the base of chemistry. They lead the subject into deeper aspects that pass on more knowledge. Part of the deeper aspects of chemistry is called organic chemistry. Organic chemistry is all about chemistry related to the element carbon. Organic chemistry ...
... topic, but in reality it is huge. Chemicals are the base of chemistry. They lead the subject into deeper aspects that pass on more knowledge. Part of the deeper aspects of chemistry is called organic chemistry. Organic chemistry is all about chemistry related to the element carbon. Organic chemistry ...
Acids and Bases
... General rule: The conjugate base of a strong acid is a weak base. Similarly, the conjugate acid of a strong base is a weak acid. ...
... General rule: The conjugate base of a strong acid is a weak base. Similarly, the conjugate acid of a strong base is a weak acid. ...
Acid K a
... Kb value is directly related to base strength. How to write out Ka and Kb rxns and expressions. The weaker the acid the stronger the conjugate base (and vice versa). Conjugate bases of strong acids have no basic ...
... Kb value is directly related to base strength. How to write out Ka and Kb rxns and expressions. The weaker the acid the stronger the conjugate base (and vice versa). Conjugate bases of strong acids have no basic ...
2013-2014
... 13. Which of the following are the differences between a well-used zinc-carbon cell and a new one? (1) The well-used one has a thinner zinc cup than the new one. (2) The voltage of the well-used one is lower than that of the new one. (3) The graphite rod of the well-used one is wetter than that of t ...
... 13. Which of the following are the differences between a well-used zinc-carbon cell and a new one? (1) The well-used one has a thinner zinc cup than the new one. (2) The voltage of the well-used one is lower than that of the new one. (3) The graphite rod of the well-used one is wetter than that of t ...
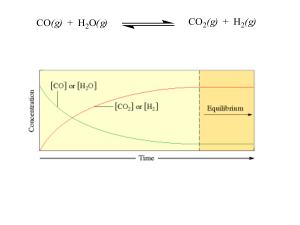
E2 and E11 Acid Deposition Past Paper Questions
... 1CO2(g) + H2O(l) H2CO3(aq) OR CO2(g) + H2O(l) 2 H (aq) + HCO3 (aq) coal contains sulfur (which burns to form SO2) S(s) + O2(g) SO2(g) SO2(g) + H2O(l) H2SO3(aq) OR … 2SO2(g) + O2(g) 2SO3(g) SO3(g) + H2O(l) H2SO4 (aq ) ...
... 1CO2(g) + H2O(l) H2CO3(aq) OR CO2(g) + H2O(l) 2 H (aq) + HCO3 (aq) coal contains sulfur (which burns to form SO2) S(s) + O2(g) SO2(g) SO2(g) + H2O(l) H2SO3(aq) OR … 2SO2(g) + O2(g) 2SO3(g) SO3(g) + H2O(l) H2SO4 (aq ) ...
acids - WordPress.com
... 2. Acids are soluble in water and give solutions with a pH below 7. They give charatcteristic colours with indicators.They turn litmus red. 3. The concentrated forms of strong acids like hydrochloric acid and sulphuric acid are corrosive. 4. Aqueous solutions of acids are able to conduct electricity ...
... 2. Acids are soluble in water and give solutions with a pH below 7. They give charatcteristic colours with indicators.They turn litmus red. 3. The concentrated forms of strong acids like hydrochloric acid and sulphuric acid are corrosive. 4. Aqueous solutions of acids are able to conduct electricity ...
Topic 14 - Fertilisers
... However, nitrogen is un-reactive but not inert. This means it is difficult getting it to react but it can be done (using electricity i.e. lightening or a spark plug). The nitrogen will form oxides which dissolve in water forming acids. Nitrogen dioxide, a brown gas, can be made when air (21% oxygen ...
... However, nitrogen is un-reactive but not inert. This means it is difficult getting it to react but it can be done (using electricity i.e. lightening or a spark plug). The nitrogen will form oxides which dissolve in water forming acids. Nitrogen dioxide, a brown gas, can be made when air (21% oxygen ...
Diazotization-Coupling Reaction--
... Azote is an old word for nitrogen. Hence, the presence of azo in the name of a chemical implies that nitrogen is present in the structure. Therefore, diazo means two nitrogen atoms. When combined with onium, we have diazonium, which means two nitrogen atoms and a positive charge (i.e., N2+). Diazoni ...
... Azote is an old word for nitrogen. Hence, the presence of azo in the name of a chemical implies that nitrogen is present in the structure. Therefore, diazo means two nitrogen atoms. When combined with onium, we have diazonium, which means two nitrogen atoms and a positive charge (i.e., N2+). Diazoni ...
A NOVEL BIOCHEMICAL METHOD FOR PRODUCTION OF AN ANTIBACTERIAL DRUG
... Objectives: Production of an antibacterial drug trimethoprim from industrial wastes by using novel biochemical methods. Methods: A few reports are available in the literature on the production of Trimethoprim through biotechnological routes. A novel route for the synthesis of trimethoprim using the ...
... Objectives: Production of an antibacterial drug trimethoprim from industrial wastes by using novel biochemical methods. Methods: A few reports are available in the literature on the production of Trimethoprim through biotechnological routes. A novel route for the synthesis of trimethoprim using the ...
Lab 13
... Azote is an old word for nitrogen. Hence, the presence of azo in the name of a chemical implies that nitrogen is present in the structure. Therefore, diazo means two nitrogen atoms. When combined with onium, we have diazonium, which means two nitrogen atoms and a positive charge (i.e., N2+). Diazoni ...
... Azote is an old word for nitrogen. Hence, the presence of azo in the name of a chemical implies that nitrogen is present in the structure. Therefore, diazo means two nitrogen atoms. When combined with onium, we have diazonium, which means two nitrogen atoms and a positive charge (i.e., N2+). Diazoni ...
Nitric acid

Nitric acid (HNO3), also known as aqua fortis and spirit of niter, is a highly corrosive mineral acid.The pure compound is colorless, but older samples tend to acquire a yellow cast due to decomposition into oxides of nitrogen and water. Most commercially available nitric acid has a concentration of 68%. When the solution contains more than 86% HNO3, it is referred to as fuming nitric acid. Depending on the amount of nitrogen dioxide present, fuming nitric acid is further characterized as white fuming nitric acid or red fuming nitric acid, at concentrations above 95%.Nitric acid is the primary reagent used for nitration – the addition of a nitro group, typically to an organic molecule. While some resulting nitro compounds are shock- and thermally-sensitive explosives, a few are stable enough to be used in munitions and demolition, while others are still more stable and used as pigments in inks and dyes. Nitric acid is also commonly used as a strong oxidizing agent.