chapter 15 acids and bases
... (b) − (c) are worked in a similar manner to part (a). However, as the initial concentration of HF becomes smaller, the assumption that x is very small compared to this concentration will no longer be valid. You must solve a quadratic equation. (b) ...
... (b) − (c) are worked in a similar manner to part (a). However, as the initial concentration of HF becomes smaller, the assumption that x is very small compared to this concentration will no longer be valid. You must solve a quadratic equation. (b) ...
85 Q.2 Pure water has a low electricity conductivity because A. it
... 90 Q.17 Zinc does NOT liberate hydrogen from a 0.5M nitric acid solution because A. zinc is an unreactive metal. B. nitric acid is an oxidizing agent. C. the concentration of hydrogen ions in the nitric acid is too low. D. a layer of oxide film is formed on the surface of zinc to prevent further rea ...
... 90 Q.17 Zinc does NOT liberate hydrogen from a 0.5M nitric acid solution because A. zinc is an unreactive metal. B. nitric acid is an oxidizing agent. C. the concentration of hydrogen ions in the nitric acid is too low. D. a layer of oxide film is formed on the surface of zinc to prevent further rea ...
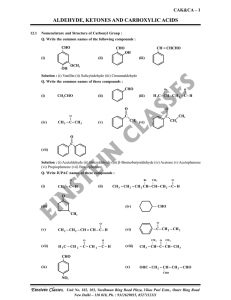
aldehyde, ketones and carboxylic acids
... Q. When ketnoes and oxidised with strong oxidant than identify the product formed. Solution : Ketones with strong oxidants and at high temperature undergo cleavage of C-C bond on either side of carbonyl group. ...
... Q. When ketnoes and oxidised with strong oxidant than identify the product formed. Solution : Ketones with strong oxidants and at high temperature undergo cleavage of C-C bond on either side of carbonyl group. ...
2014_S4_CHM_NORMAL (ALL)
... 53. Element X (atomic number 11) reacts with element Y (atomic number 16) to form an ionic compound. Each atom of X loses one electron and each atom of Y accepts two electrons to form a compound with formula X2Y. 54. Consider the following information: ...
... 53. Element X (atomic number 11) reacts with element Y (atomic number 16) to form an ionic compound. Each atom of X loses one electron and each atom of Y accepts two electrons to form a compound with formula X2Y. 54. Consider the following information: ...
Equilibrium Notes - Chemistry Teaching Resources
... demonstrated in the laboratory using the following example: N 2 O 4 (g) yellow ...
... demonstrated in the laboratory using the following example: N 2 O 4 (g) yellow ...
Journal of Molecular Catalysis A, 302
... Pd favors the second process, and the yields of benzoyl fluoride and benzoic acid then exceeds the yield of trifluoromethylbenzoic acid produced. On the other hand, with no vanadium or at extremely low vanadium concentrations, the reduced Rh or Pd cations cannot be re-oxidized sufficiently rapidly, res ...
... Pd favors the second process, and the yields of benzoyl fluoride and benzoic acid then exceeds the yield of trifluoromethylbenzoic acid produced. On the other hand, with no vanadium or at extremely low vanadium concentrations, the reduced Rh or Pd cations cannot be re-oxidized sufficiently rapidly, res ...
PRACTICAL ORGANIC CHEMISTRY
... acid and if necessary heat gently, then add another drop of acid, warm and finally add 1 ml of conc. sulphuric acid, then proceed as usual. Notes on nitration: 1. some aromatic compound are not nitrated under the above experimental conditions (e.g. polyhydroxy compound: resorcinol, ...
... acid and if necessary heat gently, then add another drop of acid, warm and finally add 1 ml of conc. sulphuric acid, then proceed as usual. Notes on nitration: 1. some aromatic compound are not nitrated under the above experimental conditions (e.g. polyhydroxy compound: resorcinol, ...
Acid - Net Texts
... Common examples of acids include acetic acid (in vinegar), sulfuric acid (used in car batteries), and tartaric acid (used in baking). As these Zinc, a typical metal, reacting with hydrochloric three examples show, acids can be solutions, liquids, or solids. Gases acid, a typical acid such as hydroge ...
... Common examples of acids include acetic acid (in vinegar), sulfuric acid (used in car batteries), and tartaric acid (used in baking). As these Zinc, a typical metal, reacting with hydrochloric three examples show, acids can be solutions, liquids, or solids. Gases acid, a typical acid such as hydroge ...
Acidic Environment
... Then the hydrogen ion reacts with water, ie this reaction occurs: H+ + H2O ...
... Then the hydrogen ion reacts with water, ie this reaction occurs: H+ + H2O ...
Physical properties study for TBP-HNO3-diluent system
... Kumar et al. (2011) have estimated PVT properties of TBP using group contribution method and from the obtained data Wagner constants for TBP in the range of 273.15K to critical temperature have been calculated. Velavendan et al. (2012) provided a complete data on solubility of 5 %, 20 %, 30 % and 10 ...
... Kumar et al. (2011) have estimated PVT properties of TBP using group contribution method and from the obtained data Wagner constants for TBP in the range of 273.15K to critical temperature have been calculated. Velavendan et al. (2012) provided a complete data on solubility of 5 %, 20 %, 30 % and 10 ...
Acidic Environment by Ahmad Shah Idil
... 2. If more water is added the equilibrium will shift to the LEFT, as more Cu(H2O)42+ is formed, and the system will become more blue. 3. If water is removed the equilibrium will shift to the RIGHT, so that the water lost will be replaced by the forward reaction. ...
... 2. If more water is added the equilibrium will shift to the LEFT, as more Cu(H2O)42+ is formed, and the system will become more blue. 3. If water is removed the equilibrium will shift to the RIGHT, so that the water lost will be replaced by the forward reaction. ...
Unit 11 acids and bases part 1
... This new bond is called a coordinate covalent bond since both new bonding electrons come from the same atom ...
... This new bond is called a coordinate covalent bond since both new bonding electrons come from the same atom ...
Kinetics of Oxidation of Benzyl Alcohol with Dilute Nitric Acid
... requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited cases, good yields have been obtained, with respec ...
... requires a heterogeneous catalyst, high temperature (210 °C), and longer reaction times (>5 h). Although the reported selectivity is quite good (75%-95%), the conversion levels are too low (maximum of 40%).14 Practically, in all the previously cited cases, good yields have been obtained, with respec ...
Structural Studies on Sulfated Glycopeptides from the Carbohydrate
... and 5 mM cysteine-HC1. The digestion was performed for 45 h at 63 ‘C; additional 0.8 mg of papain and 1.8 mg of cysteine-HC1 were added after 22 and 34 h. Following incubation the sample was lyophilized, mixed with 1.0 ml of5% trichloroacetic acid and centrifuged in a Beckman microcentrifuge for 10 ...
... and 5 mM cysteine-HC1. The digestion was performed for 45 h at 63 ‘C; additional 0.8 mg of papain and 1.8 mg of cysteine-HC1 were added after 22 and 34 h. Following incubation the sample was lyophilized, mixed with 1.0 ml of5% trichloroacetic acid and centrifuged in a Beckman microcentrifuge for 10 ...
CHAPTER 15 ACIDS AND BASES
... Without doing any calculations, could you have known that the pH of the sulfuric acid would be lower (more acidic) than that of the hydrochloric acid? ...
... Without doing any calculations, could you have known that the pH of the sulfuric acid would be lower (more acidic) than that of the hydrochloric acid? ...
Acid + Base Class # 1
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
... 20. Strong acids & strong bases have lots of ions in solution. Strong acids & strong bases are good electrolytes, they will conduct electricity well because of all the loose ions they have in solution. 21. All acids and all bases are electrolytes. 22. Their electrolyte strengths (their ability to c ...
Acids - Beck-Shop
... It is important to realise that not all compounds that contain hydrogen atoms are acids. Each molecule of ethanoic acid contains four hydrogen atoms, but only the hydrogen atom on the COOH group is released as H+. Even then, only about one molecule in every hundred dissociates, so ethanoic acid is a ...
... It is important to realise that not all compounds that contain hydrogen atoms are acids. Each molecule of ethanoic acid contains four hydrogen atoms, but only the hydrogen atom on the COOH group is released as H+. Even then, only about one molecule in every hundred dissociates, so ethanoic acid is a ...
Chapter Ten
... ► Sodium hydroxide, NaOH, or lye, is used in the production of aluminum, glass, and soap. Drain cleaners often contain NaOH because it reacts with the fats and proteins found in grease and hair. ► Calcium hydroxide, Ca(OH)2 , or slaked lime, is made industrially by treating lime (CaO) with water. I ...
... ► Sodium hydroxide, NaOH, or lye, is used in the production of aluminum, glass, and soap. Drain cleaners often contain NaOH because it reacts with the fats and proteins found in grease and hair. ► Calcium hydroxide, Ca(OH)2 , or slaked lime, is made industrially by treating lime (CaO) with water. I ...
2. 2-Isopropyl-5-methylcyclohexanol on carbon skeletal
... 116. 3-methylbutanol - 2 can be prepared by reduction of a compound: 1. 3-metilbutanaly; 2. 3-metilpentanal; 3. 3-methylbutanol-2; 4. 2-methylbutanol-3; 117. Hydroxide copper (II) in an alkaline solution under heating does not oxidize oxo compounds following: 1. formaldehyde; 2. propanaly; 3. Aceton ...
... 116. 3-methylbutanol - 2 can be prepared by reduction of a compound: 1. 3-metilbutanaly; 2. 3-metilpentanal; 3. 3-methylbutanol-2; 4. 2-methylbutanol-3; 117. Hydroxide copper (II) in an alkaline solution under heating does not oxidize oxo compounds following: 1. formaldehyde; 2. propanaly; 3. Aceton ...
Buffer Solutions
... They play an important role in chemical processes where it is essential that a fairly constant pH is maintained. In many industrial and physiological processes, specific reactions occur at some optimum pH value. When the pH varies to any extent from the optimum value, undesirable reactions and effec ...
... They play an important role in chemical processes where it is essential that a fairly constant pH is maintained. In many industrial and physiological processes, specific reactions occur at some optimum pH value. When the pH varies to any extent from the optimum value, undesirable reactions and effec ...
Buffer Solutions
... They play an important role in chemical processes where it is essential that a fairly constant pH is maintained. In many industrial and physiological processes, specific reactions occur at some optimum pH value. When the pH varies to any extent from the optimum value, undesirable reactions and effec ...
... They play an important role in chemical processes where it is essential that a fairly constant pH is maintained. In many industrial and physiological processes, specific reactions occur at some optimum pH value. When the pH varies to any extent from the optimum value, undesirable reactions and effec ...
664
... Any nitric oxide produced in small amounts may be removed by passage through FeSO4 solution. Alternatively, the gas may be obtained by warming a solution of ammonium nitrate in nitric acid containing a small amount of hydrochloric acid. Pure nitrous oxide may be prepared by treating a solution of so ...
... Any nitric oxide produced in small amounts may be removed by passage through FeSO4 solution. Alternatively, the gas may be obtained by warming a solution of ammonium nitrate in nitric acid containing a small amount of hydrochloric acid. Pure nitrous oxide may be prepared by treating a solution of so ...
Chapter 17, Section 17.3
... Chapter 17, Section 17.3 • Strong acids have large Ka; weak acids have very small Ka • Ka’s are present in your acid-base chart on pages 8-9 of the Data Booklet • % ionizations are valid for acids only over a very narrow concentration range; Ka’s better but ………. You may be required to calculate % i ...
... Chapter 17, Section 17.3 • Strong acids have large Ka; weak acids have very small Ka • Ka’s are present in your acid-base chart on pages 8-9 of the Data Booklet • % ionizations are valid for acids only over a very narrow concentration range; Ka’s better but ………. You may be required to calculate % i ...
Density functional theory and FTIR spectroscopic study of carboxyl
... cellulose acetate. The negative sign indicates that the vibrational spectrum is calculated for the transition state not for the optimum structure. Accordingly, the vibrational spectra of cellulose acetate is not considered at this level of theory. The effect of six monovalent alkalis on COOH vibrati ...
... cellulose acetate. The negative sign indicates that the vibrational spectrum is calculated for the transition state not for the optimum structure. Accordingly, the vibrational spectra of cellulose acetate is not considered at this level of theory. The effect of six monovalent alkalis on COOH vibrati ...
Nitric acid

Nitric acid (HNO3), also known as aqua fortis and spirit of niter, is a highly corrosive mineral acid.The pure compound is colorless, but older samples tend to acquire a yellow cast due to decomposition into oxides of nitrogen and water. Most commercially available nitric acid has a concentration of 68%. When the solution contains more than 86% HNO3, it is referred to as fuming nitric acid. Depending on the amount of nitrogen dioxide present, fuming nitric acid is further characterized as white fuming nitric acid or red fuming nitric acid, at concentrations above 95%.Nitric acid is the primary reagent used for nitration – the addition of a nitro group, typically to an organic molecule. While some resulting nitro compounds are shock- and thermally-sensitive explosives, a few are stable enough to be used in munitions and demolition, while others are still more stable and used as pigments in inks and dyes. Nitric acid is also commonly used as a strong oxidizing agent.