Aligned AlN Nanorods with Multi-tipped Surfaces
... substrate is higher than the best value found for carbon nanotubes[13] and SiC,[14] they are much lower than those of many other types of emitters such as carbon nitride,[15] Si nanostructures,[16] MoO3 nanobelts,[17] and ZnO nanowires.[18] In a previous study, the selection of the substrate was fou ...
... substrate is higher than the best value found for carbon nanotubes[13] and SiC,[14] they are much lower than those of many other types of emitters such as carbon nitride,[15] Si nanostructures,[16] MoO3 nanobelts,[17] and ZnO nanowires.[18] In a previous study, the selection of the substrate was fou ...
Lecture on DFT in English is available on the web page
... Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . But Hamiltionian contains only one- and two-electron operators, since electrons don’t have internal structure (n ...
... Wavefunction is a fairly complicated object! For N -electron system it depends on 4N variables. For systems of biological importance this may boil down to several thousand variables . . . But Hamiltionian contains only one- and two-electron operators, since electrons don’t have internal structure (n ...
Noncovalent interactions of molecules with single walled carbon
... was suggested that an energy correction of 1–2% for curvature could be expected, based on similar corrections for curvatureinduced changes in electronic structure,33 though p orbital misalignment may introduce larger differences in binding energy.31 Often, rather than using a discrete potential, a c ...
... was suggested that an energy correction of 1–2% for curvature could be expected, based on similar corrections for curvatureinduced changes in electronic structure,33 though p orbital misalignment may introduce larger differences in binding energy.31 Often, rather than using a discrete potential, a c ...
Question Bank (Class XI - Chemistry)
... Q3- What is a pure substance? (L- 1 ) Ans. A substance which contains only one kind of atom or molecule is called a pure substance . Q4- Define average atomic mass. (L-1) Ans. Average atomic mass is the average of atomic mass of all the isotopes of an element. Q5- What is one a.m.u. or one ‘u ,? (L- ...
... Q3- What is a pure substance? (L- 1 ) Ans. A substance which contains only one kind of atom or molecule is called a pure substance . Q4- Define average atomic mass. (L-1) Ans. Average atomic mass is the average of atomic mass of all the isotopes of an element. Q5- What is one a.m.u. or one ‘u ,? (L- ...
Introduction - St. Olaf College
... c) Measure Dihedral: This measures the angle formed by two intersecting planes, one containing the first three atoms selected and the other containing the last three atoms selected. Click once on to move to the next molecule, hydrogen peroxide, then select Measure Dihedral from the Geometry menu (o ...
... c) Measure Dihedral: This measures the angle formed by two intersecting planes, one containing the first three atoms selected and the other containing the last three atoms selected. Click once on to move to the next molecule, hydrogen peroxide, then select Measure Dihedral from the Geometry menu (o ...
5 How Chemists Measure Atoms and Molecules
... was done for the marbles.2 In the 1800s chemists, such as John Dalton, were able to assign relative masses, called atomic weights, to atoms. They did this by comparing the masses of two elements that react with each other. Dalton chose hydrogen (assumed to have an atomic weight of 1) as the referenc ...
... was done for the marbles.2 In the 1800s chemists, such as John Dalton, were able to assign relative masses, called atomic weights, to atoms. They did this by comparing the masses of two elements that react with each other. Dalton chose hydrogen (assumed to have an atomic weight of 1) as the referenc ...
5 How Chemists Measure Atoms and Molecules
... was done for the marbles.2 In the 1800s chemists, such as John Dalton, were able to assign relative masses, called atomic weights, to atoms. They did this by comparing the masses of two elements that react with each other. Dalton chose hydrogen (assumed to have an atomic weight of 1) as the referenc ...
... was done for the marbles.2 In the 1800s chemists, such as John Dalton, were able to assign relative masses, called atomic weights, to atoms. They did this by comparing the masses of two elements that react with each other. Dalton chose hydrogen (assumed to have an atomic weight of 1) as the referenc ...
THE RISE OF GRAPHENE A.K. Geim and K.S. Novoselov
... Before reviewing the earlier work on graphene, it is useful to define what 2D crystals are. Obviously, a single atomic plane is a 2D crystal, whereas 100 layers should be considered as a thin film of a 3D material. But how many layers are needed to make a 3D structure? For the case of graphene, the ...
... Before reviewing the earlier work on graphene, it is useful to define what 2D crystals are. Obviously, a single atomic plane is a 2D crystal, whereas 100 layers should be considered as a thin film of a 3D material. But how many layers are needed to make a 3D structure? For the case of graphene, the ...
Section 1.3 - The Student Room
... liquid or solid. In a gas, therefore, the volume of the particles is a very small part of the total volume and does not significantly affect it. In a liquid or solid the particles are close together and their volumes must be taken into account when deciding on the total volume. a CH4(g) + 2O2(g) Æ C ...
... liquid or solid. In a gas, therefore, the volume of the particles is a very small part of the total volume and does not significantly affect it. In a liquid or solid the particles are close together and their volumes must be taken into account when deciding on the total volume. a CH4(g) + 2O2(g) Æ C ...
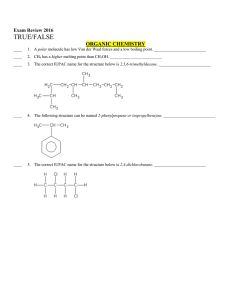
Multiple Choice Exam Review June 2016
... ____ 10. All alkali earth metals have valence electrons in s orbitals when they are in their ground state. ____________________ ____ 11. The 2p orbitals in oxygen have three unpaired electrons. ____________________ ____ 12. The shape of SO2 is trigonal planar. ____________________ ____ 13. The valen ...
... ____ 10. All alkali earth metals have valence electrons in s orbitals when they are in their ground state. ____________________ ____ 11. The 2p orbitals in oxygen have three unpaired electrons. ____________________ ____ 12. The shape of SO2 is trigonal planar. ____________________ ____ 13. The valen ...
Quantum Computer (Information) and Quantum Mechanical
... beings can perceive, this quantum mechanical behavior is normally hidden (not perceived-e(e)) by us. That it is not in the visible field; We cannot see it let alone decipher the progressive movements and dynamics of the system. Yet, if we were to look into the world of individual atoms, we would fin ...
... beings can perceive, this quantum mechanical behavior is normally hidden (not perceived-e(e)) by us. That it is not in the visible field; We cannot see it let alone decipher the progressive movements and dynamics of the system. Yet, if we were to look into the world of individual atoms, we would fin ...
Superoxide and Singlet Oxygen Produced within
... Photoinhibition1[OPEN] Daisuke Takagi, Shigeo Takumi, Masaki Hashiguchi, Takehiro Sejima, and Chikahiro Miyake* Department of Biological and Environmental Science, Graduate School of Agricultural Science, Kobe University, 1-1 Rokkodai, Nada, Kobe 657-8501, Japan (D.T., S.T., M.H., T.S., C.M.); and C ...
... Photoinhibition1[OPEN] Daisuke Takagi, Shigeo Takumi, Masaki Hashiguchi, Takehiro Sejima, and Chikahiro Miyake* Department of Biological and Environmental Science, Graduate School of Agricultural Science, Kobe University, 1-1 Rokkodai, Nada, Kobe 657-8501, Japan (D.T., S.T., M.H., T.S., C.M.); and C ...
content - Thesis Scientist
... (ii) The transition metals and many of their compounds act as good catalysts. (iii) From element to element the actinoid contraction is greater than the lanthanoid contraction. (iv) The E° value for the Mn 3+ / Mn 2+ couple is much more positive than that of Cr 3+ / Cr 2+ . (v) Scandium ( Z = 21) do ...
... (ii) The transition metals and many of their compounds act as good catalysts. (iii) From element to element the actinoid contraction is greater than the lanthanoid contraction. (iv) The E° value for the Mn 3+ / Mn 2+ couple is much more positive than that of Cr 3+ / Cr 2+ . (v) Scandium ( Z = 21) do ...
Individual Trapped Atoms for Cavity QED Quantum
... cooling beams in an optical lattice. This non-destructive imaging technique led to atomic storage times of two minutes in an optical lattice. The second part of thesis incorporated the individual atoms into a high finesse cavity. Inside this optical cavity, atoms are cooled and non-destructively obs ...
... cooling beams in an optical lattice. This non-destructive imaging technique led to atomic storage times of two minutes in an optical lattice. The second part of thesis incorporated the individual atoms into a high finesse cavity. Inside this optical cavity, atoms are cooled and non-destructively obs ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.