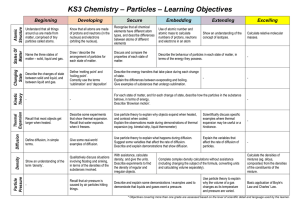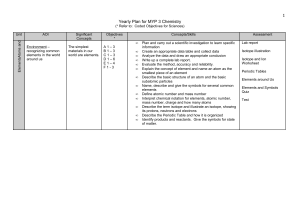
Yearly Plan for MYP 1 Science
... Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an atom and the basic subatomic particles Name, descr ...
... Analyze the data and draw an appropriate conclusion Write up a complete lab report. Evaluate the method, accuracy and reliability. Explain the concept of element and name an atom as the smallest piece of an element Describe the basic structure of an atom and the basic subatomic particles Name, descr ...
Physical Chemistry II – Exam 3 Solutions
... Note that this expectation value makes sense, because it corresponds to the Coulomb potential of interaction between the nucleus and an electron at a distance a0 , which is the ...
... Note that this expectation value makes sense, because it corresponds to the Coulomb potential of interaction between the nucleus and an electron at a distance a0 , which is the ...
Electrons in Atoms
... • Analysis of the emitted light revealed that an element’s chemical behavior is related to the arrangement of the electrons in its atoms. • In order for you to better understand this relationship and the nature of atomic structure, it will be helpful for you to first understand the nature of light. ...
... • Analysis of the emitted light revealed that an element’s chemical behavior is related to the arrangement of the electrons in its atoms. • In order for you to better understand this relationship and the nature of atomic structure, it will be helpful for you to first understand the nature of light. ...
Elements Compounds Mixtures
... • Atomic #--# of protons of an element. It identifies the element. • A#= #P • Mass # - This is the # that is = to the # of protons and the neutrons in the nucleus. ...
... • Atomic #--# of protons of an element. It identifies the element. • A#= #P • Mass # - This is the # that is = to the # of protons and the neutrons in the nucleus. ...
Webquest Review - Harrison High School
... 19. Is carbon dioxide a polar molecule? Draw the structure. CO2 is nonpolar. You have two oxygens, each double bonded to the carbon. These two identical bonds in a symmetrical species, pull in equal yet opposite directions, cancelling each other out. E: View “Intermolecular Forces.” 20. Explain why ...
... 19. Is carbon dioxide a polar molecule? Draw the structure. CO2 is nonpolar. You have two oxygens, each double bonded to the carbon. These two identical bonds in a symmetrical species, pull in equal yet opposite directions, cancelling each other out. E: View “Intermolecular Forces.” 20. Explain why ...
Chapter 4_Chemical Bonding and Molecular Structure
... A chemical bond formation is attributed to the tendency of a system to attain stability. It was observed that the inertness of noble gases was because of their fully filled outermost orbitals. Hence, it was postulated that the elements having incomplete outermost shells are unstable (reactive). Atom ...
... A chemical bond formation is attributed to the tendency of a system to attain stability. It was observed that the inertness of noble gases was because of their fully filled outermost orbitals. Hence, it was postulated that the elements having incomplete outermost shells are unstable (reactive). Atom ...
Particles - Townley Grammar School
... Know that all atoms are made of protons and neutrons (in the nucleus) and electrons (orbiting the nucleus). ...
... Know that all atoms are made of protons and neutrons (in the nucleus) and electrons (orbiting the nucleus). ...
Slide 1
... sent to three different laboratories, operated by Alice, Bob, and Claire. For simplicity, we assume the particles are not identical. The spin state of the three particles is ...
... sent to three different laboratories, operated by Alice, Bob, and Claire. For simplicity, we assume the particles are not identical. The spin state of the three particles is ...
Chapter 1: Matter, Measurement and Problem Solving
... Question: Oil floats on water is it more or less dense than ...
... Question: Oil floats on water is it more or less dense than ...
Chapter 30: Quantum Physics
... 68. Picture the Problem: An electron and proton have the same de Broglie wavelength, which means that they must also have the same momentum. However, their kinetic energies will differ because they have different masses. Strategy: Use Equation 30-16 to write the kinetic energy in terms of the de Bro ...
... 68. Picture the Problem: An electron and proton have the same de Broglie wavelength, which means that they must also have the same momentum. However, their kinetic energies will differ because they have different masses. Strategy: Use Equation 30-16 to write the kinetic energy in terms of the de Bro ...
Timeline of chemistry
... history of chemistry in its modern form is often considered to begin with the English scientist Robert Boyle, though its roots can be traced back to the earliest recorded history. Early ideas that later became incorporated into the modern science of chemistry come from two main sources. Natural phil ...
... history of chemistry in its modern form is often considered to begin with the English scientist Robert Boyle, though its roots can be traced back to the earliest recorded history. Early ideas that later became incorporated into the modern science of chemistry come from two main sources. Natural phil ...
Solution
... = 1.83 x 1083, this is a very large K indicating that the products are strongly favored. This is consistent with the negative free energy of part (c). e) The pressure of oxygen is 5 atm and the pressure of hydrogen is 10 atm at 25°C. In which direction will the reaction shift in order to regain equi ...
... = 1.83 x 1083, this is a very large K indicating that the products are strongly favored. This is consistent with the negative free energy of part (c). e) The pressure of oxygen is 5 atm and the pressure of hydrogen is 10 atm at 25°C. In which direction will the reaction shift in order to regain equi ...
Chemistry Final Exam Review 2006-2007
... nature as a diatomic molecule? b. 3 a. Nitrogen c. 4 b. Helium d. 2 c. Hydrogen 11. In the correct Lewis structure for the methane d. oxygen molecule, how many unshared electron pairs 2. Ionic compounds generally form: surround the carbon? a. Liquids a. 2 b. Gases b. 0 c. Crystals c. 8 d. molecules ...
... nature as a diatomic molecule? b. 3 a. Nitrogen c. 4 b. Helium d. 2 c. Hydrogen 11. In the correct Lewis structure for the methane d. oxygen molecule, how many unshared electron pairs 2. Ionic compounds generally form: surround the carbon? a. Liquids a. 2 b. Gases b. 0 c. Crystals c. 8 d. molecules ...
Lecture 6: 3D Rigid Rotor, Spherical Harmonics, Angular Momentum
... with the restriction that l = 0, 1, 2, .. and ml = 0, ±1, ±2, ... ± l give a degeneracy of 2l + 1 for each energy level. If these states are states corresponding to an electron orbiting around a nucleus, they become nondegenerate in the presence of a magnetic field which couples to the angular momen ...
... with the restriction that l = 0, 1, 2, .. and ml = 0, ±1, ±2, ... ± l give a degeneracy of 2l + 1 for each energy level. If these states are states corresponding to an electron orbiting around a nucleus, they become nondegenerate in the presence of a magnetic field which couples to the angular momen ...
Nuclear Forces and Mesons
... 2. Attractive at the distances > 0.6 fm – that's what binds the nucleons together in a nucleus. 3. Strongly repulsive at short distances of < 0.5 fm – that's why nuclear matter is highly incompressible, and this causes the outward “bounce” of the shock wave in a core-collapse ...
... 2. Attractive at the distances > 0.6 fm – that's what binds the nucleons together in a nucleus. 3. Strongly repulsive at short distances of < 0.5 fm – that's why nuclear matter is highly incompressible, and this causes the outward “bounce” of the shock wave in a core-collapse ...
Kinetic Energy and the Covalent Bond in H2
... the fundamental level there is only one theory of binding in H2 , and that corresponds to an exact solution of the Schrödinger equation” [14]. However, as Robert Mulliken observed, “the more accurate the calculations became the more the concepts tended to vanish into thin air” [15]. Therefore, we ex ...
... the fundamental level there is only one theory of binding in H2 , and that corresponds to an exact solution of the Schrödinger equation” [14]. However, as Robert Mulliken observed, “the more accurate the calculations became the more the concepts tended to vanish into thin air” [15]. Therefore, we ex ...
Electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is 1s2 2s2 2p6.Electronic configurations describe electrons as each moving independently in an orbital, in an average field created by all other orbitals. Mathematically, configurations are described by Slater determinants or configuration state functions.According to the laws of quantum mechanics, for systems with only one electron, an energy is associated with each electron configuration and, upon certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon.Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. The concept is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.