CLASSROOM CONNECTORS
... A property is a set of identifying characteristics about a substance. Physical properties are easy to identify because they involve your senses of sight, touch, taste, smell and hear. Common examples of physical properties include the color, size and texture of an object. The characteristics, howeve ...
... A property is a set of identifying characteristics about a substance. Physical properties are easy to identify because they involve your senses of sight, touch, taste, smell and hear. Common examples of physical properties include the color, size and texture of an object. The characteristics, howeve ...
C6-Chemical Reactions
... change is called a chemical property. Chemical properties can be used to identify a substance. But chemical properties can be observed only when a substance undergoes a chemical change. Chemical Change- the composition or identity of the matter changes. A new substance or new substances are ...
... change is called a chemical property. Chemical properties can be used to identify a substance. But chemical properties can be observed only when a substance undergoes a chemical change. Chemical Change- the composition or identity of the matter changes. A new substance or new substances are ...
How to Balance Chemical Equations
... 2. How to balance a chemical equation In balancing chemical equations you need to remember to three important points. a) You do not change the chemical _____________ of a substance to balance an equation. b) An equation is balanced by writing __________numbers before a chemical symbol or formula. c) ...
... 2. How to balance a chemical equation In balancing chemical equations you need to remember to three important points. a) You do not change the chemical _____________ of a substance to balance an equation. b) An equation is balanced by writing __________numbers before a chemical symbol or formula. c) ...
File
... 8.P.1 Understand the properties of matter and changes that occur when matter interacts in an open and closed container. 8.P.1.1 Classify matter as elements, compounds, or mixtures based on how the atoms are packed together in arrangements. 8.P.1.2 Explain how the physical properties of elements and ...
... 8.P.1 Understand the properties of matter and changes that occur when matter interacts in an open and closed container. 8.P.1.1 Classify matter as elements, compounds, or mixtures based on how the atoms are packed together in arrangements. 8.P.1.2 Explain how the physical properties of elements and ...
Candle Mass Lab and the Law of Conservation of Matter Notes.
... • Circle the letter of each correct sentence. • A. Carbon and oxygen react to form carbon monoxide. • B. Carbon and oxygen react to form carbon dioxide. • C. Carbon dioxide yields carbon and oxygen. • D. The reaction of carbon and oxygen yields carbon dioxide. ...
... • Circle the letter of each correct sentence. • A. Carbon and oxygen react to form carbon monoxide. • B. Carbon and oxygen react to form carbon dioxide. • C. Carbon dioxide yields carbon and oxygen. • D. The reaction of carbon and oxygen yields carbon dioxide. ...
Chemical properties Chemical properties can be recognized only
... Chemical properties Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. The following chemical properties can be used to help identify a substance: Ability to burn The ability to burn involv ...
... Chemical properties Chemical properties can be recognized only when substances react or do not react chemically with one another, that is, when they undergo a change in composition. The following chemical properties can be used to help identify a substance: Ability to burn The ability to burn involv ...
Physical and Chemical Changes
... For Example.... Add 3ml of the sodium iodate solution (solution A) to a test tube. Add 3ml of the sodium sulfate, citric acid and starch solution (solution B) to the test tube. Swirl the test tube around. ...
... For Example.... Add 3ml of the sodium iodate solution (solution A) to a test tube. Add 3ml of the sodium sulfate, citric acid and starch solution (solution B) to the test tube. Swirl the test tube around. ...
chemical reaction
... 3. All metal halide (halogen) compounds are soluble, except those of Ag+, Hg+ & Pb+2 4. All sulfates are soluble except: Ba+2, Sr+2, Ca+2, Pb+2,Ag+, ...
... 3. All metal halide (halogen) compounds are soluble, except those of Ag+, Hg+ & Pb+2 4. All sulfates are soluble except: Ba+2, Sr+2, Ca+2, Pb+2,Ag+, ...
13.2 Chemical Formulas
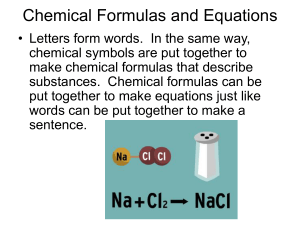
... What is a chemical formula? Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elemen ...
... What is a chemical formula? Chemical formulas have two important parts: chemical symbols for the elements in the compound and subscripts that tell how many atoms of each element are needed to form the compound. The chemical formula for water, H2O, tells us that a water molecule is made of the elemen ...
Docking
... Small Molecule/Ligand (Similarity) Predictive Methods (Kernel Methods) Why it is not hopeless ...
... Small Molecule/Ligand (Similarity) Predictive Methods (Kernel Methods) Why it is not hopeless ...
Chemical Formulas and Equations
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
... The Importance of Accuracy • CO2 is a colorless, odorless gas you exhale. • CO is a colorless, odorless, and poisonous gas. • Co is an element. ...
Introduction to Chemical Reactions
... The reactants are used up in forming the product The arrow shows the direction of the reaction ...
... The reactants are used up in forming the product The arrow shows the direction of the reaction ...
II. Classification of Matter
... Law vs. theory Scientific (natural) _____________: a general statement based on the observed behavior of matter to which no exceptions are known. __________________: a broad generalization that explains a body of facts or phenomena. Quantitative vs. qualitative data Quantitative: numerical (________ ...
... Law vs. theory Scientific (natural) _____________: a general statement based on the observed behavior of matter to which no exceptions are known. __________________: a broad generalization that explains a body of facts or phenomena. Quantitative vs. qualitative data Quantitative: numerical (________ ...
Discover Chemical Changes - gk-12
... with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 possible chemical changes that can be used. Students should move from station to station ...
... with before and after chemical changes so that students can make the chemical changes happen themselves or at least make observations of chemical changes that have happened at each station. I have listed above 9 possible chemical changes that can be used. Students should move from station to station ...
Matter- Types and Changes
... components with variable composition • Solution – material with two or more components homogeneously mixed; can have variable proportions of the components • Pure substance – material with a constant chemical composition ...
... components with variable composition • Solution – material with two or more components homogeneously mixed; can have variable proportions of the components • Pure substance – material with a constant chemical composition ...
Cosmetology Learning Module 12
... reduced (loses oxygen) and the reducing agent is oxidized (gains the oxygen) Oxidizing Agent – substance that releases oxygen. Reducing Agent – substance that adds hydrogen to a chemical compound or subtracts oxygen from the compound. ...
... reduced (loses oxygen) and the reducing agent is oxidized (gains the oxygen) Oxidizing Agent – substance that releases oxygen. Reducing Agent – substance that adds hydrogen to a chemical compound or subtracts oxygen from the compound. ...
e c n i
... Chemical reactions take place when chemical bonds are either formed or broken. Strong chemical bonds resist change: glass W eak chemical bonds breakdown easily: wood ...
... Chemical reactions take place when chemical bonds are either formed or broken. Strong chemical bonds resist change: glass W eak chemical bonds breakdown easily: wood ...
Physical Science
... Physical Change -A change that alters the form of a substance but not the chemical makeup of the substance, a change of state ...
... Physical Change -A change that alters the form of a substance but not the chemical makeup of the substance, a change of state ...
Reactions and Equations
... • The law of conservation of mass states that mass is neither created nor destroyed during a chemical reaction—it is conserved. • In other words, the mass of the reactants equals the mass of the products. ...
... • The law of conservation of mass states that mass is neither created nor destroyed during a chemical reaction—it is conserved. • In other words, the mass of the reactants equals the mass of the products. ...
1. What is a Chemical Reaction?
... reaction has occurred. Lesson Summary • A chemical reaction is the process by which atoms of one or more substances are rearranged to form different substances(s) with new chemical and physical properties. • A chemical reaction is another name for a chemical change. • When substances chemically reac ...
... reaction has occurred. Lesson Summary • A chemical reaction is the process by which atoms of one or more substances are rearranged to form different substances(s) with new chemical and physical properties. • A chemical reaction is another name for a chemical change. • When substances chemically reac ...
CHEMICAL EQUATIONS, SYMBOLS, FORULAS 7
... The law of conservation of matter states that matter can neither be created nor destroyed, but can be changed in form. The total mass of the material(s) before the reaction is the same as the total mass of material(s) after the reaction. A balanced chemical equation has the same number of each kind ...
... The law of conservation of matter states that matter can neither be created nor destroyed, but can be changed in form. The total mass of the material(s) before the reaction is the same as the total mass of material(s) after the reaction. A balanced chemical equation has the same number of each kind ...
Destruction of Syria's chemical weapons

The destruction of Syria's chemical weapons began with several international agreements that were arrived at with Syria, with an initial destruction deadline of 30 June 2014. United Nations Security Council Resolution 2118 imposed on Syria responsibilities and a timeline for the destruction of its chemical weapons and chemical weapons production facilities. The Security Council resolution incorporated and bound Syria to an implementation plan enacted in an Organisation for the Prohibition of Chemical Weapons (OPCW) Executive Council Decision. On 23 June 2014, the last declared chemical weapons were shipped out of Syria for destruction. The destruction of the most dangerous chemical weapons began at sea aboard the Maritime Administration Ready Reserve Force vessel CAPE RAY crewed with U.S. civilian merchant mariners. It took 42 days aboard ship to destroy 600 metric tons of chemical agents that would have been used to make deadly Sarin and Mustard Gas.The impetus toward destroying Syria's chemical weapons began with a 9 September 2013 rhetorical suggestion by U.S. Secretary of State John Kerry that Syria turn over all of its chemical weapons within a week. At the time, the U.S. and France headed a coalition of countries on the verge of carrying out air strikes on Syria in response to the 21 August 2013 Ghouta attacks. The suggestion received a positive response from Russia and Syria, and U.S.–Russian negotiations led to the 14 September 2013 ""Framework for Elimination of Syrian Chemical Weapons,"" which calls for the elimination of Syria's chemical weapon stockpiles by mid-2014. Following the agreement, Syria acceded to the Chemical Weapons Convention and agreed to apply that convention provisionally until its entry into force on 14 October 2013. On 21 September, Syria ostensibly provided a list of its chemical weapons to the OPCW, before the deadline set by the framework.On 27 September, the Executive Council of the OPCW adopted a decision, ""Destruction of Syrian Chemical Weapons,"" a detailed implementation plan based on the U.S./Russian agreement. Later on 27 September, the UN Security Council unanimously passed United Nations Security Council Resolution 2118, incorporating the OPCW plan and making it binding on Syria. A joint OPCW-UN mission will supervise the destruction or removal of Syria's chemical arms, while its Director-General is charged with notifying the Executive Council regarding any delay in implementation. The Executive Council would decide whether the non-compliance should be reported to the Security Council, which is responsible for making certain Syria fulfills its commitments under Resolution 2118.OPCW began preliminary inspections of Syria's chemical weapons arsenal on 1 October 2013, and actual destruction began on 6 October. Under OPCW supervision Syrian military personnel began ""destroying munitions such as missile warheads and aerial bombs and disabling mobile and static mixing and filling units."" The destruction of Syria's declared chemical weapons production, mixing, and filling equipment was successfully completed by 31 October deadline. The destruction of the chemical weapons fell well behind schedule. The entire chemical weapons stockpile had been scheduled to be completely removed from the country by 6 February 2014. Only on 23 June 2014, had Syria finished shipping the remaining declared chemicals. On 18 August 2014, all of the most toxic chemicals had been destroyed offshore. Western officials such as British Ambassador Mark Lyall Grant have expressed concerns about the completeness of Syria's disclosures, and believe the OPCW mission should remain in place following the removal of chemical weapons until verification tasks can be completed.Chlorine, a common industrial chemical, is outside the scope of the disarmament agreement; however, its use as a poison gas would violate the Chemical Weapons Convention, which Syria joined in 2013. Various parties, including Western governments, have accused Assad of conducting illegal chlorine attacks in 2014 and 2015. A late disclosure in 2014 regarding Syria's ricin program raised doubts about completeness of the government's declaration of its chemical weapons stockpile, and in early May 2015, OPCW announced that inspectors had found traces of sarin and VX nerve agent at a military research site in Syria that had not been declared previously by the Assad regime.