* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Acid-Base Balance
Survey
Document related concepts
Genetic code wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Metalloprotein wikipedia , lookup
Proteolysis wikipedia , lookup
Citric acid cycle wikipedia , lookup
Biosynthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Butyric acid wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Transcript
James Howard Acid-Base Balance [H+] maintained at 35-45 nmol/L pH 7.35 – 7.45 > 120nmol or <20nmol incompatible with life K+ Affecting Enzyme activity Hydrogen ion transporters (N.B K+) Osmolality H+ Cell Acid Production Fixed (non-volatile) acids Mainly from oxidation of amino acids 60 mmol/day 4 mmol/L Respiratory (volatile) acids Carbonic acid (H2CO3) In a state of equilibrium with CO2 Balance Usually acid excretion = acid production, through Buffering – Practically instantaneous 2. Respiratory control – Minutes 3. Renal response – Days/weeks 4. (The liver) 1. Cliché Buffering Dogs infused with 14mmol/L H+ Rise of 36 nmol/L observed Huge buffering capacity Base excess – acid required to blood pH to 7.4 Bicarbonate mainly responsible in ECF HCO3- + H+ CO2 + H2O Catalysed by Carbonic anhydrase Amongst fastest enzymes in nature Also plasma proteins, phosphates, Hgb But... 1. Buffering relies on a steady supply of base Buffering system cannot handle changes in several variables 2. pKa of the bicarbonate system is 6.1 Fortunately, the body is not a closed system! In a Nutshell (CO2 + H2O H2CO3 H+ + HCO3- ) Buffering CO2 + H2O H+ + HCO3- Controlled by lungs Controlled by kidneys Respiratory Control ΔpCO2 ΔpH Rapid– good circulation + CO2 lipid soluble Typically pCO2 drives respiratory control via pH 1A physiology with CO2 absorber CSF has little buffering capacity BBB impermeable to protein, H+, HCO3- CO2 diffuses across BBB – proportional ΔpH Chemoreceptors input to medullar respiratory centre N.B Roles of peripheral chemoreceptors Gratuitous Schematic Ventrolateral medulla CSF Blood HCO3CO2 H+ H+ + HCO3- CO2 Albumin CO2 HCO3- H+ Albumin But... We can buffer changes in pH We can blow CO2 off to reduce H+ At the expense of HCO3 But what if ↑pCO2 – respiratory acidosis ↑ H+ - metabolic acidosis AND how do we (re)generate our HCO3-? Renal Regulation So many different hypotheses, I’ll go with: We form ammonium (NH4+) and bicarbonate We reabsorb them both We secrete what we don’t want Renal Regulation Glutamine NH4+ + HCO32. Reabsorption of HCO33. Reabsorption of NH4+ 4. Secretion of NH4+ 1. The Liver Produces ~20% of daily CO2 ( HCO3- + H+) Protons can be consumed & bicarbonate formed Metabolism of organic anions (citrate, lactate, ketones etc.) Key in lactic acidosis etc. Bases can be eliminated in the urea cycle 2NH4+ + 2HCO3- H2N-CO-NH2 + 3H2O + CO2 Inhibited by pH Produces plasma proteins, important for buffering In a Nutshell (CO2 + H2O H2CO3 H+ + HCO3- ) Buffering CO2 + H2O H+ + HCO3- Controlled by lungs Controlled by kidneys The Liver Miss AM 20 y/o female Admitted with a crushed chest ABG H+ PCO2 HCO3- PO2 Result 63 nmol/L 10.1 kPa 29 mmol/L 6.4 kPa (Reference) (35-45) (4.6 - 6.0) (21 – 28) (10.5 – 13.5) High [H+] & pCO2 Bicarbonate not increased Mr. X 28 y/o male 1/7 Hx of severe vomiting (non-bilous) Self-medicating chronic dyspepsia Severely dehydrated & shallow respiration ABG H+ PCO2 HCO3- PO2 Result 28 nmol/L 7.2 kPa 43 mmol/L 13 kPa (Reference) (35-45) (4.6 - 6.0) (21 – 28) (10.5 – 13.5) Serum Na+ K+ Cl- HCO3- Urea Creat. Result 146 mmol/L 2.8 mmol/L 83 mmol/L 41 mmol/L 31 mmol/L 126 μmol/L (Ref.) (135 - 145) (3.5 – 5.0) (95 - 105) (21 – 28) (2.5 – 8.0) (40 - 130) Urine showed: Na+, K+, pH 5 Diagnosis? H+ Low [H+], high bicarb Raised pCO2 Uraemia, but normal creatinine Hypokalaemia, 3 causes Hypernatraemia Classical paradoxical acid urine K+ Cell Summary 4 key players in acid-base balance, problems in any Ventilatory failure Renal failure Metabolic – lactic acidosis, diabetic ketoacidosis 1. Look at the H+ to see if acidotic/alkalotic 2. Look at bicarb/pCO2 to see if metabolic or acidotic 3. Look at other electrolytes Hyperalosteronism, H+/K+, uraemia etc. The history is key!






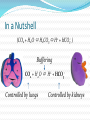





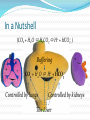









![+ [H 2 CO 3 ]](http://s1.studyres.com/store/data/001130480_1-81612c04e4e5fb35027131a7b7feb9b6-150x150.png)





