* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Forrest-ISAP-post-IC..
Survey
Document related concepts
Globalization and disease wikipedia , lookup
Behçet's disease wikipedia , lookup
Common cold wikipedia , lookup
Neonatal infection wikipedia , lookup
Childhood immunizations in the United States wikipedia , lookup
Infection control wikipedia , lookup
Signs and symptoms of Graves' disease wikipedia , lookup
Sociality and disease transmission wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Multiple sclerosis signs and symptoms wikipedia , lookup
Transcript
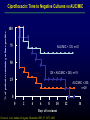
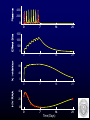
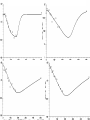
Patient Studies Modeling Surrogates and Their Linkages: MIC, Clinical Scoring and PK/PD Indices of Effect Alan Forrest, Pharm.D. State University of New York at Buffalo School of Pharmacy & Pharmaceutical Sciences, School of Medicine & Biomedical Science Institute for Clinical Pharmacodynamics Ordway Research Institute; Albany, NY CPL Associates, LLC; Buffalo, NY Overview • Difficulties in studying PK/PD of anti-infectives in humans • PD endpoints, in ID, which might be modelled – Advantages/disadvantages – Current status in drug development – Examples • Proposal to co-model pathogen replication and death, the timecourse of severity of disease manifestation & the effects of drug therapy, on these processes (PD) • Enabling study designs • Summary/Conclusions – Implications, applications – What is needed? Informative PK/PD ID Studies, in Humans, are Difficult • PD models (drug exposure & pathogen susceptibility versus outcome) usually need large numbers of evaluable patients (“100s”) – Adequate PK data usually needed in all (dose-response analyses are too insensitive, would require even larger numbers) – Positive cultures with susceptibilities needed (must consider drug concentrations in relation to the MIC, ED50, etc; concentration-response analyses are also usually too insensitive) – Relevant covariates & outcomes determined & documented • A large range of drug exposures is needed (dose ranging plus PK variability is better than relying on PK variability, alone) • “Placebo effect” (spontaneous resolution) for certain infections such as acute bacterial exacerbation of chronic bronchitis (ABECB) • To model exposure & susceptibility vs probabilities of success & failure, “adequate” numbers of both outcomes are required (& obviously, we usually cannot design for more clinical failures) Candidate PD Endpoints • Probabilities of good & bad events (categorical/ordinal responses) – Examples: mortality, microbiologic or clinical success or failure, yes/no adverse event (AE), emergence of resistance – Most available type of endpoint, in drug development trials, but least sensitive/informative (thus requires the largest sample size, especially with mortality as an endpoint!; “All-cause mortality is the gold standard”) – Accepted definitions generally available, usually largely based on observations made AFTER the course of treatment (eg, TOC visit) – Probability of clinical cure is a “noisier” endpoint than microbiologic cure, but usually has required similar activity breakpoints (eg, AUC/MIC) – Clinical resolution, after two weeks, is “counted” as being equivalent to needing only two days to achieve substantial resolution Candidate PD Endpoints (continued) • Time to good or bad events (interval, AKA ‘survival’ analyses) – Examples: eradication, clinical resolution, resistance, AE – Under-utilized, more difficult to obtain; (much) more informative; probably less sensitive to placebo effect – Time to eradication & resolution is important to patients, their family & clinicians; likely associated with probability of emergent resistance & other AE; may be associated with needed duration of treatment; probably a factor in total costs of the infectious episode – Not yet widely accepted; needs standard definitions & validation • Change in a continuous numeric or ordinal variable (regression) – Examples: pathogen titer (quantitative or semi-), such as viral load; composite disease scores; a lab value sensitive to an AE (eg, a change in creatinine clearance or platelet count) – Least commonly available, but most sensitive/informative type of data – Smallest required sample sizes Ciprofloxacin vs Lower Respiratory Tract Infections Microbiologic Cure 100 100 % probability of micro cure % probability of clinical cure Clinical Cure 80 60 40 20 0 1 2 3 4 80 60 40 20 0 1 Log10 AUC/MIC Forrest A, et al, Antimicrob Agents Chemother.1993. 37:1073–1081 2 3 Log10 AUC/MIC 4 % of patients remaining culture-positive Ciprofloxacin: Time to Negative Cultures vs AUC/MIC 100 75 AUC/MIC < 125, n=21 50 125 < AUC/MIC < 250, n=15 25 AUC/MIC > 250, n=28 0 0 2 4 6 8 10 Days of treatment Forrest A, et al, Antimicrob Agents Chemother.1993. 37:1073–1081. 12 14 Time to Emergence of Resistance Percent Susceptible 100 AUIC >100 (n=97) 80 60 GNR treated with -lactam (n=14) 40 20 0 0 AUIC <100 (n=17) 5 10 15 20 Days From Initiation of Therapy Thomas, et al. Antimicrob Agents Chemother.1998.42(3):521-527 Population Pharmacokinetics of Linezolid in Seriously Ill Adult Patients from a Compassionate Use Protocol Alison K. Meagher, Alan Forrest, Craig R. Rayner, Mary C. Birmingham, Jerome J. Schentag AAC 47(2):548-53 2003. Population Pharmacodynamics of Linezolid in Seriously Ill Adult Patients from a Compassionate Use Protocol Craig R. Rayner, Alan Forrest, Alison K. Meagher, Mary C. Birmingham, Jerome J. Schentag Clin PK 42(15):1411-23 2003. Pharmacostatistical Modelling of Hematologic Effects of Linezolid in Seriously Ill Patients Alan Forrest, Craig R. Rayner, Alison K. Meagher, Mary C. Birmingham, Jerome J. Schentag 40th ICAAC, Toronto, Ontario, Canada, Sept 2000. Probability of Eradication 1.0 1.0 0.8 0.8 0.6 0.6 0.4 0.4 Bacteremia N=68 0.2 0.2 0.0 Probability of Eradication 10 100 1000 0.0 10 1.0 1.0 0.8 0.8 0.6 0.6 0.4 Lower respiratory tract infection N=11 0.2 0.0 Bone infection N=15 10 100 AUIC (SIT-1 hr) 1000 0.4 100 1000 Skin and skin structure infection N=36 0.2 0.0 10 100 AUIC (SIT-1 hr) 1000 Linezolid AUC/MIC and Time to Negative Cultures, in Patients with Bacteremia 1.0 >105 N=39 51-105 N=23 0.8 <51 N=6 0.6 0.4 AUC/MIC T50%Erad T75%Erad 0.2 <51 4.0 - 51-105 3.0-4.0 8.0-9.0 >105 0.5-1.0 4.0-5.0 P=0.0210 * 0.0 0 10 20 Days of Therapy 30 * Log-rank test % Reduction in Platelets versus AUC and Duration 90 80 70 60 % in Platelets 50 40 30 20 10 120 100 1000 80 Duration of Therapy (Days) 800 60 600 40 400 20 200 0 AUC (mg/Lx24hr) Decision Analysis Decision Analysis % Probability or % Decrease Predicting Population Outcomes: Bacteremia, 600mg IV q.12hr x 3wk 120 % Probability of Failure 80 MIC (mg/L): 1.0 2.0 4.0 % Decrease in: Platelets 40 Hemoglobin 0 10 100 24hr Average AUC (mg/ Lxhr ) 1000 4000 2000 0 0 7 14 21 0 7 14 21 0 7 14 21 0 7 14 21 1500 1000 500 0 80 40 0 40 20 0 Time (Days) System of Models for Pathogen Growth & Death, Disease State Progression & the Effects of Drug Therapy • 1st biomath model: in the infected host, pathogen titers initially increase rapidly (output is culture +/-, semi- or quantitative titers) • 2nd model: pathogen produces toxins, etc, stimulates inflammatory processes, etc, which result in appearance & worsening of signs & symptoms of infection (output is mostly ordinal, probably a composite disease severity score) • 3rd model (PK/PD): effective treatment reduces titer (inhibits replication &/or enhances rate of death), drives titer to BLQ • Disease signs & symptoms (usually) begin to lessen later than titers start to drop & resolve more slowly (time to resolution correlated with but NOT EQUAL to time to eradication) METHODS: Protocol Highlights • Non-comparative, open label, single-center, pilot study, of gatifloxacin (for 5 days) to treat acute maxilary sinusitis • Symptoms for greater than 7 days (purulent nasal discharge from maxillary sinus orifice, nose, or back of the throat) • Radiological documentation with opacification, an air fluid level, or mucosal thickening of 5 mm • Indwelling sinus catheter allowed sampling of sinus aspirate (SA), for drug concentration & semi-quantitative culture results • C&S of SA daily x5; on day 4 serial samples of plasma & SA were obtained (6 samples over 6 hours) for gatifloxacin assay Ambrose PG, et al. Use of Pharmacodynamic Endpoints in the Evaluation of Gatifloxacin for the Treatment of Acute Maxillary Sinusitis. Clin Infect Dis. 2004. 38:1513-20 Gatifloxacin Exposure: Plasma & Sinus Aspirate Time to Bacterial Eradication Time to Resolution Correlated with Time to Eradication • 8 other signs & symptoms were also followed • 91% (29/32) of the total number of signs & symptoms resolved by the end of therapy • Time to sinus sterilization correlated with median time to resolution of signs and symptoms (rs = 0.85) Change of pneumonia score 10 5 Addition of Synercid VAN AUC/MIC ~180 0 -8 -6 -4 -2 0 2 4 6 8 10 12 14 16 18 20 22 24 26 -5 Culture (-) VAN AUC/MIC ~450 -10 Culture (-) -15 Treatment day VAN AUC/MIC ~80 + SYN AUC/MIC ?? 28 Proposed Enabling Study Designs • Data which should ‘usually’ be obtained – Plasma PK and baseline culture and sensitivities (C&S) – Yes/No clinical and microbiologic success/failure – Serial disease severity scores • Data to be obtained when possible – Serial C&S from the ‘site of infection’ (eg, ELF, CSF, blood) • Quantitative or semi-quantitative titers, if possible – PK data from the ‘site of infection’ Summary and Conclusions • We believe that time to eradication and time to (substantive) clinical resolution, for example, are strongly associated and, if they could be acceptably defined and validated, would be superior and relevant endpoints in ID drug development research – more sensitive, specific and informative • Composite ID disease severity scores – Components would differ by site/type of infection – Perspective: patients’ (how do you feel?); clinicians (signs & symptoms plus results of tests & procedures used to monitor patients); other? • What is needed? – Develop new definitions/standards, evaluate existing ones – Develop, with regulatory scientists, an approach to evaluate & validate these “new” endpoints (eg, against what/which Gold Standard(s)?) • Retrospective application to existing data? Prospective only?