* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Document
Polysubstance dependence wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
NK1 receptor antagonist wikipedia , lookup
Clinical trial wikipedia , lookup
Pharmacogenomics wikipedia , lookup
CCR5 receptor antagonist wikipedia , lookup
Drug design wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Neuropharmacology wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Drug interaction wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacognosy wikipedia , lookup
HIV vaccine wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Sol–gel process wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug discovery wikipedia , lookup
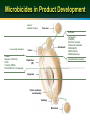
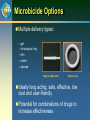
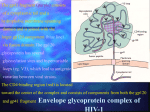
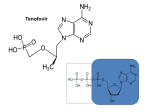
Microbicide Products in the Pipeline Regional Meeting on Regulatory Issues in Microbicide Research Dr. Zeda F. Rosenberg International Partnership for Microbicides 29 October 2007 New Delhi, India Microbicides in Product Development Lactin-V Invisible Condom Free virus BufferGel Attachment Locus small molecules S-DABO Dapivirine (TMC120) UC781 Tenofovir (PMPA) PC815 (MIV150 + Carraguard) Fusion Carraguard PRO2000 SPL7013 (VivaGel) Monoclonal antibodies DS003 (BMS) DS001 (Merck) DS006 (Maraviroc) Pyrimidinediones (Samjin) Replication (RT) Integration Protein synthesis and assembly Budding Maturation Microbicide Options Multiple delivery types: – gel – intravaginal ring – film – tablet – sponge Vaginal applicator Vaginal ring Ideally long acting, safe, effective, low cost and user-friendly Potential for combinations of drugs to increase effectiveness Rationale for Vaginal Dosing Local drug levels can be high and systemic exposure low Maximum drug:virus level Lower chance for systemic related side effects Precedent in contraception No one prevention option will satisfy all; multiple approaches available to address acceptability preferences Women-initiated Early-Generation Microbicides Non-specifically block HIV from interacting with target cells Polyanions recently or currently in HIV efficacy trials (Carraguard, CS, PRO 2000, Buffergel) Some polyanions are also acid-buffering agents Partial, low or no effectiveness Early Generation Microbicides Advantages inexpensive broad activity lack of systemic absorption some are contraceptive Disadvantages reduced or no activity against R5 viruses coitally dependent may not be at the right place at the right time activity reduced in seminal plasma Next-Generation Anti-retroviral (ARV) Microbicides Advantages Highly potent and HIV-specific Documented safety and efficacy as therapeutics Can be formulated for sustained protection • Once a day or less frequent • Gels, intravaginal rings, others Disadvantages Potential for resistance Lack of activity against other STDs Topical Tenofovir Nucleotide reverse transcriptase inhibitor Viread marketed as a therapeutic Preclinical development began in late 1990s HPTN 050 Phase I Safety Study completed in US (NIH) Well tolerated Low serum levels in 56% of subjects HPTN 059 Phase II Safety Study ongoing in US and India (NIH) CONRAD PK study of 1% Tenofovir gel to determine systemic and local tissue levels – ongoing (IPM) Topical Tenofovir (cont.) CAPRISA 004 Phase IIb study began in May 2007 (USAID) – Dosing regimen: coitally +/- 12 hours MTN 001 Comparison of once daily tenofovir oral and gel pK, adherence and acceptability in 120 women – Planned in Uganda, SA, and US MTN 003 Phase IIb Vaginal Tenofovir and Oral Tenofovir and Truvada in 4200 women – Dosing regimen: once per day – Planned in SA, Malawi, Uganda, Zambia and Zimbabwe TMC120 (Dapivirine): Background NNRTI developed by Tibotec/J&J, licensed to IPM (2004) Developed originally as therapeutic, 11 clinical studies conducted via oral administration Highly potent ARV Low cytotoxicity, non-mutagenic, non-teratogenic Easily manufactured, cheap Stable drug substance HC CH CN IP clarity N Multiple dosage forms N N N 3 3 CH3 H H Dapivirine gel/ring: Clinical studies Study Study Name Location Volunteers Status IPM003 Gel 002 Safety SA, Tanzania, Rwanda 112 completed IPM004 Gel 002 PK South Africa 18 completed IPM005B Gel 002 Expanded Safety Belgium 36 completed IPM001 PC Ring Feasibility Belgium 12 completed IPM008 WC Ring Feasibility Belgium 13 completed IPM011 WC Placebo Ring Safety /Acceptability South Africa, Kenya, Tanzania 200 ongoing IPM012 Gel 4750 and 4789 pK and Safety Belgium 36 November 2007 start IPM018 Dapivirine Matrix and Reservoir Ring pK Belgium 24 completed UC-781 Background Carboxanilide type of NNRTI H N O CH3 S O Cl Potent anti-HIV-1 activity (nM range) Tight binding to HIV-1 RT Active against cell-free and cell-associated virus Little to no cytotoxicity (>M) Active against RT inhibitor resistant strains Reduced likelihood for resistance selection Exhibits so-called “Memory Effect” Phase I safety study in 48 women completed UC-781 Ongoing Studies SAFETY STUDIES CDC/Emory ENROLLMENT GEL TYPE DURATION ENDPOINTS Twice-daily for 14 days Systemic/local toxicity, absorption, flora, acceptability 36 sexually active women 18 abstinent HIV positive women 0.1, 0.25, placebo University of Pittsburgh/NIH 60 abstinent women 0.1% or placebo at 0, 2, 4, and 8 hrs duration Single dose Safety, persistence of UC781, systemic absorption , anti-HIV-1 activity, vaginal flora, acceptability CDC/Thailand 45 sexually active women 0.1, 0.25, placebo Twice-daily for 14 days Systemic/local toxicity, absorption, flora, acceptability, anti-HIV activity CONRAD/ CFHC 36 abstinent men 0.25 %, placebo Once-daily for 7 days Systemic/local toxicity, absorption UCLA/NIH 36 women and men 0.1, 0.25, placebo 2 phases: Single dose; Once-daily for 1 week Systemic/local toxicity, rectal mucosal damage, PK subset 0.25 (12♀) UP (6♀) MIV-150 Background PC-815, Carraguard plus MIV-150 (NNRTI) Phase 1 crossover study (Dominican Republic, Profamilia) PC-815 and Carraguard – 20 women, 1 week single dose/day followed by 1 week twice dosing/day – Safety and pharmacokinetic for systemic absorption of MIV-150 – Planned start Nov 2007 Phase I male tolerance, (South Africa, Setshaba Research Center) – Safety and pharmacokinetic for systemic absorption of MIV-150 – 10 circumcised and 10 uncircumised males – IRB approved; – Planned start following Phase I in women HIV Entry HIV HIV gp120 Coreceptor Binding gp120 CD4 Binding HIV gp41 gp120 CD4 CCR5 CXCR4 CD4 gp120 gp41 CCR5 CXCR4 gp120 Gp41 Mediated Fusion Host Cell gp41 HIV-Specific Entry/Fusion Inhibitors Act on virus: gp120 or gp41 blockers DS003 (BMS793), cyanovirin, FI peptides Disadvantages - current gp41 blockers are peptides Act on target cell: CCR5 blockers DS001 (M167), Maraviroc, PSC-Rantes Disadvantages - lack of activity against X4 virus Next-Generation Product Development Tenofovir Gel Dapivirine Gel & Ring UC781 Gel MIV150 + Carraguard Gel Monoclonal antibodies DS001 (M167) DS001 (M167) + Dapivirine Dapivirine Tablet & Film S-DABO Pyrimidinediones DS003 (BMS793) Maraviroc Early Preclinical 1-2 years Preclinical 1-2 years Phase I/II 2+ years Phase III 3 years Filing Approval 1 year Future Pipeline New mechanisms of action Integrase inhibitors Small molecule fusion inhibitors ?? Need to add back-up drugs to hedge against drop-out Need to add better options In late stage development or marketed therapeutic Combination Microbicides Advantages potential increased efficacy against resistant virus coverage of multiple transmission pathways potential synergy and need for less drug Disadvantages unclear regulatory pathway possible difficulties in co-formulation possible increased cost increased potential for toxicity cross company/institutional agreements Criteria for Moving Forward: Pre-Clinical Compounds assessed to identify best candidates for clinic MECHANISMS OF ACTION LABORATORY STUDIES Earlier in life cycle is better Toxicity / Potency New mechanism of action Pre-formulation Comparison with other Stability candidates with same mechanism of action BUSINESS CASE IP access to compound Cost Drug synthesis process Ease of manufacture Criteria for Moving Forward: Early Clinical Trials Candidates assessed in small numbers of volunteers PHARMACOKINETICS ACCEPTABILITY SAFETY Where drug goes in body, Placebo formulations Drug, formulation, delivery concentration, duration assessed in diverse populations assessed for prolonged use Preferred dosage: Wide distribution in genital tract Long duration Sufficient concentration Product safety evaluated in Acceptability measured in diverse populations all clinical trials Early clinical trials cannot fully predict risk of enhancing HIV transmission Criteria for Moving Forward: Efficacy Trials Top candidates move into efficacy trials BEST-IN-CLASS Essential criteria: Potency Safety PK Acceptable formulation Secondary criteria: Mechanism of action Cost Ease of manufacture Access / IP Will Microbicides Work? Predicated on getting the RIGHT drug at the RIGHT levels in the RIGHT place at the RIGHT time Right Drugs and Right Levels Highly potent ARVs acting early in HIV lifecycle Right Place and Right Time Formulations to keep drug(s) in vaginal lumen and/or in tissue depending on drug’s MOA Long acting sustained release Drug Discovery, Development and Review Process 250 compounds 10,000 compounds 6.5 years Stage 3 Clinical trials Phase I Phase II 5 compounds 7 years Phase III Marketing application submitted Stage 2 Pre-clinical First clinical trial application submitted Stage 1 Drug discovery Stage 4 Regulatory review 1 approved drug 1.5 years Adapted from: Pharmaceutical Research and Manufacturers of America, 2006 Lessons Learned from HIV/AIDS Treatment 1981 1997 1987 1983 First AIDS case reported in the US 1995 AZT mono-therapy approved for use HIV virus identified 2003 2002 Three-drug therapy: HAART Brazil offers free universal access to treatment Two-drug therapy becomes available 2006 Drug combinations/ reducing pill burden “3 by 5” Initiative & PEPFAR launched Global Fund established 26 FDA-approved drugs and research continues