* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Chemistry - Rutgers University, Newark
Survey
Document related concepts
Elias James Corey wikipedia , lookup
Kinetic resolution wikipedia , lookup
George S. Hammond wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Petasis reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Hydroformylation wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Transcript
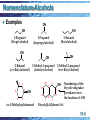
Organic Chemistry William H. Brown Christopher S. Foote Brent L. Iverson 10-1 Alcohols and Thiols Chapter 10 10-2 Structure - Alcohols The functional group of an alcohol is an -OH group bonded to an sp3 hybridized carbon • bond angles about the hydroxyl oxygen atom are approximately 109.5° H Oxygen is sp3 hybridized H O C 108.9° H H • two sp3 hybrid orbitals form sigma bonds to carbon and hydrogen • the remaining two sp3 hybrid orbitals each contain an unshared pair of electrons 10-3 Nomenclature-Alcohols IUPAC names • the parent chain is the longest chain that contains the OH group • number the parent chain to give the OH group the lowest possible number • change the suffix -e to -ol Common names • name the alkyl group bonded to oxygen followed by the word alcohol 10-4 Nomenclature-Alcohols Examples OH OH OH 1-Propanol (Propyl alcohol) 1-Butanol (Butyl alcohol) 2-Propanol (Isopropyl alcohol) OH OH OH 2-Butanol (sec-Butyl alcohol) 3 10 2 1 4 5 2-Methyl-1-propanol (Isobutyl alcohol) OH 6 cis-3-Methylcyclohexanol 9 8 7 1 6 2-Methyl-2-propanol (tert-Butyl alcohol) 2 OH 3 4 5 Numbering of the bicyclic ring takes precedence over the location of -OH Bicyclo[4.4.0]decan-3-ol 10-5 Nomenclature of Alcohols Compounds containing more than one OH group are named diols, triols, etc. CH2 CH2 OH OH 1,2-Ethanediol (Ethylene glycol) CH3 CHCH2 HO OH 1,2-Propanediol (Propylene glycol) CH2 CHCH2 HO HO OH 1,2,3-Propanetriol (Glycerol, Glycerine) 10-6 Nomenclature of Alcohols Unsaturated alcohols • show the double bond by changing the infix from -anto -en• show the the OH group by the suffix -ol • number the chain to give OH the lower number HO 1 2 3 6 4 5 (E)-2-Hexene-1-ol (trans-2-Hexen-1-ol) 10-7 Physical Properties Alcohols O are polar compounds + H +C H H H • they interact with themselves and with other polar compounds by dipole-dipole interactions Dipole-dipole interaction: the attraction between the positive end of one dipole and the negative end of another 10-8 Physical Properties Hydrogen bonding: when the positive end of one dipole is an H bonded to F, O, or N (atoms of high electronegativity) and the other end is F, O, or N • the strength of hydrogen bonding in water is approximately 21 kJ (5 kcal)/mol • hydrogen bonds are considerably weaker than covalent bonds • nonetheless, they can have a significant effect on physical properties 10-9 Hydrogen Bonding 10-10 Physical Properties Ethanol and dimethyl ether are constitutional isomers. Their boiling points are dramatically different • ethanol forms intermolecular hydrogen bonds which increase attractive forces between its molecules resulting in a higher boiling point • there is no comparable attractive force between molecules of dimethyl ether CH 3 CH 2 OH CH 3 OCH 3 Ethanol bp 78°C Dimethyl ether bp -24°C 10-11 Physical Properties In relation to alkanes of comparable size and molecular weight, alcohols • have higher boiling points • are more soluble in water The presence of additional -OH groups in a molecule further increases solubility in water and boiling point 10-12 Physical Properties MW bp (°C) Solubility in Water Structural Formula Name CH3 OH Methanol Ethane 32 30 65 -89 Infinite Insoluble CH3 CH2 CH3 Ethanol Propane 46 44 78 -42 Infinite Insoluble CH3 CH2 CH2 OH CH3 CH2 CH2 CH3 1-Propanol Butane 60 58 97 0 Infinite Insoluble CH3 ( CH 2 ) 2 CH 2 OH 1-Butanol Pentane 74 72 117 36 8 g/100 g Insoluble HOCH2 ( CH2 ) 2 CH2 OH 1,4-Butanediol 90 CH3 ( CH 2 ) 3 CH2 OH 1-Pentanol 88 230 138 69 Infinite 2.3 g/100 g Insoluble CH3 CH3 CH3 CH2 OH CH3 ( CH 2 ) 3 CH3 CH3 ( CH 2 ) 4 CH3 Hexane 86 10-13 Acidity of Alcohols In dilute aqueous solution, alcohols are weakly acidic CH3 O H + :O H H Ka = + CH3 O: + H O H [ CH3 O-] [H3 O+ ] [ CH3 OH] – H = 1 0 - 15 .5 pKa = 1 5 .5 10-14 Acidity of Alcohols Compound Structural Formula pKa Hydrogen chloride HCl -7 Acetic acid CH3 COOH Methanol CH3 OH 15.5 Water Ethanol H2 O 15.7 CH3 CH 2 OH 15.9 2-Propanol ( CH3 ) 2 CHOH 17 2-Methyl-2-propanol ( CH3 ) 3 COH 18 4.8 Stronger acid Weaker acid *Also given for comparison are pKa values for water, acetic acid, and hydrogen chloride. 10-15 Acidity of Alcohols Acidity depends primarily on the degree of stabilization and solvation of the alkoxide ion • the negatively charged oxygens of methanol and ethanol are about as accessible as hydroxide ion for solvation; these alcohol are about as acidic as water • as the bulk of the alkyl group increases, the ability of water to solvate the alkoxide decreases, the acidity of the alcohol decreases, and the basicity of the alkoxide ion increases 10-16 Reaction with Metals Alcohols react with Li, Na, K, and other active metals to liberate hydrogen gas and form metal alkoxides 2CH3 OH + 2Na - + 2CH3 O Na + H2 Sodium methoxide (MeO-Na+) Alcohols are also converted to metal alkoxides by reaction with bases stronger than the alkoxide ion • one such base is sodium hydride CH3 CH2 OH + Na+ HEthanol Sodium hydride CH3 CH2 O- Na+ + H2 Sodium ethoxide 10-17 Reaction with HX • 3° alcohols react very rapidly with HCl, HBr, and HI OH + HCl 2-Methyl-2propanol 25°C Cl + H2 O 2-Chloro-2methylpropane • low-molecular-weight 1° and 2° alcohols are unreactive under these conditions • 1° and 2° alcohols require concentrated HBr and HI to form alkyl bromides and iodides OH + 1-Butanol HBr H2 O reflux Br + H2 O 1-Bromobutane 10-18 Reaction with HX • with HBr and HI, 2° alcohols generally give some rearranged product a product of Br OH + HBr 3-Pentanol rearrangement + H2 O + heat 3-Bromopentane (major product) Br 2-Bromopentane • 1° alcohols with extensive -branching give large amounts of rearranged product Br OH + HBr 2,2-Dimethyl-1propanol + H2 O 2-Bromo-2-methylbutane (a product of rearrangement) 10-19 Reaction with HX Based on • the relative ease of reaction of alcohols with HX (3° > 2° > 1°) and • the occurrence of rearrangements, Chemists propose that reaction of 2° and 3° alcohols with HX • occurs by an SN1 mechanism, and • involves a carbocation intermediate 10-20 Reaction with HX - SN1 Step 1: proton transfer to the OH group gives an oxonium ion CH 3 + : CH3 -C-OH rapid and reversible + H O H CH3 CH3 -C H CH 3 CH3 H O + + H :O H H Step 2: loss of H2O gives a carbocation intermediate CH3 CH3 -C CH3 H O + H slow, rate determining SN 1 H CH3 CH3 - C+ CH3 + :O H A 3° carbocation intermediate 10-21 Reaction with HX - SN1 Step 3: reaction of the carbocation intermediate (an electrophile) with halide ion (a nucleophile) gives the product CH3 CH3 - C+ CH3 + :Cl fast CH3 CH3 - C- Cl CH3 2-Chloro-2-methylpropane (tert-Butyl chloride) 10-22 Reaction with HX - SN2 1° alcohols react with HX by an SN2 mechanism Step 1: rapid and reversible proton transfer : + RCH2 -OH + H O H rapid and reversible + RCH2 - O H + H H :O H H Step 2: displacement of HOH by halide ion Br:- + + RCH2 -O H H slow, rate determining SN2 H RCH2 -Br + :O H 10-23 Reaction with HX For 1° alcohols with extensive -branching • SN1 is not possible because this pathway would require a 1° carbocation • SN2 is not possible because of steric hindrance created by the -branching These alcohols react by a concerted loss of HOH and migration of an alkyl group 10-24 Reaction with HX • Step 1: proton transfer gives an oxonium ion + + H O H O H 2,2-Dimethyl-1propanol H rapid and reversible H O+ + H An oxonium ion O H H • Step 2: concerted elimination of HOH and migration of a methyl group gives a 3° carbocation H O H H slow and rate determining + O (concerted) H A 3° carbocation intermediate 10-25 Reaction with HX Step 3: reaction of the carbocation intermediate (an electrophile) with halide ion (a nucleophile) gives the product Cl - + fast Cl 2-Chloro-2-methylbutane 10-26 Reaction with PBr3 An alternative method for the synthesis of 1° and 2° bromoalkanes is reaction of an alcohol with phosphorus tribromide • this method gives less rearrangement than with HBr OH + PBr 3 2-Methyl-1-propanol Phosphorus (Isobutyl alcohol) tribromide 0° Br 1-Bromo-2-methylpropane (Isobutyl bromide) + H3 PO 3 Phosphorous acid 10-27 Reaction with PBr3 Step 1: formation of a protonated dibromophosphite converts H2O, a poor leaving group, to a good leaving group a good leaving group •• R-CH2 -O-H + Br P Br R-CH2 + O PBr2 + Br H Br Step 2: displacement by bromide ion gives the alkyl bromide + R-CH2 + O PBr2 SN 2 R-CH2 -Br + HO-PBr 2 • • • • Br - H 10-28 Reaction with SOCl2 Thionyl chloride is the most widely used reagent for the conversion of 1° and 2° alcohols to alkyl chlorides • a base, most commonly pyridine or triethylamine, is added to catalyze the reaction and to neutralize the HCl OH + 1-Heptanol SOCl 2 pyridine Thionyl chloride Cl + SO + HCl 2 1-Chloroheptane 10-29 Reaction with SOCl2 Reaction of an alcohol with SOCl2 in the presence of a 3° amine is stereoselective • it occurs with inversion of configuration OH (S)-2-Octanol + SOCl2 Thionyl chloride 3° amine Cl (R)-2-Chlorooctane + SO2 + HCl 10-30 Reaction with SOCl2 Step 1: formation of an alkyl chlorosulfite R1 R1 O C O H + Cl-S-Cl O C H-Cl Cl H R2 H R2 + O S An alkyl chlorosulfite Step 2: nucleophilic displacement of this leaving group by chloride ion gives the chloroalkane R1 Cl + O C O S H R2 Cl SN2 R1 Cl + C R2 O O S + Cl H 10-31 Alkyl Sulfonates Sulfonyl chlorides are derived from sulfonic acids • sulfonic acids, like sulfuric acid, are strong acids O R- S- Cl O A sulfonyl chloride O O R- S- OH R- S- OO O A sulfonate anion A sulfonic acid (a very strong acid) (a very weak base and stable anion; a very good leaving group 10-32 Alkyl Sulfonates A commonly used sulfonyl chloride is ptoluenesulfonyl chloride (Ts-Cl) O CH3 CH2 OH + Cl-S CH 3 O p-Toluenesulfonyl Ethanol chloride pyridine O CH 3 CH 2 O-S CH3 + HCl O Ethyl p-toluenesulfonate (Ethyl tosylate) 10-33 Alkyl Sulfonates Another commonly used sulfonyl chloride is methanesulfonyl chloride (Ms-Cl) OH + O Cl-S- CH3 pyridine O Cyclohexanol Methanesulfonyl chloride O O-S-CH3 + HCl O Cyclohexyl methanesulfonate (Cyclohexyl mesylate) 10-34 Alkyl Sulfonates Sulfonate anions are very weak bases (the conjugate base of a strong acid) and are very good leaving groups for SN2 reactions Conversion of an alcohol to a sulfonate ester converts HOH, a very poor leaving group, into a sulfonic ester, a very good leaving group 10-35 Alkyl Sulfonates This two-step procedure converts (S)-2-octanol to (R)-2-octyl acetate Step 1: formation of a p-toluenesulfonate (Ts) ester OTs OH + TsCl (S)-2-Octanol pyridine + HCl (S)-2-Octyl tosylate Tosyl chloride Step 2: nucleophilic displacement of tosylate O O OTs - + O Na Sodium acetate + (S)-2-Octyl tosylate SN 2 ethanol O + + Na OTs (R)-2-Octyl acetate 10-36 Dehydration of ROH An alcohol can be converted to an alkene by acid-catalyzed dehydration (a type of elimination) • 1° alcohols must be heated at high temperature in the presence of an acid catalyst, such as H2SO4 or H3PO4 • 2° alcohols undergo dehydration at somewhat lower temperatures • 3° alcohols often require temperatures at or slightly above room temperature 10-37 Dehydration of ROH CH 3 CH 2 OH OH H2 SO 4 180°C CH 2 = CH 2 H2 SO 4 + H2 O + H2 O 140°C Cyclohexanol CH 3 CH 3 COH Cyclohexene H2 SO 4 CH 3 2-Methyl-2-propanol (tert- Butyl alcohol) 50°C CH 3 CH 3 C= CH 2 + H2 O 2-Methylpropene (Isobutylene) 10-38 Dehydration of ROH • where isomeric alkenes are possible, the alkene having the greater number of substituents on the double bond (the more stable alkene) usually predominates (Zaitsev rule) OH CH3 CH2 CHCH3 2-Butanol 8 5 % H3 PO 4 heat CH3 CH= CH CH 3 + CH3 CH2 CH= CH2 + H2 O 2-Butene 1-Butene (80%) (20%) 10-39 Dehydration of ROH Dehydration of 1° and 2° alcohols is often accompanied by rearrangement H2 SO4 OH 140 - 170°C 3,3-Dimethyl2,3-Dimethyl2-butanol 2-butene (80%) + 2,3-Dimethyl1-butene (20%) • acid-catalyzed dehydration of 1-butanol gives a mixture of three alkenes OH 1-Butanol H2 SO 4 + 140 - 170°C trans-2-butene (56%) + cis- 2-butene (32%) 1-Butene (12%) 10-40 Dehydration of ROH Based on evidence of • ease of dehydration (3° > 2° > 1°) • prevalence of rearrangements Chemists propose a three-step mechanism for the dehydration of 2° and 3° alcohols • because this mechanism involves formation of a carbocation intermediate in the rate-determining step, it is classified as E1 10-41 Dehydration of ROH Step 1: proton transfer to the -OH group gives an oxonium ion H O + + H O H rapid and reversible H H + H O + O H H Step 2: loss of H2O gives a carbocation intermediate H +H O slow, rate determining + H2 O A 2° carbocation intermediate 10-42 Dehydration of ROH Step 3: proton transfer from a carbon adjacent to the positively charged carbon to water; the sigma electrons of the C-H bond become the pi electrons of the carbon-carbon double bond H O H rapid and reversible + H H + + + H O H H 10-43 •Dehydration of ROH alcohols with little -branching give terminal alkenes and rearranged alkenes 1° • Step 1: proton transfer to OH gives an oxonium ion O-H + 1-Butanol + H O H H rapid and reversible + O-H + H O-H H • Step 2: loss of H from the -carbon and H2O from the -carbon gives the terminal alkene H O H + + O-H H H H + E2 + H O H+ O H 1-Butene H H 10-44 Dehydration of ROH Step 3: shift of a hydride ion from -carbon and loss of H2O from the -carbon gives a carbocation + O-H H H 1,2-shift of a hydride ion + + H O-H H A 2° carbocation Step 4: proton transfer to solvent gives the alkene H H O + H + E1 + + +H O H H trans-2-Butene cis-2-Butene 10-45 Dehydration of ROH Dehydration with rearrangement occurs by a carbocation rearrangement H+ OH 3,3-Dimethyl2-butanol -H2 O + H2 O A 2° carbocation intermediate + + H3 O+ 2,3-Dimethyl2-butene A 3° carbocation intermediate H2 O + H3 O+ 2,3-Dimethyl1-butene 10-46 Dehydration of ROH Acid-catalyzed alcohol dehydration and alkene hydration are competing processes C C An alkene Principle + H2 O acid catalyst C C H OH An alcohol of microscopic reversibility: the sequence of transition states and reactive intermediates in the mechanism of a reversible reaction must be the same, but in reverse order, for the reverse reaction as for the forward reaction 10-47 Pinacol Rearrangement The products of acid-catalyzed dehydration of a glycol are different from those of alcohols HO OH 2,3-Dimethyl-2,3-butanediol (Pinacol) O H2 SO4 + H2 O 3,3-Dimethyl-2-butanone (Pinacolone) 10-48 Pinacol Rearrangement Step 1: proton transfer to OH gives an oxonium ion HO OH + rapid and reversible + H O H H HO H O H + An oxonium ion O H H Step 2: loss of water gives a carbocation intermediate HO H O H HO + H2 O A 3o carbocation intermediate 10-49 Pinacol Rearrangement Step 3: a 1,2- shift of methyl gives a more stable carbocation H O H O H O A resonance-stabilized cation intermediate Step 4: proton transfer to solvent completes the reaction H2 O + H O H3 O + + O 10-50 Oxidation: 1° ROH Oxidation of a primary alcohol gives an aldehyde or a carboxylic acid, depending on the experimental conditions OH CH3 -C H H A primary alcohol [O] O CH3 -C- H An aldehyde [O] O CH3 -C- OH A carboxylic acid • to an aldehyde is a two-electron oxidation • to a carboxylic acid is a four-electron oxidation 10-51 Oxidation of ROH A common oxidizing agent for this purpose is chromic acid, prepared by dissolving chromium(VI) oxide or potassium dichromate in aqueous sulfuric acid CrO3 + H2 O H2 SO4 Chromium(VI) oxide K2 Cr 2 O7 Potassium dichromate H2 SO4 H2 CrO 4 Chromic acid H2 Cr 2 O7 H2 O 2 H2 CrO 4 Chromic acid 10-52 Oxidation: 1° ROH Oxidation of 1-octanol gives octanoic acid • the aldehyde intermediate is not isolated O OH 1-Hexanol H2 CrO4 H2O, acetone H Hexanal (not isolated) O OH Hexanoic acid 10-53 Oxidation: 2° ROH 2° alcohols are oxidized to ketones by chromic acid OH + H2 CrO4 2-Isopropyl-5-methylcyclohexanol (Menthol) O acetone + Cr 3+ 2-Isopropyl-5-methylcyclohexanone (Menthone) 10-54 Chromic Acid Oxidation of ROH • Step 1: formation of a chromate ester O OH + HO-Cr-OH H Cyclohexanol fast and reversible O O-Cr-OH O + H2 O H An alkyl chromate O • Step 2: reaction of the chromate ester with a base, here shown as H2O chromium(VI) chromium(IV) O O H H slow, rate Cr-OH determining O H O + O+ H + H Cyclohexanone OCr-OH O O H 10-55 Chromic Acid Oxidation of RCHO • chromic acid oxidizes a 1° alcohol first to an aldehyde and then to a carboxylic acid • in the second step, it is not the aldehyde per se that is oxidized but rather the aldehyde hydrate O R-C-H + H2 O An aldehyde fast and reversible OH R-C-OH H2 CrO4 H An aldehyde hydrate O-CrO3 H R-C-OH H2 O H O R-C-OH + HCrO3 - + H3 O+ A carboxylic acid 10-56 Oxidation: 1° ROH to RCHO Pyridinium chlorochromate (PCC): a form of Cr(VI) prepared by dissolving CrO3 in aqueous HCl and adding pyridine to precipitate PCC as a solid pyridinium ion chlorochromate ion CrO3 + HCl ClCrO3 - + N Pyridine N H Pyridinium chlorochromate (PCC) • PCC is selective for the oxidation of 1° alcohols to aldehydes; it does not oxidize aldehydes further to carboxylic acids 10-57 Oxidation: 1° ROH • PCC oxidizes a 1° alcohol to an aldehyde O PCC OH H Geraniol Geranial • PCC also oxidizes a 2° alcohol to a ketone OH Menthol PCC O Menthone 10-58 Oxidation of Alcohols by NAD+ • biological systems do not use chromic acid or the oxides of other transition metals to oxidize 1° alcohols to aldehydes or 2° alcohols to ketones • what they use instead is a NAD+ A pyridine ring The business + end of NAD O OH N Nicotinic acid (Niacin; Vitamin B6) O An amide group NH2 N Ad Nicotinamide adenine dinucleotide (NAD+ ) The plus sign in NAD+ represents this charge on nitrogen • the Ad part of NAD+ is composed of a unit of the sugar D-ribose (Chapter 25) and one of adenosine diphosphate (ADP, Chapter 28) 10-59 Oxidation of Alcohols by NAD+ • when NAD+ functions as an oxidizing agent, it is reduced to NADH • in the process, NAD+ gains one H and two electrons; NAD+ is a two-electron oxidizing agent O CNH2 reduction + H+ + 2 e- N+ Ad NAD+ (oxidized form) oxidation H H O CNH2 N Ad NADH (reduced form) 10-60 Oxidation of Alcohols by NAD+ • NAD+ is the oxidizing in a wide variety of enzymecatalyzed reactions, two of which are CH3 CH2 OH + NAD+ Ethanol OH + CH3 CHCOO + NAD Lactate alcohol dehydrogenase O CH3 CH + NADH + H+ Ethanal (Acetaldehyde) lactate dehydrogenase O CH3 CCOO + NADH + H+ Pyruvate 10-61 Oxidation of Alcohols by NAD+ • mechanism of NAD+ oxidation of an alcohol - B • • 1 E B H H • • • • • • • • C C O 2 3 HH O O C-NH2 4-5 N+ Ad NAD+ E reduction of NAD+ oxidation of NADH H H O C-NH2 •• N Ad NADH • hydride ion transfer to NAD+ is stereoselective; some enzymes catalyze delivery of hydride ion to the top face of the pyridine ring, others to the bottom face10-62 Oxidation of Glycols Glycols are cleaved by oxidation with periodic acid, HIO4 OH + OH cis-1,2-Cyclohexanediol HIO 4 Periodic acid CHO CHO Hexanedial + HIO 3 Iodic acid 10-63 Oxidation of Glycols The mechanism of periodic acid oxidation of a glycol is divided into two steps Step 1: formation of a cyclic periodate O O C OH + O C OH I O C O OH I C OH + H2 O O O A cyclic periodate Step 2: redistribution of electrons within the fivemembered ring O O C O C I C O OH + I OH C O O O O 10-64 Oxidation of Glycols • this mechanism is consistent with the fact that HIO4 oxidations are restricted to glycols that can form a five-membered cyclic periodate • glycols that cannot form a cyclic periodate are not oxidized by HIO4 OH OH HO OH The trans isomer is unreactive toward periodic acid O HIO4 O The cis isomer forms a cyclic periodate and is cleaved 10-65 Thiols: Structure The functional group of a thiol is an SH (sulfhydryl) group bonded to an sp3 hybridized carbon The bond angle about sulfur in methanethiol is 100.3°, which indicates that there is considerably more p character to the bonding orbitals of divalent sulfur than there is to oxygen 10-66 Nomenclature IUPAC names: • the parent is the longest chain that contains the -SH group • change the suffix -e to -thiol • when -SH is a substituent, it is named as a sulfanyl group Common names: • name the alkyl group bonded to sulfur followed by the word mercaptan OH HS 2-Sulfanylethanol 1-Butanethiol 2-Methyl-1-propanethiol (2-Mercaptoethanol) (Butyl mercaptan) (Isobutyl mercaptan) SH SH 10-67 Thiols: Physical Properties Because of the low polarity of the S-H bond, thiols show little association by hydrogen bonding • they have lower boiling points and are less soluble in water than alcohols of comparable MW Thiol bp (°C) Methanethiol 6 Ethanethiol 35 1-Butanethiol 98 bp (°C) Alcohol Methanol 65 Ethanol 78 1-Butanol 117 • the boiling points of ethanethiol and its constitutional isomer dimethyl sulfide are almost identical CH3 CH2 SH Ethanethiol (bp 35°C) CH3 SCH3 Dimethyl sulfide (bp 37°C) 10-68 Thiols: Physical Properties Low-molecular-weight thiols = STENCH • the scent of skunks is due primarily to these two thiols SH SH 2-Butene-1-thiol 3-Methyl-1-butanethiol (Isopentyl mercaptan) • a blend of low-molecular weight thiols is added to natural gas as an odorant; the two most common of these are SH SH 2-Methyl-2-propanethiol 2-Propanethiol (tert-Butyl mercaptan) (Isopropyl mercaptan) 10-69 Thiols: preparation The most common preparation of thiols depends on the very high nucleophilicity of hydrosulfide ion, HS- SN 2 + + CH3 (CH2 ) 8 CH2 I Na SH ethanol Sodium 1-Iododecane hydrosulfide O + - - + Na SH + ICH2 CO Na Sodium Sodium hydrosulfide iodoacetate SN 2 + - CH3 (CH2 ) 8 CH2 SH + Na I 1-Decanethiol O - + + - HSCH2 CO Na + Na I Sodium mercaptoacetate (Sodium thioglycolate) 10-70 Thiols: acidity Thiols are stronger acids than alcohols CH3 CH2 OH + H2 O CH3 CH2 O + H3 O - + pKa = 15.9 CH3 CH2 SH + H2 O CH3 CH2 S - + H3 O+ pKa = 8.5 • when dissolved an aqueous NaOH, they are converted completely to alkylsulfide salts + CH3 CH2 SH + Na OH pK a 8.5 (Stronger acid) CH3 CH2 S- Na+ + H2 O pKa 15.7 (Weaker acid) 10-71 Thiols: oxidation The sulfur atom of a thiol can be oxidized to several higher oxidation states [O] R-S-H A thiol [O] R-S-S-R A disulfide O R-S-OH A sulfinic acid [O] O R-S-OH O A sulfonic acid • the most common reaction of thiols in biological systems in interconversion between thiols and disulfides, -S-S2 RSH + A thiol 1 O 2 2 RSSR + H2 O A disulfide 10-72 Alcohols and Thiols End of Chapter 10 10-73