* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Aldehydes And Ketones
Survey
Document related concepts
Enantioselective synthesis wikipedia , lookup
Kinetic resolution wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Volatile organic compound wikipedia , lookup
Metal carbonyl wikipedia , lookup
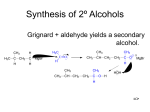
George S. Hammond wikipedia , lookup
Petasis reaction wikipedia , lookup
Elias James Corey wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aldol reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Transcript
Organic Lecture Series Aldehydes And Ketones Chap 16 1 Organic Lecture Series IUPAC names • the parent alkane is the longest chain that contains the carbonyl group • for ketones, change the suffix -e to -one • number the chain to give C=O the smaller number • the IUPAC retains the common names acetone, acetophenone, and benzophenone O Prop anone (Aceton e) O O A cetophen on e Benzophen on e Commit to memory O 1-Phenyl-1-pen tanone 2 Common Names Organic Lecture Series – for an aldehyde, the common name is derived from the common name of the corresponding carboxylic acid O HCH Formaldehyde O HCOH Formic acid O CH3 CH A cetaldeh yd e O CH3 COH Acetic acid – for a ketone, name the two alkyl or aryl groups bonded to the carbonyl carbon and add the word ketone O O O Ethyl isopropyl ketone D iethyl ketone D icyclohexyl ketone 3 Organic Lecture Series Drawing Mechanisms • Use double-barbed arrows to indicate the flow of pairs of e• Draw the arrow from higher e- density to lower e- density i.e. from the nucleophile to the electrophile • Removing e- density from an atom will create a formal + charge • Adding e- density to an atom will create a formal - charge • Proton transfer is fast (kinetics) and usually reversible 4 Organic Lecture Series Reaction Themes One of the most common reaction themes of a carbonyl group is addition of a nucleophile to form a tetrahedral carbonyl addition compound (intermediate). O R Nu - C + O Nu - C R R R Tetrahedral carbonyl add ition comp ou nd 5 Organic Lecture Series Reaction Themes A second common theme is reaction with a proton or other Lewis acid to form a resonancestabilized cation-– protonation increases the electron deficiency of the carbonyl carbon and makes it more reactive toward nucleophiles R R B fast B + C O + H-B R R + C O H R - R + H-Nu + + C O H R + C O H R O-H s low Nu C + H-B R R Tetrah edral carbonyl addition compoun d 6 Organic Lecture Series – often the tetrahedral product of addition to a carbonyl is a new chiral center – if none of the starting materials is chiral and the reaction takes place in an achiral environment, then enantiomers will be formed as a racemic mixture Ap proach from the top face Nu Nu OH O Nu - R R R' C O H3 O+ R' + R' R R R' + R' R OH O A pproach from the bottom face Nu A n ew chiral cen ter is created Nu A racemic mixture 7 Organic Lecture Series Addition of C Nucleophiles Addition of carbon nucleophiles is one of the most important types of nucleophilic additions to a C=O group – a new carbon-carbon bond is formed in the process – Focus on addition of these carbon nucleophiles: RMgX A Grignard reagent RLi RC C An organolithium An alkyne reagent anion - C N Cyanide ion 8 Organic Lecture Series Grignard Reagents – addition of a Grignard reagent to formaldehyde followed by H3O+ gives a 1° alcohol O CH 3 CH2 -MgBr + H-C-H ether Formaldehyde - O [ MgBr] + HCl H2 O CH3 CH2 -CH2 A magn esium alkoxide OH 2+ CH3 CH2 -CH2 + Mg 1-Prop anol (a 1° alcoh ol) 9 Organic Lecture Series Grignard Reagents – addition to any other RCHO gives a 2°alcohol MgBr O + ether H Acetaldehyde (an ald ehyde) - O [ MgBr] + OH HCl H2 O A magn esiu m alk oxid e + Mg2 + 1-Cycloh exyleth anol (a 2° alcohol; (racemic) 10 Organic Lecture Series Grignard Reagents – addition to a ketone gives a 3°alcohol O Ph-MgBr Ph enylmagnesiu m b romide eth er + Aceton e (a ketone) - O [ MgBr] Ph A magnes ium alk oxid e + OH HCl H2 O + Mg 2+ Ph 2-Phenyl-2-propanol (a 3° alcohol) 11 Organic Lecture Series Problem: 2-phenyl-2-butanol can be synthesized by three different combinations of a Grignard reagent and a ketone. Show each combination. OH C-CH2 CH3 CH3 12 Organic Lecture Series A Simple Retrosynthetic Analysis O CH3 OH CH3 O CH3 O CH3 13 Organic Lecture Series 14 Organic Lecture Series Addition Reactions to Carbonyl Compounds Note: Water can also be added to ketones & aldehydes. 15 Organic Lecture Series Organolithium Reagents Organolithium compounds are generally more reactive in C=O addition reactions than RMgX, and typically give higher yields O Li O- Li+ HCl H2 O + Phenyl- 3,3-D imeth yl-2lithiu m butan on e OH A lithiu m alk oxid e (racemic) 3,3-D imethyl-2-phen yl2-bu tanol (racemic) 16 Organic Lecture Series Salts of Terminal Alkynes • Addition of an alkyne anion followed by H3O+ gives an α-acetylenic alcohol • Note: this is a 2-C homologation O HC C O - Na + HCl H2 O - HC C: Na + + S od ium acetylid e HC C OH Cycloh exanone A sodium alkoxide 1-Eth yn ylcyclohexan ol Homologation is a term used for extending a carbon chain 17 Organic Lecture Series Oxidation of Terminal Alkynes O H2 O HO C CH HO α CCH3 H2 SO4 , HgSO4 An α-hydroxyketone 1. (sia) 2 BH 2. H2 O2 , NaOH O α HO CH2 CH β A β-hydroxyaldehyde (these reactions are from O-chem I) Pg 285: use of (sia)2BH 18 Organic Lecture Series Addition of HCN • HCN adds to the C=O group of an aldehyde or ketone to give a cyanohydrin • Cyanohydrin: a molecule containing an OH group and a -CN group bonded to the same carbon O C HO + HCN C N C H3C H3C H Acetaldehyde H 2-Hydroxypropanenitrile (Acetaldehyde cyanohydrin) 19 Organic Lecture Series Addition of HCN • Mechanism of cyanohydrin formation – Step 1: nucleophilic addition of cyanide to the carbonyl carbon H3 C • • H3 C C O + C N H3 C O - C H3 C C N – Step 2: proton transfer from HCN gives the cyanohydrin and regenerates cyanide ion H3 C O - H3 C + H C N H3 C C N pKa=9.3 O-H + C - • • C H3 C C N C N 20 Organic Lecture Series Cyanohydrins – catalytic reduction of the cyano group gives a 1°amine OH OH CHC N + 2 H2 Ben zaldeh yd e cyanohydrin (racemic) Ni CHCH2 NH2 β α 2-Amino-1-ph enylethanol (racemic) β−Phenethylamines 21 Organic Lecture Series Biosynthesis of Dopamine L-Phenylalanine → L-Tyrosine → L-DOPA → Dopamine 22 Organic Lecture Series β−Phenethylamines (Natural) NH2 HO OH OH NH2 Dopamine HO OH Norepinephrine 23 β−Phenethylamines (synthetic) Organic Lecture Series H N CH3 CH3 N-methylamphetamine NH2 CH3 amphetamine 24 Organic Lecture Series Overall Synthetic Transformation of Wittig Reagents & Its Variations R R' O C Ketone Can Be Cyclic Alkenes or Olefins 25 Organic Lecture Series Wittig Reaction The Wittig reaction is a very versatile synthetic method for the synthesis of alkenes (olefins) from aldehydes and ketones O + + Ph3 P-CH2 Cycloh exanone A p hosp honium ylide CH2 + Ph 3 P=O MethyleneTrip hen ylcycloh exane phosp hine oxide Ylides are reagents (or reactive intermediates) which have adjacent charges: Ph3P CH2 Ph3P CH2 26 Organic Lecture Series Phosphonium ylides Phosphonium ylides are formed in two steps: Step 1: nucleophilic displacement of iodine by triphenylphosphine Ph 3 P SN 2 + CH3 -I Trip henylph os phin e + Ph3 P-CH3 I Methyltriphen ylp hosph on ium iod ide (an alk yltriph enylph os phin e salt) Step 2: treatment of the phosphonium salt with a very strong base, most commonly BuLi, NaH, or NaNH2 - CH3 CH2 CH2 CH2 Li Bu tyllith ium + + + H-CH2 -PPh3 I+ CH3 CH2 CH2 CH3 + - CH2 -PPh3 + LiI Bu tane A phosp honium ylide Not exam material 27 Organic Lecture Series Wittig Reaction Phosphonium ylides react with the C=O group of an aldehyde or ketone to give an alkene Step 1: nucleophilic addition of the ylide to the electrophilic carbonyl carbon O CR2 + Ph3 P - :O + - Ph3 P CH2 CR2 O CR2 CH2 Ph3 P CH2 An oxaph os ph etane A betaine Step 2: decomposition of the oxaphosphatane O CR2 Ph3 P CH2 Ph3 P=O + Triphen ylp hosph ine oxide R2 C=CH2 An alkene 28 Organic Lecture Series Wittig Reaction O + Ph3 P + Acetone 2-Methyl-2-hepten e O + Ph3 P Ph Ph 3 P=O + Ph + Ph P=O Ph 3 (Z)-1-Ph enyl-2- (E)-1-Phen yl-2b utene b utene (87%) (13%) H Phen ylacetald ehyde Resonance stabilized Wittig reagent: O H Ph O Phen ylacetald ehyde + Ph3 P O OEt Ph + Ph 3 P=O OEt Eth yl (E)-4-phen yl-2-b utenoate (only the E is omer is formed) 29 Organic Lecture Series Addition Reactions to Carbonyl Compounds Note: Water can be added to ketones & aldehydes. 30 Organic Lecture Series Addition of H2O to Carbonyls Addition of water (hydration) to the carbonyl group of an aldehyde or ketone gives a geminal diol, commonly referred to a gemdiol – gem-diols are also referred to as hydrates C O + acid or b ase OH C H2 O OH A hydrate (a gem-diol) Carbonyl group of an aldeh yd e or k eton e 31 Organic Lecture Series Addition of H2O to Carbonyls – when formaldehyde (g) is dissolved in water at 20°C, the carbonyl group is more than 99% hydrated H H O + H2 O H Formaldehyde OH OH H Formald ehyde h yd rate (>99%) – the equilibrium concentration of a hydrated ketone is considerably smaller O + H2 O Acetone (99.9%) OH OH 2,2-Propan ediol (0.1%) 32 Organic Lecture Series Addition of Alcohols to Carbonyls • Addition of one molecule of alcohol to the C=O group of an aldehyde or ketone gives a hemiacetal • Hemiacetal: a molecule containing an OH and an -OR or -OAr bonded to the same carbon acid or base O + H-OR OH OR A hemiacetal 33 Organic Lecture Series Formation of a hemiacetal-- base catalyzed – Step 1: proton transfer from HOR gives an alkoxide B - + H OR fas t and reversib le B H + - Na+ -OCH3 OR – Step 2: attack of RO- on the carbonyl carbon O CH3 -C-CH3 + O:– – :O-R CH3 -C-CH3 OR – Step 3: proton transfer from the alcohol to O- gives the hemiacetal and generates a new base catalyst – O: CH3 -C-CH3 + H OR OR OH CH3 -C-CH3 + - OR OR 34 Organic Lecture Series Formation of a hemiacetal --acid catalyzed Step 1: proton transfer to the carbonyl oxygen O CH3 -C-CH3 + H-A + fas t and reversib le O H CH3 -C-CH3 + A - Step 2: attack of ROH on the carbonyl carbon + H O O-H CH3 -C-CH 3 O+ H R CH3 -C-CH3 + H-O-R Step 3: proton transfer from the oxonium ion to A- gives the hemiacetal and generates a new acid catalyst OH A - CH3 -C-CH3 + O H R OH CH3 -C-CH 3 + H-A OR 35 Organic Lecture Series Addition of Alcohols to Carbonyls • Hemiacetals react with alcohols to form acetals Acetal: a molecule containing two -OR or -OAr groups bonded to the same carbon OH + + H-OR OR A hemiacetal H OR OR acetal + H2 O Nota Bene: acetals are STABLE in base 36 Organic Lecture Series Begin Mechanism from the hemiacetal stage: Step 1: proton transfer from HA gives an oxonium ion H + H O HO R-C-OCH3 + H A R-C-OCH3 H - + A: H An oxonium ion Step 2: loss of water gives a resonancestabilized cation + H O R-C OCH3 H + R-C OCH3 + R-C OCH3 + H2 O H H A resonan ce-stabilized cation H 37 Organic Lecture Series Step 3: reaction of the cation (an electrophile) with methanol (a nucleophile) gives the conjugate acid of the acetal H + CH3 -OH + R-C OCH3 + CH 3 O R-C OCH3 H A p rotonated acetal H Step 4: proton transfer to A- gives the acetal and generates a new acid catalyst H + CH3 O A: + R-C OCH3 H (4) OCH3 R-C-OCH3 + H-A H An acetal 38 Organic Lecture Series Addition of Alcohols to Carbonyls – with ethylene glycol and other glycols, the product is a five-membered cyclic acetal – this a method of “protecting” ketones O + HO + OH O H O Cyclic acetal + H2 O 39 Organic Lecture Series Dean-Stark Trap 40 Organic Lecture Series Acetals as Protecting Groups • How to bring about a Grignard reaction between these compounds: O OH O H Br + Benzald ehyde H ?? 4-Bromobutanal O O H 5-Hydroxy-5-phen ylpen tanal (racemic) This Grignard cannot be made!! BrMg H 41 Organic Lecture Series Acetals as Protecting Groups • a Grignard reagent prepared from 4bromobutanal will self-destruct (decompose). – first protect the -CHO group as an acetal: O Br H + HO OH H O + Br O A cyclic acetal + H2 O – then prepare the Grignard reagent: O- MgBr+ O O Br 1 . Mg, ether O O 2 . C6 H5 CHO A chiral magnesiu m alk oxide (produced as a racemic mixtu re) – hydrolysis (not shown) gives the target molecule 42 Organic Lecture Series Addition of Nitrogen Nucleophiles • Ammonia, 1°aliphatic amines, and 1°aromatic amines react with the C=O group of aldehydes and ketones to give imines (Schiff bases) • Water is removed by Dean-Stark trap or chemical dehydration (e.g. molecular sieves) O CH3 CH + H2 N Acetaldehyde H + CH3 CH =N Aniline O Cyclohexanone + + H2 O An imine (a Schiff base) H N H3 + Ammonia N H + H2 O An imine (a Schiff base) 43 Organic Lecture Series Addition of Nitrogen Nucleophiles – a value of imines is that the carbon-nitrogen double bond can be reduced to a carbonnitrogen single bond O + H+ - H2 O H2N Cyclohexanone Cyclohexylamine H N H2 / Ni (An imine) N Dicyclohexylamine Does not have to isolated 44 Organic Lecture Series Addition of Nitrogen Nucleophiles • Secondary amines react with the C=O group of aldehydes and ketones to form enamines (alkene and amine) + O Cyclohexanone H H-N + + H2 O N Piperidine (a secondary amine) An enamine – the mechanism of enamine formation involves formation of a tetrahedral carbonyl addition compound followed by its acid-catalyzed dehydration 45 Organic Lecture Series Acidity of α-Hydrogens Hydrogens alpha to a carbonyl group are more acidic than hydrogens of other hydrocarbons (e.g. alkanes, alkenes, aromatic). O H H α H α H 46 Organic Lecture Series H 3O + + A - H A + H 2O K eq = Note: l and s are not used in K [H 2 O ] K e q = Ka = [H 3 O + ] [A - ] [H A ] [H 2 O ] [H 3 O + ] [A - ] [H A ] Freshman Flashback!! [H 3 O + ] [A - ] [H A ] 47 Organic Lecture Series Acidity of α-Hydrogens Hydrogens alpha to a carbonyl group are more acidic than hydrogens of alkanes, alkenes, and alkynes but less acidic than the hydroxyl hydrogen of alcohols pKa = -log Ka Type of Bond pKa CH3 CH2 O-H O CH3 CCH2 -H CH3 C C-H CH2 =CH-H CH3 CH2 -H 16 20 25 44 51 48 Organic Lecture Series α-Hydrogens are more acidic because the enolate anion is stabilized by: 1. delocalization of its negative charge (resonance effect) 2. the electron-withdrawing inductive effect of the adjacent electronegative oxygen O- O O CH3 -C-CH2 - H + :A - CH3 -C=CH2 + H-A CH3 -C CH2 Resonance-stabilized en olate anion 49 Organic Lecture Series Keto-Enol Tautomerism – protonation of the enolate anion on oxygen gives the enol form*; protonation on carbon gives the keto form O - O- CH3 - C-CH2 CH3 - C= CH2 Enolate anion O H- A A - + CH3 - C-CH3 Keto form H- A OH CH3 - C= CH2 + A - Enol form *Enol: made from 2 functional groups-alkene and alcohol 50 Organic Lecture Series Keto-Enol Tautomerism – acid-catalyzed equilibration of keto and enol tautomers occurs in two steps Step 1: proton transfer to the carbonyl oxygen • • O CH3 -C-CH3 + H-A Keto form fas t and reversib le + O H CH3 -C-CH3 + A - The conju gate acid of the ketone Step 2: proton transfer to the base A+ O H slow - CH3 -C-CH2 -H + :A OH CH3 -C=CH2 + H-A En ol form 51 Organic Lecture Series Keto-Enol Tautomerism Keto-enol equilibria for simple aldehydes and ketones lie far toward the keto form Keto form Enol form O OH CH2 = CH CH3 CH O OH CH3 CCH3 CH3 C= CH2 O % Enol at Equilibrium 6 x 10-5 6 x 10-7 OH 1 x 10-6 O OH 4 x 10-5 52 O O C C R Oxidation R OH R C a rb o x y lic A c id s & E s te rs OR' O O C C H Organic Lecture Series R K e to n e s & A ld e h y d e s R' OH A lc o h o ls C R R' H H H y d r o c a rb o n (lo w e s t o x id a tio n ) C R R' 53 H Organic Lecture Series Oxidation from O-Chem I 54 Organic Lecture Series Oxidation of Aldehydes Aldehydes are oxidized to carboxylic acids by a variety of oxidizing agents, including *H2CrO4 CHO H 2 Cr O4 COOH Hexanal Hexanoic acid 55 * See 10.8 for Jones reagent & PCC: pyridine+•ClCrO3- Organic Lecture Series Oxidation of Aldehydes • They are also oxidized by Ag(I) – in one method, a solution of the aldehyde in aqueous ethanol or THF is shaken with a slurry of silver oxide O CH CH3 O HO Vanillin T HF, H 2 O + A g2 O N aOH HCl H2 O CH3 O O COH + Ag HO Vanillic acid 56 Organic Lecture Series Oxidation of Aldehydes Aldehydes are oxidized by O2 in a radical chain reaction – liquid aldehydes are so sensitive to air that they must be stored under N2 O 2 O CH + O2 2 COH Benzaldehyde Benzoic acid 57 Organic Lecture Series Reduction – aldehydes can be reduced to 1°alcohols – ketones can be reduced to 2°alcohols – the C=O group of an aldehyde or ketone can be reduced to a -CH2- group Aldehydes Can Be Reduced to Ketones Can Be Reduced to OH O RCH 2 OH O RCHR' RCR' RCH RCH 3 RCH 2 R' 58 Organic Lecture Series Metal Hydride Reduction The most common laboratory reagents for the reduction of aldehydes and ketones are NaBH4 and LiAlH4 – both reagents are sources of hydride ion, H: - a very powerful nucleophile H H Al H H L i+ H H B H Na+ H Hydride H Lithium Aluminum Hydride LAH Sodium Borohydride NaBH4 59 Organic Lecture Series Sodium Borohydride Reduction – reductions with NaBH4 are most commonly carried out in aqueous methanol, in pure methanol, or in ethanol – one mole of NaBH4 reduces four moles of aldehyde or ketone O methanol 4 RCH + NaBH4 - + ( RCH2 O) 4 B Na A tetraalkyl borate H2 O 4 RCH2 OH + borate salts 60 Organic Lecture Series Sodium Borohydride Reduction • The key step in metal hydride reduction is transfer of a hydride ion to the C=O group to form a tetrahedral carbonyl addition compound This H comes from w ater du rin g hydrolys is H O + Na H-B-H + R-C-R' H O BH3 Na + O-H R-C-R' H2 O R-C-R' H H This H comes from the hydride reducing agen t Not exam material 61 Organic Lecture Series LAH Reduction – unlike NaBH4, LiAlH4 reacts violently with water, methanol, and other protic solvents – reductions using it are carried out in dry (anhydrous) diethyl ether or tetrahydrofuran (THF) O ether 4 RCR + LiAlH4 - ( R2 CHO) 4 Al Li A tetraalk yl alu min ate OH H2 O + + - H or OH 4 RCHR + aluminum salts 62 Organic Lecture Series Catalytic Reduction • Catalytic reductions are generally carried out at from 25°to 100°C and 1 to 5 atm H2 OH O + H2 Pt 25 o C, 2 atm Cyclohexanone Cyclohexanol O H trans- 2-Butenal (Crotonaldehyde) 2 H2 Ni OH 1-Butanol Note: Both the olefin and the carbonyl are reduced 63 Organic Lecture Series 64 Organic Lecture Series Catalytic Reduction • A carbon-carbon double bond may also be reduced under these conditions: O 2 H2 H Ni trans-2-Butenal (Crotonaldehyde) OH 1-Butanol – by careful choice of experimental conditions, it is often possible to selectively reduce a carbon-carbon double in the presence of an aldehyde or ketone O 1 . NaBH4 RCH=CHCR' 2 . H2 O OH RCH=CHCHR' O O RCH=CHCR' + H2 Rh RCH2 CH2 CR' 65 Organic Lecture Series Clemmensen Reduction – refluxing an aldehyde or ketone with amalgamated zinc in concentrated HCl converts the carbonyl group to a methylene group – Classic reaction but harsh conditions limit its use OH O OH Zn( H g) , HCl 66 Organic Lecture Series Wolff-Kishner Reduction – in the original procedure, the aldehyde or ketone and hydrazine are refluxed with KOH in a high-boiling solvent – the same reaction can be brought about using hydrazine and potassium tert-butoxide in DMSO (Dimethyl sulfoxide) O + H2 NN H2 Hydrazine KOH diethylene glycol (reflux) + N 2 + H2 O 67 Organic Lecture Series Racemization • Racemization at an α-carbon may be catalyzed by either acid or base • Once stereochemistry is set, this is usually an undesirable side reaction. O Ph (R)-3-Phenyl-2b utanone OH Ph An achiral enol O Ph (S)-3-Phenyl-2butan one 68 Organic Lecture Series α-Halogenation α-Halogenation: aldehydes and ketones with at least one α-hydrogen react at an αcarbon with Br2 and Cl2 O O Br + Br2 + HBr CH3 COOH Acetop henone α -Bromoacetoph enone – reaction is catalyzed by both acid and base – Caution!! These are lachrymators 69 Organic Lecture Series α-Halogenation • Acid-catalyzed α-halogenation Step 1: acid-catalyzed enolization- forms the enol OH R'-C-C-R H-O slow R C R' R C R Step 2: nucleophilic attack of the enol on halogen R H-O C R' C H O + Br R Br fast Br C C R R' + - Br: R Step 3: (not shown) proton transfer to solvent completes the reaction 70 Organic Lecture Series α-Halogenation • Base-promoted α-halogenation Step 1: formation of an enolate anion OH slow - R'-C-C-R + :OH O C O: - R •• C - R C + H2 O C R' R' R R Res on ance-stab ilized enolate anion R Step 2: nucleophilic attack of the enolate anion on halogen O: C R' R O + Br C R Br fast Br C C R + R' α-Halogenation Br R 71 Organic Lecture Series • Acid-catalyzed α-halogenation: – introduction of a second halogen is slower than the first – introduction of the electronegative halogen on the αcarbon decreases the basicity of the carbonyl oxygen toward protonation • Base-promoted α-halogenation: – each successive halogenation is more rapid than the previous one – the introduction of the electronegative halogen on the α-carbon increases the acidity of the remaining αhydrogens and, thus, each successive α-hydrogen is removed more rapidly than the previous one 72