* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
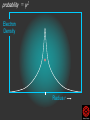
Download probability = ψ 2
Woodward–Hoffmann rules wikipedia , lookup
Photoelectric effect wikipedia , lookup
Marcus theory wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Hartree–Fock method wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Electron scattering wikipedia , lookup
Physical organic chemistry wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Chemical bond wikipedia , lookup
Coupled cluster wikipedia , lookup
Molecular orbital wikipedia , lookup
Heat transfer physics wikipedia , lookup
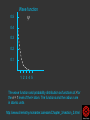
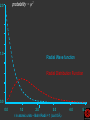
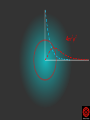
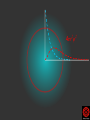
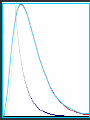
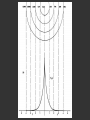
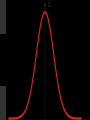
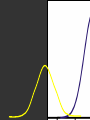
Hψ = E ψ Hamiltonian for the H atom The wave function is usually represented by ψ 2 The electron density is given by ψ probability = ψ2 Electron Density Radius r Wave function 0.5 ψ 0.4 0.3 0.2 0.1 1 2 3 4 5 The wave function and probability distribution as functions of r for the n = 1 level of the H atom. The functions and the radius r are in atomic units http://www.chemistry.mcmaster.ca/esam/Chapter_3/section_2.html Wave function 0.5 0.3 Electron density ψ 0.4 ψ2 0.2 0.3 0.2 0.1 0.1 1 2 3 4 5 1 2 3 4 The wave function and probability distribution as functions of r for the n = 1 level of the H atom. The functions and the radius r are in atomic units http://www.chemistry.mcmaster.ca/esam/Chapter_3/section_2.html probability = ψ2 2.0 1.0 Radial Wave function Radial Distribution Function 0.0 0.0 1.0 2.0 3.0 r in atomic units – Bohr Radii = 1 (ca 0.5Ǻ) 4.0 5.0 4πr2ψ2 4πr2ψ2 4πr2ψ2 32 5 4 3 2 1 0 1 2 3 4 5 % 32 74 % 5 4 3 2 1 0 1 2 3 4 5 32 74 93 % 5 4 3 2 1 0 1 2 3 4 5 32 74 93 99 % 5 4 3 2 1 0 1 2 3 4 5 Wave function Electron density ψ ψ2 The wave function and probability distribution as functions of r for the n = 1 level of the H atom. The functions and the radius r are in atomic units http://www.chemistry.mcmaster.ca/esam/Chapter_3/section_2.html Radial Distribution Function 2.0 1.0 0.0 0.0 1.0 2.0 3.0 4.0 5.0 Hydrogen. The two electrons in the hydrogen molecule may both be accommodated in the 1sg orbital if their spins are paired and the molecular orbital configuration for H2 is 1sg2. Since the 1sg orbital is the only occupied orbital in the ground state of H2, the density distribution shown previously in Fig. 62 for H2 is also the density distribution for the 1sg orbital when occupied by two electrons. The remarks made previously regarding the binding of the nuclei in H2 by the molecular charge distribution apply directly to the properties of the 1sg charge density. Because it concentrates charge in the binding region and exerts an attractive force on the nuclei the 1sg orbital is classified as a bonding orbital. http://www.chemistry.mcmaster.ca/esam/Chapter_3/section_2.html Hydrogen. II Excited electronic configurations for molecules may be described and predicted with the same ease within the framework of molecular orbital theory as are the excited configurations of atoms in the corresponding atomic orbital theory. For example, an electron in H2 may be excited to any of the vacant orbitals of higher energy indicated in the energy level diagram. The excited molecule may return to its ground configuration with the emission of a photon. The energy of the photon will be given approximately by the difference in the energies of the excited orbital and the 1sg ground state orbital. Thus molecules as well as atoms will exhibit a line spectrum. The electronic line spectrum obtained from a molecule is, however, complicated by the appearance of many accompanying side bands. These have their origin in changes in the vibrational energy of the molecule which accompany the change in electronic energy. The nucleus is the very dense region consisting of nucleons (protons and neutrons) at the center of an atom. Almost all of the mass in an atom is made up from the protons and neutrons in the nucleus, with a very small contribution from the orbiting electrons. The diameter of the nucleus is in the range of 1.6 fm (1.6 × 10−15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium). These dimensions are much smaller than the diameter of the atom itself, by a factor of about 23,000 (uranium) to about 145,000 (hydrogen). The branch of physics concerned with studying and understanding the atomic nucleus, including its composition and the forces which bind it together, is called nuclear physics. http://tannerm.com/diatomic.htm