* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 5.3_Matter_Waves
Quantum chaos wikipedia , lookup
Quantum entanglement wikipedia , lookup
Quantum mechanics wikipedia , lookup
Bell's theorem wikipedia , lookup
Quantum potential wikipedia , lookup
Renormalization wikipedia , lookup
ATLAS experiment wikipedia , lookup
History of quantum field theory wikipedia , lookup
Relational approach to quantum physics wikipedia , lookup
Interpretations of quantum mechanics wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Photoelectric effect wikipedia , lookup
Eigenstate thermalization hypothesis wikipedia , lookup
Identical particles wikipedia , lookup
Introduction to gauge theory wikipedia , lookup
Quantum state wikipedia , lookup
Future Circular Collider wikipedia , lookup
Canonical quantization wikipedia , lookup
Quantum logic wikipedia , lookup
Wave packet wikipedia , lookup
Quantum vacuum thruster wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Uncertainty principle wikipedia , lookup
Elementary particle wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Old quantum theory wikipedia , lookup
Photon polarization wikipedia , lookup
Electron scattering wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Quantum tunnelling wikipedia , lookup
Probability amplitude wikipedia , lookup
Double-slit experiment wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
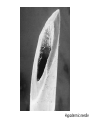
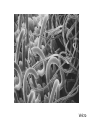
QUANTUM MECHANICS Matter Waves 1. De Broglie and Schrodinger 2. Electron microscopes 3. Quantum Tunneling (microscopes) Matter Waves Everything (photons, electrons, SMU students, planets, ..) has a probability wave - de Broglie Wavelength λ = h = Planck’s constant p momentum Q. What is your wavelength? Electron Waves A. About 10-35 m (Practically Unobservable) But… photons, electrons, other elementary particles can have very small p, hence observable wavelength Schrodinger’s Equation • Based on Conservation of Energy principle • Describes how probability waves move • Output is `wavefunction’ Ψ - height of the wave at any one place and time (probability is Ψ2) • Visible light -> Microscopes use lenses and mirrors to guide • Electrons -> Electron microscopes use electricity to guide Momentum larger than for visible photons, wavelength smaller, see more details E.g. cancer cell Hypodermic needle Velcro Staple Spider’s foot Mascara brush Dental drill tip Energy Barriers Classical physics – Energy needed to surmount barrier Quantum Physics – Small probability to pass through Waves can pass through `forbidden’ regions Quantum wave exists within and beyond energy barrier Probability to `tunnel’ through grows rapidly as width/height of barrier decreases QM applies to everything … including you Very (very) small probability that you can walk through walls Scanning Tunneling Microscope (STM) • Electrons quantum tunnel from tip to sample through (air) barrier • Tunneling rate (current) extremely sensitive to tip-sample separation • Measured current provides topographical map of sample surface Particle Colliders Accelerate to very large p and collide Quark particles “Image” smallest, simplest things known Elementary particles – characterized by a few numbers Atlas Detector E.g. Large Hadron Collider