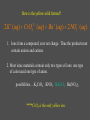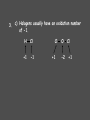* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download + H 2 O(l )
Radical (chemistry) wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Process chemistry wikipedia , lookup
Marcus theory wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Atomic theory wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Rate equation wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Oxidation state wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Transition state theory wikipedia , lookup
Acid–base reaction wikipedia , lookup
Click chemistry wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Stoichiometry wikipedia , lookup
Water splitting wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Electrolysis of water wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Chemical reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Electrochemistry wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
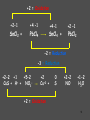
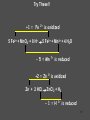
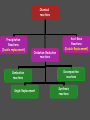
Chemistry Chapter 11 Chemical Reactions Describing Chemical Reactions Chemical Reaction- process by which one or more substances are changed into one or more different substances. Reactant- original substance Product- resulting substance Chemical Equation - Represents the identities and relative amounts of the reactants and products in a chemical reaction How do you know a chemical reaction occurred? Evidence that one or more substance has undergone a change in identity. 1. Evolution of heat and light. 2. Production of a gas 3. Formation of a precipitate (solid produced in solution that separates from the solution) Characteristics of Chemical Equations Must represent the known facts. The equation must contain the correct formulas for the reactants and products. The law of conservation of mass must be satisfied. (coefficients are used to equalize the # of moles of a substance) Word and Skeleton Equations Word equation methane + oxygen Skeleton Equation CH4(g) + O2(g) carbon dioxide + water CO2(g) + H2O(g) Balance Equation Usually balance Hydrogen and Oxygen last. CH4(g) + 2O2(g) CO2(g) + 2H2O(g) Sample Problem 1 •Write word and formula equations for the chemical reaction that occurs when solid sodium oxide is added to water at room temperature and forms sodium hydroxide (dissolved in the water). Include symbol for physical states and then balance. Answer 1 Sodium oxide + water sodium hydroxide Na2O + H20 NaOH Na2O(s) + H20(l) 2NaOH(aq) Balancing Equations Identify the names of the reactants and products and write a word equation. water hydrogen + oxygen Write the formula equation. H2O(l) H2 (g) + O2 (g) Balance the equation 1. balance 1 at a time. 2. balance atoms that are combined and appear only once on each side. 3. balance polyatomic ions as single units 4. balance H and O last. Balancing cont. 2H2O(l) H2 (g) + O2 (g) 2H2O(l) 2H2 (g) + O2 (g) Count the atoms. 2H2O(l) 2H2 (g) + O2 (g) Sample Problem 2 •The reaction of zinc with aqueous hydrochloric acid produces a solution of zinc chloride and hydrogen gas. Write a balanced equation for the reaction. Answer 2 • Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) Sample Problem 3 • Solid aluminum carbide, Al4C3, reacts with water to produce methane gas and solid aluminum hydroxide. Write a balanced chemical equation for this reaction. Answer 3 Al4C3(s) + 12H20(l) 3CH4(g) + 4Al(OH)3(s) What are the driving forces of reactions? • • • • Formation of a solid Formation of water Transfer of electrons Formation of a gas Types of Solution Reactions Precipitation reactions AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) Acid-base reactions NaOH(aq) + HCl(aq) NaCl(aq) + H2O(l) Oxidation-reduction reactions Fe2O3(s) + Al(s) Fe(l) + Al2O3(s) Precipitation Reaction K2CrO4(aq) + Ba(NO3)2 --> KNO3 + Ba(CrO4) Figure 4.13: When yellow aqueous potassium chromate is added to a colorless barium nitrate solution, yellow barium chromate precipitates. Precipitation reaction (Double Replacement) Yellow potassium chromate with colorless barium nitrate yields a yellow precipitation K2 CrO4 (aq) Ba( NO3 ) 2 (aq) product In virtually every case when a solid containing ions dissolves in water, the ions separate and move around independently. What happens when an Ionic compound Dissolves in Water? Strong electrolytes: 2 K (aq) CrO4 2 (aq) Ba 2 (aq) NO3 product How is the yellow solid formed? 2 2 K (aq) CrO4 (aq) Ba (aq) 2 NO3 (aq) 1. Ions form a compound; zero net charge. Thus the product must contain anions and cations. 2. Most ionic materials contain only two types of ions: one type of cation and one type of anion. possibilities….K2CrO4 KNO3 BaCrO4 Ba(NO3)2 ****CrO4 is the only yellow ion. What happens to the K+ and NO3-? 2 K (aq) CrO4 2 (aq) Ba (aq) 2 NO3 (aq) They are not involved in the reaction Therefore….. K2 CrO4 (aq) Ba( NO3 ) 2 (aq) Ba(CrO4 )( s) 2 K ( NO3 )(aq) Remain in solution Simple Rules for Solubility 1.Most nitrate (NO3) salts are soluble. 2.Most alkali (group 1A) salts and NH4+ are soluble. 3.Most Cl, Br, and I salts are soluble (NOT Ag+, Pb2+, Hg22+) 4.Most sulfate salts are soluble (NOT BaSO4, PbSO4, HgSO4, CaSO4) 5.Most OH salts are only slightly soluble (NaOH, KOH are soluble, Ba(OH)2, Ca(OH)2 are marginally soluble) 6.Most S2, CO32, CrO42, PO43 salts are only slightly soluble. Molecular Equation K2 CrO4 (aq) Ba( NO3 ) 2 (aq) Ba(CrO4 )( s) 2 K ( NO3 )(aq) Complete ionic equation 2 2 K (aq) CrO 4 (aq) Ba 2 (aq) NO3 (aq) BaCrO 4 ( s ) 2 K (aq ) NO3 (aq) Net ionic equation 2 CrO 4 (aq) Ba 2 (aq) BaCrO 4 ( s ) Predict the following and write the molecular and net ionic equations: 1. Aqueous sodium sulfide is mixed with aqueous copper(II)nitrate to produce… 2. Aqueous ammonium chloride and aqueous lead(II)nitrate react to form… Acid-Base Reactions • Acid substance that produces H+ ions when it is dissolved in water. • Base substance that produces hydroxide ions (OH-) in water. Product of strong acid + strong base • When a strong acid and a strong base react the chemical change that always occurs is that H+ ions react with OHto form water. • Strong acid- HCl, HNO3, H2SO4 • Strong base-NaOH, KOH Products of a strong acid/strong base reaction: 1. Water 2. Ionic compound (salt) which may precipitate or remain dissolved Aqueous nitric acid and aqueous aluminum hydroxide react to form… Molecular HNO 3 (aq ) Al (OH ) 3 (aq) H 2O(l ) Al ( NO 3 ) 3 (aq ) Ionic H (aq ) NO 3 (aq ) Al 3 (aq ) OH (aq ) H 2O (l ) Al 3 (aq ) NO 3 (aq ) Net Ionic H (aq ) OH (aq ) H 2O(l ) Weak acids and bases do not ionize. sodium hydroxide + oxalic acid → 2 NaOH + H2C2O4 (aq) Na2C2O4 + 2 H2O (l) Ionic : 2Na+ + 2OH- + H2C2O4 (aq) 2Na+ + C2O42- +2H2O (l) Net: H2C2O4 (aq) + 2 OH- C2O42- +2 H2O (l) Formation of Gases 1. CO2 2. H2S 3. NH3 (H2CO3 H2O + CO2) (sulfide salt + acid) (NH4OH NH3(aq) + H2O(l)) Ammonia solution + acetic acid Weak base + weak acid salt NH4OH NH3(aq) + H2O(l) NH3(aq) + HC2H3O2 (aq) NH4C2H3O2(aq) NH3(aq) + HC2H3O2 (aq) NH4+ (aq) + C2H3O2-(aq) Oxidation Reduction Reaction Synthesis Single replacement Combustion Decomposition Synthesis (Composition) Reactions Two or more substances combine to form a new compound. A + X AX Reaction of elements with oxygen and sulfur Reactions of metals with Halogens Synthesis Reactions with Oxides Synthesis Reactions of Elements w/ Oxygen and Sulfur Most metals react w/ oxygen to form oxides. They react similarly w/ sulfur to form sulfides. 2Mg(s) + O2(g) 8Ba(s) + S8(s) 2 MgO(s) 8BaS(s) Synthesis Reactions of Metals with Halogens Most group 1 and 2 metals react with the halogens 2Na(s) + Cl2(g) 2NaCl(s) Synthesis Reactions w/ Oxides Metal oxides react with water to produce metal hydroxides. CaO(s) + H2O(l) Ca(OH) 2(s) Synthesis w/ oxides cont. Many oxides of nonmetals in the upper right portion of the periodic table react w/water to produce oxyacids. SO2(g) + H2O(l) 2H2SO3(aq) + O 2(g) H2SO3(aq) 2H2SO4(aq) Synthesis reactions cont. Certain metal oxides and nonmetal oxides react w/ each other to form salts. CaO(s) + SO2(g) CaSO3(s) Decomposition Reactions A single compound undergoes a reaction that produces two or more simpler substances AX A + X Decomposition of: Binary compounds Metal carbonates Metal hydroxides Metal chlorates Oxyacids 2H2O(l ) 2H2(g) + O2(g) CaCO3(s) CaO(s) + CO2(g) Ca(OH)2(s) CaO(s) + H2O(g) 2KClO3(s) 2KCl(s) + 3O2(g) H2CO3(aq) CO2(g) + H2O(l ) Formation of a Gas • Zn(s) + 2HCl(aq) H2(g) + ZnCl2(aq) • How would you classify this reaction? precip, acid-base, redox It is an electron transfer process Also called... Single replacement redox Single Replacement Reactions A + BX AX + B BX + Y BY + X Replacement of: Metals by another metal Hydrogen in water by a metal Hydrogen in an acid by a metal Halogens by more active halogens The Activity Series of the Metals Lithium Potassium Calcium Sodium Magnesium Aluminum Zinc Chromium Iron Nickel Lead Hydrogen Bismuth Copper Mercury Silver Platinum Gold Metals can replace other metals provided that they are above the metal that they are trying to replace. Metals above hydrogen can replace hydrogen in acids. Metals from sodium upward can replace hydrogen in water The Activity Series of the Halogens Fluorine Chlorine Bromine Iodine Halogens can replace other halogens in compounds, provided that they are above the halogen that they are trying to replace. ??? + Cl2(g) 2NaCl(s) + F2(g) 2NaF(s) ???Reaction MgCl2(s) + Br2(g) No Combustion Reactions A substance combines with oxygen, releasing a large amount of energy in the form of light and heat. Usually produce CO2 and H20 (special form of oxidation-reduction) The burning of natural gas, wood, gasoline C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) Oxidation-Reduction (Redox) Reactions Oxidation Number (State): A value which indicates whether an atom is neutral, electronrich, or electron-poor. Rules for Assigning Oxidation Numbers 1. An atom in its elemental state has an oxidation number of 0. Na H2 Br2 S Oxidation number 0 Ne 2. A monatomic ion has an oxidation number identical to its charge. Na1+ Ca2+ Al3+ Cl1- O2- +1 +2 +3 -1 -2 3. An atom in a polyatomic ion or in a molecular compound usually has the same oxidation number it would have if it were a monatomic ion. a) Hydrogen can be either +1 or -1. H +1 O 1- -2 H Ca -1 +2 H -1 b) Oxygen usually has an oxidation number of -2. H O H H O O H +1 -2 +1 with halogens it’s (+2) OF2 +1 -1 -1 peroxide +1 3. c) Halogens usually have an oxidation number of -1. H Cl +1 -1 Cl +1 O -2 Cl +1 4. The sum of the oxidation numbers is 0 for a neutral compound and is equal to the net charge for a polyatomic ion. H2SO3 +1 x -2 Cr2O72x -2 2(+1) + x + 3(-2) = 0 (net charge) x = +4 2(x) + 7(-2) = -2 (net charge) x = +6 Assign Oxidation #s • UF6 • H2SO4 • ClO3- Examples - assigning oxidation numbers Assign oxidation states to all elements: SO3 SO42- K NH3 MnO4 Cr2O72- CH3OH PO43- ClO3 HSO3 Cu H2 + 53 The chemical changes that occur when electrons are transferred between reactants are called oxidation – reduction reactions 54 Oxidation and Reduction • Oxidation: complete or partial loss of electrons or gain of oxygen. Not all reactions involve burning. example: Iron rusting: Fe + O2 Fe2O3 Bleaching: 4 NaClO + 2 H2O → 4 Na+ + 4 OH- + 2 Cl2 + O2 The O2 then attaches to stains Reduction: opposite of oxidation complete or partial gain of electrons or loss of oxygen. Iron rusting: Fe + O2 Fe2O3 Bleaching: 4 NaClO + 2 H2O → 4 Na+ + 4 OH- + 2 Cl2 + O2 OIL RIG • Oxidation involves loss of electrons • Reduction involves gain of electrons LEO says GER • For every oxidation there is a reduction. The electrons are transferred from one spies to the other. REDOX +2 = Oxidation +2 -1 +4 -1 SnCl2 + PbCl4 +4 -1 +2 -1 SnCl4 + PbCl2 -2 = Reduction -3 = Reduction +2-2 +1 +5-2 CuS + H+ + NO3- +2 Cu+2 + 0 S +2-2 NO +1 -2 H2O +2 = Oxidation 58 Try These!! +1 = Fe 2+ is oxidized 5 Fe2+ + MnO4- + 8 H+ 5 Fe3+ + Mn2+ + 4 H2O - 5 = Mn +2 = Zn 0 7+ is reduced is oxidized Zn + 2 HCl ZnCl2 + H2 - 1 = H 1+ is reduced 59 How to write net ionic equations • 1) write a balanced equation (aq) Cu(s) + 2NaCl(aq) 2Na(s) + CuCl2 2) Ionize any aqueous substances Cu(s) + 2Na1+(aq) 2Cl1-(aq) 2Na(s) + Cu2+ (aq) 2Cl 1- (aq) 3) Remove any like substances (spectators) Cu(s) + 2Na1+(aq) 2Cl1-(aq) 2Na(s) + Cu2+ (aq) 2Cl 1- (aq) 4) Sum up what’s left Cu(s) + 2Na1+(aq) 2Na(s) + Cu2+ (aq) The Net Ionic Equation (the reaction that60 is really occurring) Oxidation Reduction Reaction Synthesis • Reaction that involves a transfer of electrons. • Presence of oxygen **hint** 2Mg(s) + O2(g) 2MgO(s) MgMg2+ + 2eoxidation O + 2e- O2reduction Most REDOX reactions used for energy production Thermite Reaction (single replacement) Fe2O3(s) + 2Al(s) 2Fe(l) + Al2O3(s) Al Al3+ +3e- Fe3+ 3e- Fe (molten) Production of Bleach Cl2 + 2 NaOH → NaCl + NaClO + H2O Identify the oxidize and reduced species What do you notice different about this reaction? Hence, chlorine is simultaneously reduced and oxidized; this process is known as disproportionation. Balancing Equations with the Half-Reaction Method 1) Identify what is oxidized and what is reduced. Split the equation into two half-reactions. In each half-reaction, follow these steps: 2) Balance all elements except “H” and “O”. 3) Balance the “O’s” by adding water, H2O. 4) Balance the “H’s” by adding hydrogen ions, H+. 5) Balance the charges by adding electrons. If necessary, multiply one or both half reactions by an integer to equalize the number of electrons transferred in the two half-reactions. 6) Recombine the ½ reactions into a complete balanced equation, cancel identical species. 64 Example: Fe2+ + Cr2O72- Fe3+ + Cr3+ acidic solution 6( Fe2+ 21( 6 e-+ 14 H+ + Cr2O7 Fe3+ + 1e- ) 2Cr3+ + 7 H2O) Cr2O72- + 6 Fe2+ + 14 H+ 2 Cr3+ + 6 Fe3+ + 7 H2O 65 Table 12.1 Strength of oxidizing and reducing agents Inquiry into Chemistry Chapter 12 Oxidizing Agent Reduction Oxidation Reducing Agent Stronger Oxidizing Agent Cu 2+ Cu Zn 2+ Zn Stronger Reducing Agent 66 Oxidation Reduction Strongest Oxidizing Agent Weakest Reducing Agent Ba 2+ (aq) Ba (s) Ca 2+ (aq) Ca (s) Mg 2+ (aq) Mg (s) Al 3+ (aq) Al (s) Zn 2+ (aq) Zn (s) Cr 3+ (aq) Cr (s) Fe 2+ (aq) Fe (s) Cd 2+ (aq) Cd (s) Tl + (aq) Tl (s) Co 2+ (aq) Co (s) Ni 2+ (aq) Ni (s) Sn 2+ (aq) Sn (s) Cu 2+ (aq) Cu (s) Hg 2+ (aq) Hg (s) Ag 2+ (aq) Ag (s) Pt 2+ (aq) Pt (s) Au 1+ (aq) Au (s) Weakest Oxidizing Agent Strongest Reducing Agent 67 Spontaneous Reaction Compare Reducing Agents Loses 2 e Pt (s)+ Sn 2+ (aq) Pt 2+ (aq)+ Sn (s) Gains 2 eStronger Reducing Agent Stronger Oxidizing Agent Compare Oxidizing Agents 68 Non Spontaneous Reaction Compare Reducing Agents Loses 2 e - Mg (s) + Fe2+ (aq) Mg 2+ (aq) + Fe (s) Gains 2 eCompare Oxidizing Agents Stronger Oxidizing Agent Stronger Reducing Agent 69 What if the solution was basic? Notice that the method has assumed the solution was acidic - we added H+ to balance the equation. The [H+] in a basic solution is very small. The [OH-] is much greater. For this reason, we will add enough OH- ions to both sides of the equation to neutralize the H+ added in the reaction. The hydrogen and hydroxide ions will combine to make water, and you may have to do some canceling before you’re done. Cr2O72- + Fe2+ + H2O Cr3+ + Fe3+ Try this in a basic solution!!! 70 Cr2O72- + Fe2+ + H2O Cr3+ + Fe3+ Basic Solution 6 ( Fe2+ ) 1(6 e-+ 14OH 14 H-214H O + ++ ) Cr2O72- Fe3+ + 1e2 Cr3+ + 7 H2O + 14OH- Cr2O72- + 6 Fe2+ + 7 H2O 2 Cr3+ + 6 Fe3+ + 14 OH- 71 Balancing Redox Equations Practice Balance in acidic solution: H2C2O4 + MnO4- Mn2+ + CO2 5 H2C2O4 + 2 MnO4- + 6 H+ 2 Mn2+ + 10 CO2 + 8 H2O Balance in basic solution: CN- + MnO4- CNO- + MnO2 3 CN- + 2 MnO4- + H2O 3 CNO- + 2 MnO2 + 2 OH- 72 Chemical reactions Precipitation Reactions (Double replacement) Oxidation-Reduction reactions Combustion reactions Single Replacement Acid-Base Reactions (Double Replacement) Decomposition reactions Synthesis reactions