* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Meandering Right Pulmonary Vein Simulating the Scimitar Syndrome*
Survey
Document related concepts
Electrocardiography wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Heart failure wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Myocardial infarction wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Coronary artery disease wikipedia , lookup
Cardiac surgery wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Atrial septal defect wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Transcript
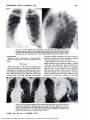
GOODMAN, JAMSHIDI, HIPONA 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 paralysis in left ventricular failure. Am J Med Sci 188: 691-697, 1934 Tashneck AB: Left recurrent laryngeal nerve paralysis in patient with arteriosclerotic heart disease. Southern Med J, 46:718-719, 1953 Zelman S, Nice GW: Recurrent left laryngeal nerve paralysis in atherosclerotic heart disease. JAMA 149: 12911292, 1952. Huppler EG, Schmidt H, Devine KD, et al: Causes of vocal cord paralysis. Proc Staff Meet Mayo Clin 30:518521, 1955 Erlanger H, Levine SA: Atrial septa1 defect: A report of two cases in which there was recurrent laryngeal nerve paralysis. Am Heart J 26:520-527, 1943 Dolowitz DA, Lewis CS: Left vocal cord paralysis associated with cardiac disease. Am J Med 4:856-862, 1948 Mead KC: Persistent patency of the ductus arteriosus. JAMA 50:2205-2210, 1910 Talley JD, Fowler K: Tetralogy of Fallot with hypoplasia of dextroposed aorta. Am J Med Sci 191:618-626, 1936 Camishion RC, Gibbon JH, Pierucci L, et al: Paralysis of the left recurrent laryngeal nerve secondary to mitral valve disease: Report of two cases and literature review. Ann Surg 163:818-827, 1960 Fife R, Miur A: Laryngeal paralysis associated with mitral stenosis. ( Ortner's syndrome ) : Report of two cases. Glasgow Med J 36: 184-167, 1955 Malcomson K, Hillman LM: Ortner's syndrome. Guy Hosp Rep 105:307-319, 1956 Fetterolf G , Nonis GW: The anatomical explanation of the paralysis of the left recurrent laryngeal nerve found in certajn cases of mitral stenosis. Am J Med Sci 141:625638, 1911 Ari R, Harvey WP, Hufnagel CA: Etiology of hoarseness associated with mitral stenosis: Improvement following mitral surgery. Am Heart J 50: 153-160,1955 Brinton WD: Primary pulmonary hypertension. Br Heart J 12:305-311, 1950 Soothill JF: A case of primary pulmonary hypertension with paralyzed left vocal cord. Guy Hosp Rep 100:232237, 1951 Bohme Von H, Bohme G: Das Ortner-Syndrome. Munchen Med Wschr 104:681-887, 1962 Pamell FW, Brandenburg JH: Vocal cord paralysis: A review of 100 cases. Laryngoscope 80: 1036-45, 1970 Clerf LH: Unilateral vocal cord paralysis. JAMA 151:900903, 1953 Goff WF: Letter to the Editor. JAMA 212:1378-1379, 1970 Meandering Right Pulmonary Vein Simulating the Scimitar Syndrome* Lawrence R. Goodman, M.D.,OV Ahmd Jamshidi, M.D.,t and Florencio A. Hipona, M.D.1 'From the Department of Medicine, Cardiovascular Laboratory, Bridgeport Hospital, Bridgeport, Connecticut and the Department of Radiology, The Boston City Hospital, Boston. ''Resident in Radiology, Boston University Medical Center. tAssistant Clinical Professor in Pediatrics, Yale University School of Medicine; Director, Cardiovascular Laboratory, Bridgeport Hospital. :Associate Professor of Radiology, Harvard Medical School. Reprint requests: Dr. Hipona, Department of Radiology. Boston City Hospital, Boston 02118 A 51-year-old woman presented with clinical and chest x-ray film findings simulating partial anomalous pulmonary venous rebelow the diaphragm (scimitar syndrome). Angiography, however, revealed the scimitm vein turned cephalad at the diaphragm and draiied into the left side of the left alrium. It is postulated that the abnormal left atrial drainage is via persistent thebestan veins. 0 n frontal chest x-ray film, a large pulmonary vein running caudad and medially along the right heart border to the diaphragm is referred to as a scimitar sign. This commonly indicates partial anomalous pulmonary venous return to the inferior vena cava or low right atrium. Associated findings in the scimitar syndrome include displacement of the heart to the right, abnormal segmentation of the right lung, hypoplastic right hemithorax and hypoplastic right pulmonary artery. Varicosities of the pulmonary veins or arteriovenous malformations may cause a scimitar-like vein adjacent to the right This communication describes a patient whose condition simulated the classic scimitar syndrome except that the right pulmonary vein, after a meandering course, drained into the left atrium rather than the inferior vena cava. A 51-year-old woman was admitted to the Bridgeport Hospital for episodes of coughing small amounts of bright red blood every several weeks over a 20-year period. The last episode had been three weeks prior to admission. These episodes were associated with difficult breathing when lying in the left lateral decubitus position. For the last 15 years, the patient had had multiple episodes of right-sided pleurisy, fever and productive cough. Recurrent asthmatic attacks required use of bronchodilators. X-ray films of this period described a large tubular density over the right mid-lung, as well as right pleural thickening. Past medical history revealed one bout of hematemesis about 12 years previously. This was believed due to a "stomach ulcer." Physical examination was unremarkable except for dullness and decreased breath sounds at the right base. Frontal chest x-ray 6lm (Fig 1A) revealed dextroposition of a normal-sized heart. There was pleural thickening at the right base. A 9 mm diameter vessel was seen adjacent to the right heart border. The main right pulmonary artery a p peared small. On the lateral film (Fig 1B) the 9 mm vessel was seen posteriorly overlapping the vertebral column. A p parently, continuous with this vessel was a 12 mm vessel coursing upward and forward to the upper posterior heart border. Right anterior oblique examination c o n h e d their continuity. Angiography (Fig 2A) showed the right main pulmonary artery to be smaller than the left. A lingular artery was present on the right (mirror image circulation). The levophase (Fig 2B, C ) demonstrated the posterior vessel (described above) drained the entire right lung toward the diaphragm. Instead of piercing the diaphragm, it was continuous with the larger mid-thoracic vessel. This vein drained cephalad, crossed the mediastinum and entered the left side CHEST, VOL. 62, NO. 4, OCTOBER, 1972 Downloaded From: http://publications.chestnet.org/pdfaccess.ashx?url=/data/journals/chest/21547/ on 05/03/2017 MEANDERING RIGHT PULMONARY VEIN FIGURE1A. Frontal chest film shows dextroposed normal-sized heart. The right hemithorax and pulmonary artery are small. A 9 m m vessel is seen adjacent to the right heart border (arrow). B. Lateral chest film shows the 9 m m vase1 running posteroinferiorly over the vertebral column (posterior arrow). It is continuous with a 12 mm vessel running anteriosuperiorly (anterior arrow). of the left atrium. Right-sided cardiac catheterization, including bilateral pulmonary wedge pressures and oxygen saturations, was normal. With the exception of the left atrial drainage, this case has all the features of the scimitar syndrome. There is hypoplasia of the right main pulmonary artery and a mirror image lung. The initial course of the draining vein also mimics the scimitar vein. Clinically, dyspnea and recurrent bouts of the right chest pain are commonly associated symptoms. Although the cause of the hemoptysis is uncertain in this case, hemoptysis is common in patients with pulmonary venous vari~osities.~ Embryologically, the trachea and lungs arise as a midline structure from the primitive f o r e g ~ t . A ~ .plexus ~ of channels course through the loose mesenchyme and communicate with both the cardinal and umbilicomesenteric veins. As the pulmonary veins enlarge, the other connections atrophy. If the cardinal or umbilicomesenteric channels are abnormally large, they may persist as abnormal vessels with various aberrant venous connections. It would seem to follow that abnormally FIGURE 2A. Dextro-phase of angiogram shows normal-sized right atrium and ventricle with minimal rotation to the right. The right pulmonary artery is small and shows a lingular artery ( arrow). B and C. Levo-phase shows the entire right lung draining via a posteroinferiorly oriented vein. At the diaphragm, it takes a meandering course upward, crosses the midline and drains into the left side of the left atrium (arrows outline direction of flow). CHEST, VOL. 62, NO. 4, OCTOBER, 1972 Downloaded From: http://publications.chestnet.org/pdfaccess.ashx?url=/data/journals/chest/21547/ on 05/03/2017 large but normally draining vessels might persist in a similar manner. During fetal life, the superior vena cava is continuous with the coronary sinus. Small thebesian veins drain from this area into the left atrium. If the coronary sinus should be atretic or 0ccluded,2.~.gblood drains retrogradely into the left superior vena cava. If the latter atrophies too, dilated thebesian veins drain into the left atrium (foramen of Bochdalek). Aberrant pulmonary veins may join this system at any point. Usually the coronary sinus or left superior vena cava, or occasionally both,*^^*^^ drain the abnormal vessel. If neither is patent, blood drains via the thebesian system into the left atrium. We postulate this type of drainage to explain the aberrant left-sided insertion into the left atrium. Becu et ale and Nelson et a14 emphasize the importance of distinguishing abnormal connections from abnormal drainage. The latter causes the return of oxygenated pulmonary venous blood to the right side of the heart. These left-to-right shunts are often surgically correctable. With abnormal connections, the route back to the left atrium may be circuitous, but hemodynamically is normal. This case illustrates the value of cardiac catheterization in ruling out left-to-right shunts and the value of angiography in assessing the direction of flow. 1 Bender F, Hilgenberg F, Jungehulsing C: Dextrokardie mit Pulmonalvenentransposition bei Partieuer Lungenagenesie. Ztschr Kreiglaufforch 46: 172-179,1957 2 Blake HA, Hill RJ. Manion WC: Anomalous pulmonary venous return. Circulation 32:406-414, 1965 3 Gottesman L, Weinstein A: Varicosities of the pulmonary veins. Dis Chest 35:322-327, 1959 Isolated Lobular Pulmonary Artery Atresia with Systemic Collaterals in Tetralogy of allot* Robert Klint, M.D.; Alexis Hartmnn, Jr., M.D.; Nickohuss Schad, M.D., F.C.C.P.; C h Weldon, M.D.; and D a d Coldring, M.D. Thishad report describes a patient with tetralogy of Fallot absence or hypoplasia of the right upper who lobe pulmonary artery and unusual collateral blood supply to that lobe. The collateral vessel was used to create a systemic-pulmonary artery shunt. The patient was the full-term product of a normal pregnancy, labor and delivery. The child was asymptomatic until 13 months of age when he became cyanotic while crying. He experienced six similar episodes during the next nine months. Physical examination revealed a faintly cyanotic boy in no distress. He demonstrated cardiomegaly, a prominent thrill in the third left intercostal space, a grade IV/VI pansystolic murmur maximal at the second and third left intercostal spaces with radiation into the back, and a diminished second heart sound in the pulmonic area. The nailbeds were faintly cyanotic; clubbing was absent. The electrocardiogram demonstrated right axis deviation 'From the Edward Mallinckrodt Department of Pediatrics Division of Cardiology, Edward Mallinckrodt Institute ol Radiology and the Mary Culver Department of Surgery, Division of Cardiothoracic Surgery, Washinqton University School of Medicine, and St. Louis Children s Hospital, St. Louis. Aided in part by the Arthur Fund, St. Louis Children's Hospital Heart Mothers' Fund, Scott Centsch Memorial Fund, William T. Beauchamp Memorial Fund, and the John Clay Seier Fund. Reprint requests: Dr. Klint, St. Louts Children's Hospital, St. Louts 63110 4 Nelson WP, Hall RJ, Garcia E: Varicosities of the pulmonary veins simulating arteriovenous fistulas. JAMA 195:103107, 1966 5 Vengsarker AS, Kincaid OW, Weidman WH: Selective angiocardiography in diagnosis of varicosities of the pulmonary veins. Am Heart J 88:296-298,1963 6 Becu LM, Tauxe WN, D u Shane JW, et al: Anomalous connection of pulmonary veins with normal pulmonary drainage. Arch Path01 59:463-470.1955 7 Patten B: Human Embryology (3rd ed). New York. McCraw Hill, 1968 8 Fieldstein LE, Pick J: Drainage of the coronary sinus into the left atrium. Am J Clin Path 12:66-89,1942 9 Reed AF: Left superior vena cava draining the blood from a closed coronary sinus. J Anatomy 73:195-197, 1938 10 Nakib A, Moller JH, Kanjuk VI, et al: Anomalies of the pulmonary veins. Am J Cardiol20:77-90, 1967 FIGURE1. PA projection. This roentgenogram shows cardiomegaly, left aortic arch, and inferior displacement of the right main pulmonary artery. CHEST, VOL. 62, NO. 4, OCTOBER, 1972 Downloaded From: http://publications.chestnet.org/pdfaccess.ashx?url=/data/journals/chest/21547/ on 05/03/2017