* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Singapore Scientists Discover Genetic Link in Kawasaki Disease
Survey
Document related concepts
Gene therapy wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Transmission (medicine) wikipedia , lookup
Fetal origins hypothesis wikipedia , lookup
Race and health wikipedia , lookup
Compartmental models in epidemiology wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Seven Countries Study wikipedia , lookup
Epidemiology wikipedia , lookup
Public health genomics wikipedia , lookup
Transcript
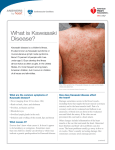
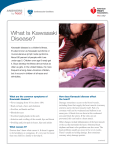
RESEARCH FINDINGS Singapore Scientists Discover Genetic Link in Kawasaki Disease S cientists at the Infectious Diseases department of the Genome Institute of Singapore (GIS), an institute of the Agency for Science, Technology and Research (A*STAR), led a genome-wide association study (GWAS) on a world-wide collaboration on Kawasaki disease. The study, published in Nature Genetics on 13 November 2011, indicates that the gene FCGR2A is associated with an increased risk of contracting the disease. The world-wide investigation, linking five separate consortia, was initiated by GIS, which was responsible for the study design and coordination, as well as the laboratory work and data analysis of samples collected by its research partners. The study examined the genetic profiles of 405 children with Kawasaki disease and contrasted them with 6,252 healthy controls, in Europe, USA and Australia. Genetic markers showing potential association with the disease were re-assessed and validated in a further 740 affected families, as well as a further 1,028 affected children and 1,512 healthy controls from Asian countries. From the study, the scientists noted very strong evidence of increased risk of disease at a gene encoding a protein called FCGR2A. This protein is well known to Kawasaki disease doctors because it is a receptor for intravenous immunoglobulin (IVIG), which is used for the treatment of Kawasaki disease. Their observation highlights the importance of such receptors in the pathogenesis of this inflammatory disease, and thereby provides a biological basis for the use of IVIG for Kawasaki disease treatment. Until now, researchers were unclear as to how or why the infusion of IVIG worked in treating the disease. Co-lead author of the paper, Senior Group Leader and Associate Director for Infectious Diseases at the GIS, Dr Martin Hibberd said, “The cause and events that lead to Kawasaki disease have been difficult mysteries, that are now starting to be revealed, but only because of world-wide efforts such as this.Now we have added a very significant jigsaw piece to the big picture, I hope we can start to impact on patients through the development of early diagnosis and new therapeutic approaches.” Dr Khor Chiea Chuen, co-first author and Research Scientist at the GIS added, “I saw my first patient with Kawasaki disease as a fourth year medical student five years ago, and when asked by the child’s parents regarding the cause of the disease, I didn’t know much apart from the observation that Kawasaki disease could be triggered by an infectious episode. Now, we know more. We plan to extend this study to involve Singaporean patients in the near future.” Kawasaki disease is an autoimmune disease in which patient’s blood vessels throughout the body become inflamed. It affects mostly children, causing prolonged fever of usually more than five days. It is unresponsive to antibiotics and antipyrectics, such as paracetomol and aspirin. A very visible rash develops, and the child is in obvious discomfort, but it cannot be relieved by traditional means. Furthermore, there are other conditions, such as scarlet fever and measles, which can mimic it, and thus delay its diagnosis. Early diagnosis is important, so that IVIG can be administered straightaway. If IVIG is delayed, there is a significant risk of irreversible heart damage. Stem Cells Engineered to Kill Cancer U .S. researchers say they’ve shown that blood stem cells can be engineered to create cancer-killing T-cells that seek out and attack a human melanoma. Jerome Zack -- a scientist with the University of California, Los Angeles, Jonsson Comprehensive Cancer Center and the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research -- said the tests, conducted on mice, prove blood stem cells can be genetically altered in a living organism to fight cancer. “We knew from previous studies that we could generate engineered T-cells, but would they work to fight cancer in a relevant model 44 of human disease, such as melanoma,” Zack said Tuesday in a release. In four of the nine mice studied, the antigen-expressing melanomas were eliminated. In the other five mice, the antigen-expressing melanomas decreased in size. Researcher Dimitrios Vatakis said the approach turned a few engineered stem cells into an army of T-cells that responded to the presence of the melanoma antigen. “These cells can exist in the periphery of the blood and if they detect the melanoma antigen, they can replicate to fight the cancer,” he said. The study appeared in the early online edition of the Proceedings of the National Academy of Sciences.