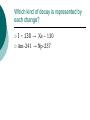* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Nuclear Decay (Radioactivity)
Survey
Document related concepts
Introduction to quantum mechanics wikipedia , lookup
Standard Model wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Future Circular Collider wikipedia , lookup
ATLAS experiment wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Nuclear structure wikipedia , lookup
Elementary particle wikipedia , lookup
Transcript
Nuclear Decay (Radioactivity) Subatomic particles Electron -negatively charged particle found outside the nucleus. Proton – positively charged subatomic particle found inside the nucleus. Neutron – Neutrally charged (no charge) subatomic particle found inside the nucleus. Some Surprises A neutron is a proton with a captured electron. A neutron can become a proton by losing that electron. That electron, when it comes flying out of the nucleus, will be called a beta particle. It’s symbol is the Greek letter β. Beta radiation What would happen to the nuclear make-up as a result of beta radiation? Consider: carbon – 14. -+ = neutron + +-+ + -+ -+ +-++ + -+ + -+ -+ = proton Beta radiation What would happen to the nuclear make-up as a result of beta radiation? Consider: carbon – 14. -+ = neutron + +-+ + -+ -+ +-++ + -+ + -+ - + = proton Beta radiation How many protons does it have now? What is its mass number? -+ = neutron + +-+ + -+ -+ +-++ + -+ + -+ + = proton Alpha Radiation An alpha (α) particle is the same as a helium nucleus and is therefore symbolized by: 42 He -+ = neutron + = proton -+ -+ + + -+ + -+ + + -+ +-+ + -+ -+ -+ + -+ -+ + -+ -+ + ++ + + ++ -+ -+ + -+ + -+ -+ +-+-+ -+ + + -+ ++ + -+ -+ Alpha Decay When an alpha particle is emitted from a nucleus, what is left? Consider Uranium – 238. 238 92 U XTh 234 90 + 4 2 He Which kind of decay is represented by each change? I – 130 → Xe – 130 Am-241 → Np-237 Three Kinds of Decay Alpha – a helium nucleus, cannot penetrate paper. Beta - an electron which is emitted from the nucleus. Can penetrate paper, but cannot penetrate aluminum foil. Gamma ray – not a particle. 50% emitted will penetrate 1cm of lead.